Chemical looping gasification of biomass based on the oxygen carrier derived from the layered double hydroxide (LDH) precursor
-
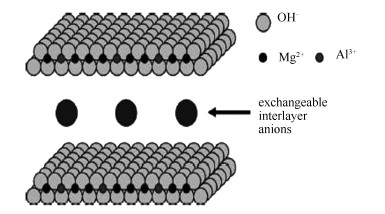
摘要: 通过低饱和共沉淀法合成了类水滑石结构的层状氢氧化物(Layered Double Hydroxide, LDH) 前驱体, 经煅烧获得衍生Cu/Al/Zn、Cu/Al/Ni、Cu/Al/Ni/Zn高分散复合氧载体.采用XRD、XRF、H2-TPR、SEM及BET等分析手段对氧载体的结构及反应性能进行了表征, 并通过固定床反应器开展了氧载体与生物质化学链气化实验.结果表明, 合成的三种前驱体都具有典型的水滑石特征衍射峰, 且层板稳定.Cu/Al/Zn前驱体层间厚度为0.2642nm, Ni2+引入后, 层间距减小.前驱体煅烧后形成的复合氧载体中元素含量与制备试剂基本一致.氧载体中Zn、Ni元素的引入可提升CuO的反应活性, 降低H2还原的反应温度, Zn元素与Cu具有更好的协同作用.Cu/Al/Ni/Zn氧载体在固定床化学链气化中具有较好的碳转化率和气体产率, 其碳转化率为82.03%.反应后氧载体比表面积为5.995m2/g, 具有较好的可再生性与抗烧结性, 是生物质化学链气化反应较为理想的氧载体.Abstract: Layered double hydroxide (LDH) precursors with different metal elements were prepared by co-precipitation method at constant pH value; highly dispersed Cu/Al/Zn, Cu/Al/Ni and Cu/Al/Ni/Zn mixed metal oxygen carriers were obtained by calculation of the corresponding precursors. These oxygen carriers were characterized by XRD, XRF, H2-TPR, SEM and nitrogen adsorption and their activity in the chemical looping gasification (CLG) of biomass was investigated in a fixed bed reactor. The results indicated that typical hydrotalcite structure appears in the three precursors with stable layer board. The interlayer spacing of Cu/Al/Zn precursor is 0.2642nm, larger than that of the Cu/Al/Ni precursor. The oxygen carriers derived from the corresponding precursors display similar elements contents as the preparation reagents. Due to the synergy among various metals, the Cu/Al/Ni/Zn oxygen carrier shows the highest reaction activity and anti-sintering ability. The addition of Ni and Zn has a positive effect on the activity of CuO and reduce its reduction temperature; Zn shows a better synergistic effect than Ni with Cu. The carbon conversion reaches 82.03% for the CLG of biomass with Cu/Al/Ni/Zn as oxygen carrier; the surface area of Cu/Al/Ni/Zn remanis 5.995m2/g after the CLG reaction, suggesting that it could be an ideal candidate for the CLG of biomass.
-
Key words:
- layered double hydroxide /
- hydrotalcite /
- chemical looping gasification /
- oxygen carrier /
- biomass
-
图 2 前驱体及煅烧后产物氧载体的XRD谱图
Figure 2 XRD patterns of the LDH precursor and oxygen carriers
a: Cu/Al/Zn LDH; b: Cu/Al/Ni LDH; c: Cu/Al/Ni/Zn LDH; d: Cu/Al/Zn oxygen carrier; e: Cu/Al/Ni oxygen carrier; f: Cu/Al/Ni/Zn oxygen carrier ◆: Cu/Al/Zn LDH; ▲: Cu/Al/Ni LDH; ●: Cu/Al/Ni/Zn LDH; ○: CuO; ■: Al2O3; △: NiO; ◇: ZnO
表 1 氧载体前驱体晶胞参数
Table 1 Cell parameters of the oxygen carrier precursors
Cell
parameterCu/Al/Zn
LDHCu/Al/Ni
LDHCu/Al/Ni/Zn
LDHa 3.072 0 3.054 0 3.063 b 3.072 0 3.054 0 3.063 c 22.620 22.966 0 22.713 d003 /nm 0.264 2 0.263 2 0.263 7 d006 /nm 0.228 7 0.194 0 0.211 4 d009 /nm 0.171 7 0.152 6 0.162 2 d110 /nm 0.153 6 0.141 8 0.147 7 Cell volume
(106 /pm3)183.56 185.50 184.53 表 2 层状氢氧化物前驱体及相应氧载体的元素分析
Table 2 Elemental analysis of the LDH precursors and corresponding oxygen carrier
Content w/% Cu/Al/Zn LDH Cu/Al/Zn OC Cu/Al/Ni LDH Cu/Al/Ni OC Cu/Al/ Ni/Zn LDH Cu/Al/ Ni/Zn OC O 43.1 27.91 48.72 23.59 59.74 26.22 Zn 18.15 22.56 0.026 5 0.028 4 8.865 18.29 Cu 17.75 21.52 15.7 30.27 8.133 17.06 Na 6.147 13.56 6.383 8.64 3.524 7.373 Al 7.831 13.4 5.362 11.66 1.947 9.973 Ni 0.715 0.937 4 16.91 33.72 9.81 20.71 C 6.216 - 6.834 - 7.925 - Ca 0.021 0.028 0.017 0.084 0.012 0.033 Si 0.015 0.023 0.012 0.470 0.007 0.234 Fe 0.014 0.019 0.012 0.037 0.008 0.021 Mn 0.015 0.015 0.006 0.018 0.009 0.017 note: “-” means the element was undetected 表 3 生物质的元素分析与工业分析
Table 3 Elemental analysis and proximate analysis of biomass
Proximate analysis wdb/% Element analysis wdb/% QLHV/
(MJ·kg-1, db)M V FC A C H O N S 8.39 84.31 6.88 0.42 46.44 6.21 47.29 0.05 0.01 18.707 note: M moisture, V volatile matter, FC fixed carbon, A ash, db dry basis, LHV lower heating value 表 4 合成氧载体生物质化学链气化实验结果
Table 4 Experimental results of biomass CLG on the basis of various oxygen carriers
Item Cu/Al/Zn Cu/Al/Ni Cu/Al/Ni/Zn Fe/Al CH4/% 7.05 7.60 7.14 13.15 C2Hm/% 2.39 1.71 2.34 - CO2/% 24.12 24.01 23.88 18.09 CO/% 32.32 45.43 32.37 47.94 H2/% 34.12 21.25 34.26 20.32 Gas yield/(m3·kg-1) 1.09 0.95 1.12 0.95 QLHV/(MJ·m-3) 11.92 11.88 11.90 12.65 Carbon conversion x/% 80.23 82.16 82.03 78.47 Gasification efficiency η% 69.46 60.36 71.25 64.27 表 5 氧载体生物质化学链气化反应后BET比表面积
Table 5 Surface area of the oxygen carriers after CLG reaction
BET area A/(m2·g-1) Cu/Al/Zn OC Cu/Al/Ni OC Cu/Al/Ni/Zn OC Before 2.480 2.287 3.821 After 5.243 2.828 5.995 -
[1] RICHTER H, KNOCHE K, RICHTER H, KNOCHE K. Reversibility of combustion processes[J]. ACS Symp Ser, 1983, 235(3):71-86. doi: 10.1021/bk-1983-0235.ch003 [2] UDOMSIRICHAKORN J, BASU P, ABDUL SALAM P, ACHARYA B. CaO-based chemical looping gasification of biomass for hydrogen-enriched gas production with in situ CO2 capture and tar reduction[J]. Fuel Process Technol, 2014, 127: 7-12. doi: 10.1016/j.fuproc.2014.06.007 [3] HUANG Z, HE F, FENG Y, ZHAO K, ZHENG A, CHANG S, LI H. Synthesis gas production through biomass direct chemical looping conversion with natural hematite as an oxygen carrier[J]. Bioresour Technol, 2013, 140: 138-145. doi: 10.1016/j.biortech.2013.04.055 [4] TIAN H, FISHER J C. Isotopic steam investigations of hematite (Fe2O3) for chemical looping combustion of methane[J]. Catal Commun, 2015, 67: 83-86. doi: 10.1016/j.catcom.2015.04.015 [5] MEI D, ABAD A, ZHAO H, ADÁNEZ J. Characterization of a sol-gel derived CuO/CuAl2O4 oxygen carrier for chemical looping combustion (CLC) of gaseous fuels: Relevance of gas-solid and oxygen uncoupling reactions[J]. Fuel Process Technol, 2015, 133: 210-219. doi: 10.1016/j.fuproc.2015.02.007 [6] SHEN L H, WU J H, XIAO J. Experiments on chemical looping combustion of coal with a NiO based oxygen carrier[J]. Combust Flame, 2009, 156(3): 721-728. doi: 10.1016/j.combustflame.2008.08.004 [7] FORUTAN H R, KARIMI E, HAFIZI A, RAHIMPOUR M R, KESHAVARZ P. Expert representation chemical looping reforming: A comparative study of Fe, Mn, Co and Cu as oxygen carriers supported on Al2O3[J]. J Ind Eng Chem, 2015, 21: 900-911. doi: 10.1016/j.jiec.2014.04.031 [8] ADáNEZ-RUBIO I, GAYÁN P, ABAD A, DE DIEGO L F, GARCÍA-LABIANO F, ADÁNEZ J. Evaluation of a spray-dried CuO/MgAl2O4 oxygen carrier for the chemical looping with oxygen uncoupling process[J]. Energy Fuels, 2012, 26(5): 3069-3081. doi: 10.1021/ef3002229 [9] SIRIWARDANE R, TIAN H, MILLER D, RICHARDS G. Fluidized bed testing of commercially prepared MgO-promoted hematite and CuO-Fe2O3 mixed metal oxide oxygen carriers for methane and coal chemical looping combustion[J]. Appl Energy, 2015, 157: 348-357. doi: 10.1016/j.apenergy.2015.04.042 [10] LI K Z, WANG H, WEI Y G, YAN D X. Direct conversion of methane to synthesis gas using lattice oxygen of CeO2-Fe2O3 complex oxides[J]. Chem Eng J, 2010, 156 (3): 512-518. doi: 10.1016/j.cej.2009.04.038 [11] 刘洁翔, 张晓光.阴离子插层镁铝水滑石结构及相互作用的理论研究[J].燃料化学学报, 2013, 41(6): 761-768. http://rlhxxb.sxicc.ac.cn/CN/Y2013/V41/I06/761LIU Jie-xiang, ZHANG Xiang-guang. Theoretical investigation on the structure of anions intercalated MgAl-layered double hydroxides and the interaction between anions and host layer[J]. J Fuel Chem Technol, 2013, 41(6): 761-768. http://rlhxxb.sxicc.ac.cn/CN/Y2013/V41/I06/761 [12] WANG L, DALIN L, WATANABE H, TAMURA M, NAKAGAWA Y, TOMISHIGE K. Catalytic performance and characterization of Co/Mg/Al catalysts prepared from hydrotalcite-like precursors for the steam gasification of biomass[J]. Appl Catal B: Environ, 2014, 150: 82-92. https://www.researchgate.net/publication/259518080_Catalytic_performance_and_characterization_of_CoMgAl_catalysts_prepared_from_hydrotalcite-like_precursors_for_the_steam_gasification_of_biomass [13] SONG Q L, LIU W, CHRISTOPHER D B, RYAN N H, EASAN S, STUART A S, JOHN S D. A high performance oxygen storage material for chemical looping processes with CO2 capture[J]. Energy Environ Sci, 2013, 6(1): 288-298. doi: 10.1039/C2EE22801G [14] LONG H, XU Y, ZHANG X Q, HU S J, SHANG S Y, YIN Y X, DAI X Y. Ni-Co/Mg-Al catalyst derived from hydrotalcite-like compound prepared by plasma for dry reforming of methane[J]. J Energy Chem, 2013, 22(5):733-739. doi: 10.1016/S2095-4956(13)60097-2 [15] LI B S, YUAN S L. Synthesis, characterization, and evaluation of TiMgAlCu mixed oxides as novel SOx removal catalysts[J]. Ceram Int, 2014, 40(8): 11559-11566. doi: 10.1016/j.ceramint.2014.03.112 [16] GALINDO R, LÓPEZ-DELGADO A, PADILLA I, YATES M. Hydrotalcite-like compounds: A way to recover a hazardous waste in the aluminium tertiary industry[J]. Appl Clay, 2014, 95: 41-49. doi: 10.1016/j.clay.2014.03.022 [17] BHUIYAN M R, LIN S D, HSIAO T C. Effect of calcination on Cu-Zn-loaded hydrotalcite catalysts for C-C bond formation derived from methanol[J]. Catal Today, 2014, 226: 150-159. doi: 10.1016/j.cattod.2013.10.053 [18] DALIN L, KOIKE M, CHEN J H, NAKAGAWA Y, TOMISHIGE K. Preparation of Ni-Cu/Mg/Al catalysts from hydrotalcite-like compounds for hydrogen production by steam reforming of biomass tar[J]. Int J Hydrogen Energy, 2014, 39(21): 10959-10970. doi: 10.1016/j.ijhydene.2014.05.062 [19] SIRIWARDANE R, TIAN H J, SIMONYI T, POSTON J. Synergetic effects of mixed copper-iron oxides oxygen carriers in chemical looping combustion[J]. Fuel, 2013, 108: 319-333. doi: 10.1016/j.fuel.2013.01.023 [20] HUANG Z, HE F, FENG Y P, ZHAO K, ZHENG A Q, CHANG S, WEI G Q, ZHAO Z L, LI H B. Biomass char direct chemical looping gasification using NiO-modified iron ore as an oxygen carrier[J]. Energy Fuels, 2014, 28(1): 183-191. doi: 10.1021/ef401528k [21] 朱锡锋.生物质热解原理与技术[M].合肥:中国科学技术大学出版社, 2006.ZHU Xi-feng. Biomass Pyrolysis Principle and Technology[M]. Hefei: University of Science and Technology of China Press, 2006. [22] LUCRÉDIO A F, ASSAF J M, ASSAF E M. Reforming of a model sulfur-free biogas on Ni catalysts supported on Mg (Al) O derived from hydrotalcite precursors: Effect of La and Rh addition[J]. Biomass Bioenergy, 2014, 60: 8-17. doi: 10.1016/j.biombioe.2013.11.006 [23] GAO P, LI F, XIAO F K, ZHAO N, WEI W, ZHONG L S, SUN Y H. Effect of hydrotalcite-containing precursors on the performance of Cu/Zn/Al/Zr catalysts for CO2 hydrogenation: Introduction of Cu2+ at different formation stages of precursors[J]. Catal Today, 2012, 194(1): 9-15. doi: 10.1016/j.cattod.2012.06.012 -





 下载:
下载: