Effect of kaolin on the ash fusion characteristics of high alkali Zhundong coal: A quantum chemistry and experimental study
-
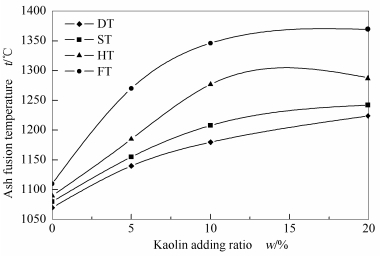
摘要: 采用量子化学理论计算与实验表征相结合的方法, 研究了高岭土对准东高碱煤煤灰熔融特性及其熔融过程中矿物质演变规律的影响, 并从矿物质微观结构特性角度阐述了高岭土对改变准东煤煤灰熔融特性的影响机制。结果表明, 准东高碱煤煤灰熔融温度随高岭土的添加呈现先快速升高后逐渐变缓的趋势, 当高岭土添加比例大于10%时, 其提高煤灰熔融温度的趋势变缓; 准东高碱煤添加高岭土后, 其在1 000-1200 ℃下的低熔融矿物钙长石、硬石膏等量明显减少, 1 200-1 300 ℃下有一定量的莫来石生成, 是其煤灰熔点升高的主要原因; 高岭石分子结构中的O (26)、O (22)、Si (6)、Si (8) 的反应活性较高, 能够与灰中的Fe2+等金属离子成键, 促使高岭石的铝氧键断裂。煤中的碱金属或碱土金属(Na或Ca) 氧化物中的的O2-, 作为亲核试剂, 与高岭石的Si (6) 和Si (8) 发生亲核反应, 使桥氧键S-O-Si断裂。Abstract: The effect of kaolin on the ash fusion characteristics of high alkali Xinjiang Zhundong coal was investigated by quantum chemistry calculation and experimental measurement methods. The results show that the ash fusion temperature is increased significantly by adding kaolinite; the ash fusion temperature increases rapidly at first with the increase of kaolinite content added in the Zhundong coal and then levels off when the fraction of kaolinite exceeds 10%. By adding kaolin in the Zhundong coal, the content of minerals with a low melting point (1 100-1 200 ℃), such as anorthite and anhydrite, is reduced, whereas mullite is found at 1 200-1 300 ℃. O (26), Si (6), O (22) and Si (8) atoms in the kaolinite molecular structure exhibit relatively high reactivity; Al-O bond, which is connected with O (26) and O (22), can be ruptured by reacting with Fe2+ or other metal ions in ash as an electrophilic reagent. The O2- of alkali oxides in ash, such as Na2O and CaO, can react as a nucleophilic reagent with Si (6) and Si (8) in kaolinite, breaking the oxygen bridge bond of Si-O-Si in kaolinite.
-
Key words:
- Zhundong coal /
- kaolin /
- density functional theory /
- ash fusion characteristics /
- high alkali coal /
- reactivity
-
表 1 实验煤样与高岭土分析数据
Table 1 Properties of coal and kaolinite
Project Zhundong coal Kaolin Proximate analysis Mar/% 20.48 0.0 Mad/% 8.86 0.0 Aar/% 14.23 100 Vdaf/% 35.98 0.0 Qnet, ar/(MJ·kg-1) 18.3 0.0 Elemental analysis Cdaf w/% 78.77 - Hdaf w/% 4.59 - Odaf w/% 15.06 - Ndaf w/% 0.84 - Sdaf w/% 0.74 - Melting temperature (reducing atmosphere) DT t/℃ 1 070 > 1 500 ST t/℃ 1 080 > 1 500 HT t/℃ 1 090 > 1 500 FT t/℃ 1 110 > 1 500 Chemical compositions of coal ash SiO2 w/% 35.95 61.99 Al2O3 w/% 14.60 21.73 Fe2O3 w/% 16.32 1.39 CaO w/% 11.72 0.61 MgO w/% 5.69 0.48 K2O w/% 0.78 0.61 Na2O w/% 4.46 0.21 TiO2 w/% 0.96 0.59 SO3 w/% 9.10 0.83 表 2 高岭石Mulliken原子布居数及简缩Fukui函数(f -和f+)
Table 2 Atomic charges (q) and condensed Fukui Functions (f- and f+) of Kaolinite
Atom q f- f+ atom q f- f+ Al (1) 1.441 0.047 0.058 O (18) -0.967 0.011 -0.023 Al (2) 1.463 0.044 0.047 O (19) -0.793 0.042 -0.020 Al (3) 1.441 0.047 0.058 O (20) -0.775 0.056 -0.010 Al (4) 1.464 0.044 0.047 O (21) -0.811 0.037 -0.008 Si (5) 1.884 0.036 0.158 O (22) -0.787 0.061 -0.013 Si (6) 1.853 0.037 0.168 O (23) -0.793 0.042 -0.020 Si (7) 1.884 0.036 0.158 O (24) -0.775 0.055 -0.010 Si (8) 1.853 0.037 0.168 O (25) -0.811 0.037 -0.008 O (9) -0.994 0.017 -0.015 O (26) -0.787 0.062 -0.013 O (10) -0.982 0.017 -0.014 H (27) 0.335 0.012 0.076 O (11) -0.992 0.012 -0.023 H (28) 0.337 0.017 0.050 O (12) -0.952 0.015 -0.030 H (29) 0.393 0.015 0.032 O (13) -0.967 0.011 -0.023 H (30) 0.347 0.025 0.064 O (14) -0.994 0.017 -0.015 H (31) 0.335 0.012 0.077 O (15) -0.982 0.017 -0.014 H (32) 0.337 0.017 0.050 O (16) -0.992 0.012 -0.022 H (33) 0.393 0.015 0.032 O (17) -0.952 0.015 -0.030 H (34) 0.347 0.025 0.065 -
[1] 杨忠灿, 刘家利, 何红光.新疆准东煤特性研究及其锅炉选型[J].热力发电, 2010, 39(8): 38-40. http://www.cnki.com.cn/Article/CJFDTOTAL-RLFD201008013.htmYANG Zhong-can, LIU Jia-li, HE Hong-guang. Study on properties of Zhundong coal in xinjiang region and type-selection for boilers burning this coal sort[J]. Therm Power Gen, 2010, 39(8): 38-40. http://www.cnki.com.cn/Article/CJFDTOTAL-RLFD201008013.htm [2] ZHOU J B, ZHUANG X G, ALASTUEY A, QUEROLD X, LI J H. Geochemistry and mineralogy of coal in the recently explored Zhundong large coal field in the Junggar basin, Xinjiang province, China[J]. Int J Coal Geol, 2010, 82(1): 51-67. https://www.researchgate.net/publication/238379445_Geochemistry_and_mineralogy_of_coal_in_the_recently_explored_Zhundong_large_coal_field_in_the_Junggar_basin_Xinjiang_province_China [3] 范建勇.准东煤结渣特性及其配煤灰熔融性试验研究[D].杭州:浙江大学, 2014. http://cn.bing.com/academic/profile?id=5196f1c98871c2d7cf78821122ac2fd4&encoded=0&v=paper_preview&mkt=zh-cnFAN Jian-yong. Experimental research about Zhundong coal slagging characteristics and its coal blending ash fusion[D]. Hangzhou: Zhejiang University, 2014. http://cn.bing.com/academic/profile?id=5196f1c98871c2d7cf78821122ac2fd4&encoded=0&v=paper_preview&mkt=zh-cn [4] 岑可法, 樊建人, 池作和, 沈珞婵.锅炉和热交换器的积灰、结渣、磨损和腐蚀的防止原理与计算[M].北京:科学出版社, 1993: 237-265.CEN Ke-fa, FAN Jian-ren, CHI Zuo-he, SHEN Luo-chan. Boiler and Heat Exchanger Product Ash, Slag, Wear and Corrosion Prevention Principle and Calculation[M]. Beijing: Science Press, 1993: 237-265. [5] 姜英.动力煤和动力配煤[M].北京:化学出版社, 2011: 65-67.JIANG Ying. Power Coal and Power Coal Blending[M]. Beijing: Chemical Industry Press, 2011: 65-67. [6] 王勤辉, 景妮洁, 骆仲泱, 李小敏, 揭涛.灰成分影响煤灰烧结温度的实验研究[J].煤炭学报, 2010, 35(6): 1015-1020. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201006035.htmWANG Qin-hui, JING Ni-jie, LUO Zhong-yang, LI Xiao-min, JIE Tao. Experiments on the effect of chemical components of coal ash on the sintering temperature[J]. J China Coal Soc, 2010, 35(6): 1015-1020. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201006035.htm [7] 杨建国, 邓芙蓉, 赵虹, 岑可法.煤灰熔融过程中的矿物演变及其对灰熔点的影响[J].中国电机工程学报, 2006, 26(17): 122-126. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC200617021.htmYANG Jian-guo, DENG Fu-rong, ZHAO Hong, CEN Ke-fa. Mineral conversion of coal-ash in fusing process and the influence to ash fusion point[J]. Proc CSEE, 2006, 26(17): 122-126. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC200617021.htm [8] 唐黎华, 王福明, 朱学栋, 吴勇强, 朱子彬.煤焦中矿物质行为与灰熔融温度的关系[J].华东理工大学学报, 2003, 29(3): 243-247. http://www.oalib.com/references/19425655TANG Li-hua, WANG Fu-ming, ZHU Xue-dong, ZHU Zi-bin. Relationship between mineral behavior in coke and ash melting temperature[J]. J East China Univ Sci Technol, 2003, 29(3): 243-247. http://www.oalib.com/references/19425655 [9] 代百乾, 乌晓江, 陈玉爽, 张忠孝.煤灰熔融行为及其矿物质作用机制的量化研究[J].动力工程学报, 2014, 34(1): 70-76. http://www.cnki.com.cn/Article/CJFDTOTAL-DONG201401012.htmDAI Bai-qian, WU Xiao-jiang, CHEN Yu-shuang, ZHANG Zhong-xiao. Experimental study and quantum chemistry calculation on coal ash fusion characteristics and mineral reaction mechanism[J]. J Chin Soc Power Eng, 2014, 34(1): 70-76. http://www.cnki.com.cn/Article/CJFDTOTAL-DONG201401012.htm [10] LINJEWILE T M, MANZOORI A R. Role of additives in controlling agglomeration and defluidization during fluidized bed combustion of high-sodium, high-sulphur low-rank coal[J]. Eng Fund Conf, 2007, 11: 2-7. doi: 10.1007/0-306-46920-0_24 [11] 李洁, 杜梅芳, 闫博, 张忠孝.添加硼砂助熔剂煤灰熔融性的量子化学与实验研究[J].燃料化学学报, 2008, 36(5): 519-523. doi: 10.1016/S1872-5813(08)60032-8LI Jie, DU Mei-fang, YAN Bo, ZHANG Zhong-xiao. Quantum and experimental study on coal ash fusion with borax fluxing agent[J]. J Fuel Chem Technol, 2008, 36(5): 519-523. doi: 10.1016/S1872-5813(08)60032-8 [12] 洪汉烈, 铁丽云, 闵新民, 肖睿娟, 周泳, 边秋娟.高岭石矿物表面化学的量子化学研究[J].武汉理工大学学报, 2005, 27(1): 25-29. http://www.cnki.com.cn/Article/CJFDTOTAL-WHGY200501008.htmHONG Han-lie, TIE Li-yun, MIN Xin-min, XIAO Rui-juan, ZHOU Yong, BIAN Qiu-juan. Surface chemistry of kaolinite by quantum chemistry calculations[J]. J Wuhan Univ Technol, 2005, 27(1): 25-29. http://www.cnki.com.cn/Article/CJFDTOTAL-WHGY200501008.htm [13] FORESMAN J B, FRISCH A. Exploring chemistry with electronic structure methods (2nd ed). Gaussian, Inc, Pittsburg, PA, 1996. [14] 陈玉爽, 张忠孝, 乌晓江, 李洁, 管荣清, 闫博.配煤对煤灰熔融特性影响的实验与量化研究[J].燃料化学学报, 2009, 37(5): 521-526. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17485.shtmlCHEN Yu-shuang, ZHANG Zhong-xiao, WU Xiao-jiang, LI Jie, GUAN Rong-qing, YAN Bo. Quantum chemistry calculation and experimental study on coal ash fusion characteristics of blend coal[J]. J Fuel Chem Technol, 2009, 37(5): 521-526. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17485.shtml [15] PARR R G, YANG W. Density Functional approach to the frontier-electron theory of chemical reactivity[J]. J Am Chem Soc, 1984, 106: 4049-4050. doi: 10.1021/ja00326a036 [16] 付蓉, 卢天, 陈飞武.亲电取代反应中活性位点预测方法的比较[J].物理化学学报, 2014, 30(4): 628-639. http://www.cnki.com.cn/Article/CJFDTOTAL-WLHX201404004.htmFU Rong, LU Tian, CHEN Fei-wu. Comparing methods for predicting the reactive site of electrophilic substitution[J]. Acta Phys-Chim Sin, 2014, 30(4): 628-639. http://www.cnki.com.cn/Article/CJFDTOTAL-WLHX201404004.htm [17] NNABUK O E, STANISLAV R S, ENO E E. Fluoroquinolones as corrosion inhibitors for mild steel in acidic medium; experimental and theoretical studies[J]. Int J Electrochem Sci, 2010, 5: 1127-1150. https://www.researchgate.net/publication/228507114_Fluoroquinolones_as_Corrosion_Inhibitors_for_Mild_Steel_in_Acidic_Medium_Experimental_and_Theoretical_Studies [18] STOYANOV S R, GUSAROV S, KUZNICKI M S, KOVALENKO A. Theoretical modeling of zeolite nanoparticle surface acidity for heavy oil upgrading[J]. J Phys Chem, 2008, 112(17): 6794-6810. doi: 10.1021/jp075688h [19] STOYANOV S R, GUSAROV S, KOVALENKO A. Modelling of thiophene and benzene adsorption on Cu2+ and Ag+ exchanged chabazite surface. In 'Theoretical Aspect of Catalysis'. Eds. G.Vayssilov and T. Mineva; Heron press, Sofia, Bulgaria, 2008. [20] YANG W, MORTIER W J. The use of global and local molecular parameters for the analysis of the gas-phase basicity of amines[J]. J Am Chem Soc, 1986, 108(19): 5708-5711. doi: 10.1021/ja00279a008 [21] BRINDLEY G W, ROBINSON K. The structure of kaolinite[J]. Miner Mag, 1946, 31: 781-786. http://www.minersoc.org/pages/Archive-MM/Volume_27/27-194-242.htm [22] PERDEW J P. Unified theory of exchange and correlation beyond the local density approximation. Ziesche P, Esching H. Electronic Structure of Solids' 91.Berlin, Akademic Verlag, 1991: 11-20. [23] PERDEW J P, WANG Y. Accurate and simple analytic representation of the electron-gas correlation energy[J]. Phys Rev B, 1992, 45(23): 13244-13249. doi: 10.1103/PhysRevB.45.13244 [24] WANG Y, PERDEW J P. Spin scaling of the electron-gas correlation energy in the high-density limit[J]. Phys Rev B, 1991, 43(11): 8911-8916. doi: 10.1103/PhysRevB.43.8911 [25] POLITZER P, MURRAY J S. The electrostatic potential as a guide to molecular interactive behavior[C]// In Chemical Reactivity Theory: A Density Functional View. CRC Press: Boca Raton, 2009. [26] MICHAELIAN K H, YARIV S, NASSER A. Study of the interaction between caesium bromide and kaolinite by photoacoustic and fiffuse reflectance infrared spectroscopy[J]. Can J Chem, 1991, 69: 749-754. doi: 10.1139/v91-110 [27] 张利孟, 董信光, 刘科, 谭厚章, 王学斌, 魏博.高岭土对准东煤结渣特性及矿物质演变的影响[J].燃料化学学报, 2015, 43(10): 1176-1181. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18706.shtmlZHANG Li-meng, DONG Xin-guang, LIU Ke, TAN Hou-zhang, WANG Xue-bin, WEI Bo. Effect of kaolin on ash slagging and mineral conversion of Zhundong coal[J]. J Fuel Chem Technol, 2015, 43(10): 1176-1181. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18706.shtml [28] 马永静.矿物学角度研究添加剂对煤灰熔融性的作用及其机理[D].太原:太原理工大学, 2009.MA Yong-jing. Study the Effect of additives on the fusibility of coal ash and its mechanism from a mineralogical point of view[D]. Taiyuan: Taiyuan University of Technology, 2009. [29] 沈铭科, 邱坤赞, 黄镇宇, 王智化, 刘建忠.准东煤掺烧高岭土对固钠率及灰熔融特性影响研究[J].燃料化学学报, 2015, 43(9): 1044-1051. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18688.shtmlSHEN Ming-ke, QIU Kun-zan, HUANG Zhen-yu, WANG Zhi-hua, LIU Jian-zhong. Influence of kaolin on sodium retention and ash fusion characteristic during combustion of Zhundong coal[J]. J Fuel Chem Technol, 2015, 43(9): 1044-1051. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18688.shtml [30] KYI S, CHADWICK B L. Screening of potential mineral additives for use as fouling preventatives in Victorian brown coal combustion[J]. Fuel, 1999, 78(7): 845-855. doi: 10.1016/S0016-2361(98)00205-1 [31] 李勇, 肖军.燃煤过程中碱金属赋存迁移规律及相关研究进展[J].洁净煤技术, 2005, 11(1): 39-44. http://www.cnki.com.cn/Article/CJFDTOTAL-JJMS20050100A.htmLI Yong, XIAO Jun. The occurrence and migration mechanism of alkali metal during coal-fired process and research progress[J]. Clean Coal Technol, 2005, 11(1): 39-44. http://www.cnki.com.cn/Article/CJFDTOTAL-JJMS20050100A.htm -





 下载:
下载: