Effect of heat treatment time on the performance of CuZnAl catalysts in the synthesis of higher alcohols from syngas
-
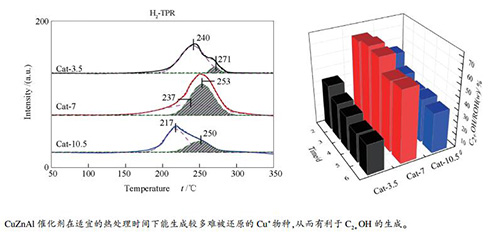
摘要: 采用完全液相法在不同热处理时间下制备了CuZnAl催化剂,利用X射线光电子能谱(XPS)、X射线衍射(XRD)、H2程序升温还原(H2-TPR)、NH3吸附-脱附(NH3-TPD-MS)和N2物理吸附-脱附等方法对其结构进行了表征分析,并在浆态床反应器上对其催化合成气制C2+OH的性能进行了研究。研究发现,延长热处理时间增强了催化剂中Cu和Al物种之间的相互作用力,改变了其中Cu+的量,从而影响Cu+-Cu0活性位的协同作用。同时,热处理时间的延长减少了催化剂的表面酸量,增大了孔容和孔径;催化剂表面较少的弱酸位及较大孔容和孔径均有利于C2+OH的生成。热处理时间为7 h时所制备的CuZnAl催化剂表现出了优良的低碳醇合成催化活性,CO转化率和总醇中C2+OH的质量分数分别达到了38.1%和65.9%。Abstract: A series of CuZnAl catalysts were prepared by the complete liquid-phase method with different heat treatment times and characterized by XPS, XRD, H2-TPR, NH3-TPD-MS and N2 adsorption-desorption; their catalytic performances in the synthesis of higher alcohols from syngas were investigated in a slurry bed reactor. The results indicate that an increase in the heat treatment time can enhance the interaction between the Cu and Al species and alter the amount of Cu+ species over the CuZnAl catalysts, influencing the synergistic effect of Cu+-Cu0 sites. In addition, with the increase of heat treatment time, the surface acidity of CuZnAl catalyst decreases, accompanying with an increase in the pore volume and pore size; small amount of surface weak acid sites, large pore volume and large pore size are beneficial to the formation of higher alcohols. The CuZnAl catalyst obtained by heat-treating for 7 h exhibits excellent performance in the synthesis of higher alcohols, with a CO conversion of 38.1% and a higher alcohols mass fraction of 65.9% in the total alcohols.
-
Key words:
- complete liquid-phase method /
- CuZnAl catalyst /
- heat treatment time /
- higher alcohols /
- Cu+-Cu0
-
图 1 CuZnAl催化剂在不同热处理时间下的催化性能
Figure 1 Performance of CuZnAl catalysts prepared with different heat treatment times in the synthesis of higher alcohols from syngas
(a): CO conversion as a function of reaction time;
(b): higher alcohols mass fraction in total alcohols as a function of reaction time reaction conditions: 280 ℃, 4.0 MPa, V(H2)/V(CO) = 2, feed flow rate = 150 mL/min表 1 CuZnAl催化剂在不同热处理时间下的表面元素组成
Table 1 Surface compositions of the CuZnAl catalysts prepared with different heat treatment times
Catalyst Atom/% Molar ratio C O Cu Zn Al Cu/Zn Cu/Al Zn/Al Cat-3.5 38.3 37.5 1.1 4.5 18.6 0.24 0.06 0.24 Cat-7 22.3 48.7 1.3 4.4 23.4 0.30 0.06 0.19 Cat-10.5 10.5 55.3 1.4 5.5 27.3 0.25 0.05 0.20 表 2 CuZnAl催化剂在不同热处理时间下的织构参数
Table 2 Textural properties of the CuZnAl catalysts with different heat treatment times
Catalyst ABET/(m2·g-1) vBJH/(cm3·g-1) d/nm before reaction after reaction before reaction after reaction before reaction after reaction Cat-3.5 175.4 186.1 0.41 0.44 9.4 10.0 Cat-7 168.9 139.2 0.52 0.43 12.6 15.5 Cat-10.5 155.9 129.3 0.54 0.41 15.0 14.1 notes: ABET: BET surface area; vBJH: pore volume; d: average pore diameter -
[1] SUBRAMANI V, GANGWAL S K. A review of recent literature to search for an efficient catalytic process for the conversion of syngas to ethanol[J]. Energy Fuels, 2008, 22(2):117-136. doi: 10.1021-ef700411x/ [2] GUPTA M, SMITH M L, SPIVEY J J. Heterogeneous catalytic conversion of dry syngas to ethanol and higher alcohols on cu-based catalysts[J]. Acs Catal, 2011, 1(6):641-656. doi: 10.1021/cs2001048 [3] LUK H T, MONDELLI C, FERRE D C, STEWART J A, PEREZ R. Status and prospects in higher alcohols synthesis from syngas[J]. Chem Soc Rev, 2017, 46(5):1358-1426. doi: 10.1039/C6CS00324A [4] JAKOBSEN J G, JAKOBSEN M, CHORKENDORFF I, SEHESTED J. Methane steam reforming kinetics for a rhodium-based catalyst[J]. Catal Lett, 2010, 140(3):90-97. doi: 10.1007/s10562-010-0436-7 [5] BAEK S C, BAE J W, CHEON J Y, JUN K W, LEE K Y. Combined steam and carbon dioxide reforming of methane on Ni/MgAl2O4:Effect of CeO2 promoter to catalytic performance[J]. Catal Lett, 2011, 141(2):224-234. doi: 10.1007/s10562-010-0483-0 [6] MEI D, ROUSSEAU R, KATHMANN S M. Ethanol synthesis from syngas over Rh-based/SiO2 catalysts:A combined experimental and theoretical modeling study[J]. J Catal, 2010, 271(2):325-342. https://www.sciencedirect.com/science/article/pii/S002195171000062X [7] YANG X M, WEI Y, SU Y L, ZHOU L P. Characterization of fused Fe-Cu based catalyst for higher alcohols synthesis and DRIFTS investigation of TPSR[J]. Fuel Process Technol, 2010, 91(9):1168-1173. doi: 10.1016/j.fuproc.2010.03.032 [8] HERACLEOUS E, LIAKAKOU E T, LAPPAS A A, LEMONIDOU A A. Investigation of K-promoted Cu-Zn-Al, Cu-X-Al and Cu-Zn-X (X=Cr, Mn) catalysts for carbon monoxide hydrogenation to higher alcohols[J]. Appl Catal A:Gen, 2013, 455(2):145-154. https://www.sciencedirect.com/science/article/abs/pii/S0926860X1300077X [9] GAO Z H, HAO L F, HUANG W, XIE K C. A novel liquid-phase technology for the preparation of slurry catalysts[J]. Catal Lett, 2005, 102(3/4):139-141. doi: 10.1007/s10562-005-5845-7 [10] HUANG W, YU L M, LI W H, MA Z L. Synthesis of methanol and ethanol over CuZnAl slurry catalyst prepared by complete liquid-phase technology[J]. Front Chem Eng China, 2010, 4(4):472-475. http://d.old.wanfangdata.com.cn/Periodical/zggdxxxswz-hxgc201004016 [11] 喻仕瑞.浆态床合成乙醇Cu基催化剂制备及其性能研究[D].太原: 太原理工大学, 2013.YU Shi-rui. Study on the preparation and performance of Cu-based catalyst for ethanol synthesis in slurry reactor[D]. Taiyuan: Taiyuan University of Technology, 2013. [12] 刘勇军. CuZnAl催化剂催化合成气直接制乙醇的研究[D].太原: 太原理工大学, 2016.LIU Yong-jun. Study on the ethanol synthesis from syngas over CuZnAl catalysts[D]. Taiyuan: Taiyuan University of Technology, 2016. [13] 董伟兵.热处理条件对CuZnAl催化剂合成低碳醇性能的影响[D].太原: 太原理工大学, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10112-1017832721.htmDONG Wei-bing. Effect of heat treatment conditions on catalytic performance of CuZnAl catalysts for higher alcohol synthesis[D]. Taiyuan: Taiyuan University of Technology, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10112-1017832721.htm [14] SCHULZ H. Short history and present trends of Fischer-Tropsch synthesis[J]. Appl Catal A:Gen, 1999, 186(1/2):3-12. https://www.sciencedirect.com/science/article/abs/pii/S0926860X9900160X [15] 叶同奇, 张朝霞, 徐勇, 颜世志, 朱九方, 刘勇, 李全新. Na促进的CuCoMn催化剂催化生物质合成气合成高醇[J].物理化学学报, 2011, 27(6):1493-1500. doi: 10.3866/PKU.WHXB20110610YE Tong-qi, ZHANG Zhao-xia, XU Yong, YAN Shi-zhi, ZHU Jiu-fang, LIU Yong, LI Quan-xin. Higher alcohol synthesis from bio-syngas over Na-promoted CuCoMn catalyst[J]. Acta Phys-Chim Sin, 2011, 27(6):1493-1500. doi: 10.3866/PKU.WHXB20110610 [16] BOZ I. Higher alcohol synthesis over a K-promoted Co2O3/CuO/ZnO/Al2O3 catalyst[J]. Catal Lett, 2003, 87(3/4):187-194. doi: 10.1023/A:1023499324647 [17] LI Z H, ZUO Z J, HUANG W, XIE K C. Research on Si-Al based catalysts prepared by complete liquid-phase method for DME synthesis in a slurry reactor[J]. Appl Surf Sci, 2011, 257(6):2180-2183. doi: 10.1016/j.apsusc.2010.09.069 [18] SUH Y W, MOON S H, RHEE H K. Active sites in Cu/ZnO/ZrO2 catalysts for methanol synthesis from CO/H2[J]. Catal Today, 2000, 63(2/4):447-452. https://www.sciencedirect.com/science/article/pii/S0920586100004909 [19] GAO W, ZHAO Y F, LIU J M, HUANG Q W, HE S, LI C M, ZHAO J W, WEI M. Catalytic conversion of syngas to mixed alcohols over CuFe-based catalysts derived from layered double hydroxides[J]. Catal Sci Technol, 2013, 3(5):1324-1332. doi: 10.1039/c3cy00025g [20] FIGUEIREDO R T, MARTINEZ-ARIAS A, GRANADOS M L, FIERRO J L G. Spectroscopic evidence of Cu-Al interactions in Cu-Zn-Al mixed oxide catalysts used in CO hydrogenation[J]. J Catal, 1998, 178(1):146-152. https://www.sciencedirect.com/science/article/pii/S0021951798921066 [21] 高志华, 黄伟, 李俊芳, 阴丽华, 谢克昌.以拟薄水铝石为铝源制备浆态床二甲醚合成催化剂[J].高等学校化学学报, 2009, 30(3):534-538. doi: 10.3321/j.issn:0251-0790.2009.03.020GAO Zhi-hua, HUANG Wei, LI Jun-fang, YIN Li-hua, XIE Ke-chang. Liquid-phase preparation of DME slurry catalysts using pseudo-boehmite as aluminum source[J]. Chem J Chin Univ, 2009, 30(3):534-538. doi: 10.3321/j.issn:0251-0790.2009.03.020 [22] 房德仁, 刘中民, 刘德臣, 张慧敏, 孟霜鹤, 王立刚.铝盐加入方式对CuO/ZnO/Al2O3系催化剂性能的影响[J].石油化工, 2004, 33(11):1041-1045. doi: 10.3321/j.issn:1000-8144.2004.11.008FANG De-ren, LIU Zhong-min, LIU De-chen, ZHANG Hui-min, MENG Shuang-he, WANG Li-gang. Influence of Al salt addition methods on performance of CuO/ZnO/Al2O3 catalysts[J]. Petrochem Technol, 2004, 33(11):1041-1045. doi: 10.3321/j.issn:1000-8144.2004.11.008 [23] LIU Y J, ZUO Z J, LIU C B, LI C, DENG X, HUANG W. Higher alcohols synthesis via CO hydrogenation on Cu/Zn/Al/Zr catalysts without alkalis and F-T elements[J]. Fuel Process Technol, 2016, 144:186-190. doi: 10.1016/j.fuproc.2016.01.005 [24] 董伟兵, 郝树宏, 高志华.预热液体石蜡对CuZnAl催化剂合成低碳醇的影响[J].天然气化工, 2017, 42(5):27-33. doi: 10.3969/j.issn.1001-9219.2017.05.006DONG Wei-bing, HAO Shu-hong, GAO Zhi-hua. Effect of preheating liquid paraffin on synthesis of higher alcohols by CuZnAl catalyst[J]. Nat Gas Chem Ind, 2017, 42(5):27-33. doi: 10.3969/j.issn.1001-9219.2017.05.006 [25] 毛东森, 郭强胜, 俞俊, 韩璐蓬, 卢冠忠. Ce添加对Cu-Fe/SiO2催化合成气制低碳醇性能的影响[J].物理化学学报, 2011, 27(11):2639-2645. doi: 10.3866/PKU.WHXB20111125MAO Dong-sen, GUO Qiang-sheng, YU Jun, HAN Lu-peng, LU Guan-zhong. Effect of cerium addition on the catalytic performance of Cu-Fe/SiO2 for the synthesis of lower alcohols from syngas[J]. Acta Phys-Chim Sin, 2011, 27(11):2639-2645. doi: 10.3866/PKU.WHXB20111125 [26] 徐慧远, 储伟, 士丽敏, 张辉, 邓思玉.射频等离子体对合成低碳醇用CuCoAl催化剂的改性作用[J].燃料化学学报, 2009, 37(2):212-216. doi: 10.3969/j.issn.0253-2409.2009.02.016XU Hui-yuan, CHU Wei, SHI Li-min, ZHANG Hui, DENG Si-yu. Effect of glow discharge plasma on copper-cobalt-aluminum catalysts for higher alcohols synthesis[J]. J Fuel Chem Technol, 2009, 37(2):212-216. doi: 10.3969/j.issn.0253-2409.2009.02.016 [27] CHU W, KIEFFER R, KIENNEMANN A, HINDERMANN J P. Conversion of syngas to C1-C6 alcohol mixtures on promoted CuLa2Zr2O7 catalysts[J]. Appl Catal A:Gen, 1995, 27(121):95-111. https://www.researchgate.net/publication/232358075_Conversion_of_syngas_to_C1-C6_alcohol_mixtures_on_promoted_CuLa2Zr2O7_catalysts [28] OKAMOTO Y, FUKINO K, IMANAKA T, TERANISHI S. Surface characterization of copper(Ⅱ) oxide-zinc oxide methanol-synthesis catalysts by x-ray photoelectron spectroscopy. 2. Reduced catalysts[J]. J Phys Chem, 1983, 87(19):3740-3747. doi: 10.1021/j100242a034 [29] 樊金串, 杨瑞卿, 赵杰, 黄伟.液体石蜡体系中含铜物种的化学变化[J].应用化学, 2013, 30(1):67-72. http://d.old.wanfangdata.com.cn/Periodical/yyhx201301012FAN Jin-chuan, YANG Rui-qing, ZHAO Jie, HUANG Wei. Chemical change of copper species in liquid paraffin[J]. Chin J Appl Chem, 2013, 30(1):67-72. http://d.old.wanfangdata.com.cn/Periodical/yyhx201301012 [30] 吕晓东. Cu-Zn-Al催化剂的完全液相制备及催化合成乙醇的研究[D].太原: 太原理工大学, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10112-1015603877.htmLV Xiao-dong. The study of preparation and synthesis of ethanol of Cu-Zn-Al catalyst by complete liquid-phase technology[D]. Taiyuan: Taiyuan University of Technology, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10112-1015603877.htm [31] 孙凯, 张小雨, 张琳, 边仲凯, 黄伟, 赵志换.酸碱性硅溶胶对浆状Cu/Zn/Al催化剂性能的影响[J].燃料化学学报, 2015, 43(10):1221-1229. doi: 10.3969/j.issn.0253-2409.2015.10.010SUN Kai, ZHANG Xiao-yu, ZHANG Lin, BIAN Zhong-kai, HUANG Wei, ZHAO Zhi-huan. Influence of acidic and alkaline silica sols on the performance of Cu/Zn/Al slurry catalysts[J]. J Fuel Chem Technol, 2015, 43(10):1221-1229. doi: 10.3969/j.issn.0253-2409.2015.10.010 [32] 马强, 黄伟, 樊金串, 赵杰, 任杰.完全液相法制备的Cu-Zn-Si-Al浆状催化剂一步法合成二甲醚的失活研究[J].分子催化, 2009, 23(6):499-505. http://d.old.wanfangdata.com.cn/Periodical/fzch200906004MA Qiang, HUANG Wei, FAN Jin-chuan, ZHAO Jie, REN Jie. Study on the deactivation of Cu-Zn-Si-Al slurry catalyst prepared by complete liquid-phase for one-step dimethyl ether synthesis[J]. J Mol Catal(China), 2009, 23(6):499-505. http://d.old.wanfangdata.com.cn/Periodical/fzch200906004 [33] GAO Z H, LIU Y, LI L L, LI S S, HUANG W. CuZnAl catalysts prepared by precipitation-hydrothermal method for higher alcohols synthesis from syngas[J]. Energy Source Part A, 2017, 39(6):1-7. https://www.researchgate.net/publication/319918792_CuZnAl_catalysts_prepared_by_precipitation-hydrothermal_method_for_higher_alcohols_synthesis_from_syngas [34] 刘建国, 定明月, 王铁军, 马隆龙. Cu-Fe基双孔载体催化剂结构和低碳醇合成反应性能[J].物理化学学报, 2012, 28(8):1964-1970. doi: 10.3866/PKU.WHXB201205213LIU Jian-guo, DING Ming-yue, WANG Tie-jun, MA Long-long. Structure and performance of Cu-Fe bimodal support for higher alcohol syntheses[J]. Acta Phys-Chim Sin, 2012, 28(8):1964-1970. doi: 10.3866/PKU.WHXB201205213 -





 下载:
下载: