Study on the oxidative desulfurization performance of SiO2-supported divanadium-substituted phosphotungstate hybrid material
-
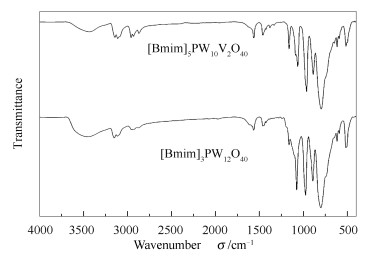
摘要: 利用磷酸氢二钠、偏钒酸纳和钨酸钠为原料,合成了具有Keggin结构的磷钨钒杂多化合物(H5PW10V2O40),并与1-丁基-3-甲基咪唑溴(BmimBr)离子液体反应生成一种杂多酸杂化材料([Bmim]5PW10V2O40)。利用红外光谱(FT-IR)、X射线衍射光谱(XRD)和紫外可见光谱(UV-vis)对所合成的杂多酸杂化材料进行表征。结果表明,[Bmim]5PW10V2O40具有咪唑阳离子基团和Keggin型杂多阴离子基团的结构特征,并且两种基团之间存在相互作用。以SiO2为载体制备负载型的杂多酸杂化材料催化剂[Bmim]5PW10V2O40/SiO2,以H2O2作为氧化剂,考察该催化剂对模拟油中DBT的氧化性能,并优化氧化反应条件,在反应温度40℃,O/S物质的量比为3.0的条件下,反应50 min,模拟油品中的DBT的转化率可以达到100%。催化剂可以通过离心法分离,经过干燥之后,可以循环使用至少七次,而对DBT的氧化活性没有降低。Abstract: The Keggin type H5PW10V2O40 was synthesized by Na2HPO4, NaVO3 and Na2WO4·12H2O. The divanadium-substituted phosphotungstate hybrid material [Bmim]5PW10V2O40 was synthesized by reacting H5PW10V2O40 and 1-butyl-3-methylimidazolium bromide ([Bmim]Br). The FT-IR, XRD and UV-vis characterization results show that the [Bmim]5PW10V2O40 hybrid materials possess Keggin structure and the interactions between the [Bmim]+ and the [PW10V2O40]5-. The SiO2-supported [Bmim]5PW10V2O40/SiO2 was prepared and used for oxidative desulfurization of dibenzothiophene (DBT) with the H2O2 as the oxidant. The experimental results show that the DBT conversion can reach 100% in the [Bmim]5PW10V2O40/SiO2-H2O2 oxidation system under the conditions of 40℃, oxigen/sulfur mol ratio 3.0 and 50 min reaction time. The catalyst was easily separated by centrifugation and could be reused for seven times without decreasing in oxidative desulfurization activity after drying treatment.
-
Key words:
- divanadium-substituted phosphotungstate /
- H2O2 /
- DBT /
- oxidative desulfurization /
- hybrid material
-
表 1 H3PW12O40和H5PW10V2O40的元素分析
Table 1 Elemental analysis of H3PW12O40 and H5PW10V2O40
Sample Actual content of atom w/% Atomic ratio W P V W/P V/P H3PW12O40 76.82 1.09 - 11.97 - H5PW10V2O40 69.85 1.15 3.98 10.28 2.10 表 2 不同催化剂的比表面积及其氧化DBT的活性
Table 2 Specific surface area and catalytic activity in DBT oxidation on different catalysts
Catalyst Specific surface area A/(m2·g-1) DBT conversion x/% SiO2 263.5 0.0 [Bmim]3PW12O40 17.3 13.5 [Bmim]5PW10V2O40 16.8 21.4 10% [Bmim]5PW10V2O40/SiO2 220.1 43.8 20% [Bmim]5PW10V2O40/SiO2 196.4 100.0 30% [Bmim]5PW10V2O40/SiO2 168.7 78.4 40% [Bmim]5PW10V2O40/SiO2 139.2 64.9 20% [Bmim]3PW12O40/SiO2 194.9 62.8 -
[1] SONG C H. An overview of new approaches to deep desulfurization for ultra-clean gasoline, diesel fuel and jet fuel[J]. Catal Today, 2003, 86(1/4): 211-263. https://www.researchgate.net/profile/Chunshan_Song/publication/222545098_An_Overview_of_New_Approaches_to_Deep_Desulfurization_for_Ultra-Clean_Gasoline_Diesel_Fuel_and_Jet_Fuel/links/00b4953458f8ac5beb000000.pdf [2] ETEMADI O, YEN T F. Aspects of selective adsorption among oxidized sulfur compounds in fossil fuels[J]. Energy Fuels, 2007, 21(3): 1622-1627. doi: 10.1021/ef070016b [3] 张存, 王峰, 潘小玉, 刘晓勤.酸性离子液体萃取-氧化模拟油品脱硫研究[J].燃料化学学报, 2011, 3(9): 689-693. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17804.shtmlZHANG Cun, WANG Feng, PAN Xiao-yu, LIU Xiao-qin. Study on extraction-oxidation desulfurization of model oil by acidic ionic liquid[J]. J Fuel Chem Technol, 2011, 3(9): 693-693. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17804.shtml [4] MEILLE V, SHULZ E, VRINAT M. A new route towards deep desulfurization: Selective charge transfer complex formation[J]. Chem Commun, 1998, 29(19): 305-306. https://www.researchgate.net/publication/244537334_A_new_route_towards_deep_desulfurization_Selective_charge_transfer_complex_formation [5] 张健, 白秀梅, 李翔, 王安杰, 马学虎.溶胶-凝胶法制备MoO3-CeO2-SiO2氧化脱硫催化剂[J].催化学报, 2009, 30(10): 1017-1021. doi: 10.1016/S1872-2067(08)60135-7ZHANG Jian, BAI Xiu-mei, LI Xiang, WANG An-jie, MA Xue-hu. Preparation of MoO3-CeO2-SiO2 oxidative desulfurization catalysts by a sol-gel procedure[J]. Chin J Catal, 2009, 30(10): 1017-1021. doi: 10.1016/S1872-2067(08)60135-7 [6] JIANG W, ZHU W S, LI H M, CHAO Y H, XUN S H, CHANG Y H, LIU H, ZHAO Z. Mechanism and optimization for oxidative desulfurization of fuels catalyzed by Fenton-like catalysts in hydrophobic ionic liquid[J]. J Mol Catal A, 2014, 382: 8-14. doi: 10.1016/j.molcata.2013.10.017 [7] 苏建勋, 艾东, 赵荣祥, 李秀萍. CuWO4/C复合物的制备和其在模拟油氧化脱硫中的应用[J].燃料化学学报, 2015, 43(12): 1476-1481. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18747.shtmlSU Jian-xun, AI Dong, ZHAO Rong-xiang, LI Xiu-ping. Study on preparation of CuWO4/C composite and it's application in oxidative desulfurization of model oil[J]. J Fuel Chem Technol, 2015, 43(12): 1476-1481. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18747.shtml [8] ZHANG M, ZHU W S, XUN S H, LI H M, GU Q Q, ZHAO Z, WANG Q. Deep oxidative desulfurization of dibenzothiophene with POM-based hybrid materials in ionic liquids[J]. Chem Eng J, 2013, 220(6): 328-336. https://www.researchgate.net/publication/257566607_Deep_oxidative_desulfurization_of_dibenzothiophene_with_POM-based_hybrid_materials_in_ionic_liquids [9] OTSUKI S, NONAKA T, TAKASHIMA N. Oxidative desulfurization of light gas oil and vacuum gas oil by oxidation and solvent extraction[J]. Energy Fuels, 2000, 14(6): 1232-1239. doi: 10.1021/ef000096i [10] LIANG W D, ZHANG S, LI H F, ZHANG G D. Oxidative desulfurization of simulated gasoline catalyzed by acetic acid-based ionic liquids at room temperature[J]. Fuel Process Technol, 2013, 109(2): 27-31. https://www.researchgate.net/publication/257210210_Oxidative_desulfurization_of_simulated_gasoline_catalyzed_by_acetic_acid-based_ionic_liquids_at_room_temperature [11] SHIRAISHI Y, NAITO T, HIRAI T. Vanadosilicate molecular sieve as a catalyst for oxidative desulfurization of light oil[J]. Ind Eng Chem Res, 2003, 42(24): 6034-6039. doi: 10.1021/ie030328b [12] WANG R, ZHANG G, ZHAO H. Polyoxometalate as effective catalyst for the deep desulfurization of diesel oil[J]. Catal Today, 2010, 149(1): 117-121. https://www.researchgate.net/publication/244322923_Polyoxometalate_as_effective_catalyst_for_the_deep_desulfurization_of_diesel_oil [13] LU H Y, DENG C L, REN W H, YANG X. Oxidative desulfurization of model diesel using[(C4H9)4N]6Mo7O24 as a catalyst in ionic liquids[J]. Fuel Process Technol, 2014, 119(1): 87-91. [14] 张薇, 丁永萍, 宫敬, 宋溪明.羧基功能化离子液体催化二苯并噻吩氧化脱硫[J].燃料化学学报, 2012, 40(5): 626-629. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17954.shtmlZHANG Wei, DING Yong-ping, GONG Jing, SONG Xi-ming. Oxidative desulfurization of dibenzothiophene catalyzed by carboxy-functionalized ionic liquid[cmmim]BF4[J]. J Fuel Chem Technol, 2012, 40(5): 626-629. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17954.shtml [15] 刘丹, 桂建舟, 王利, 张晓彤, 宋丽娟, 孙兆林.功能化离子液体催化柴油氧化脱硫的研究[J].燃料化学学报, 2008, 36(5): 601-605. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18400.shtmlLIU Dan, GUI Jian-zhou, WANG Li, ZHANG Xiao-tong, SONG Li-juan, SUN Zhao-lin. A study of oxidative desulfurzaiton of diesel catalyzed by acidic ionic liquid[J]. J Fuel Chem Technol, 2008, 36(5): 601-605. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18400.shtml [16] HUANG D, ZHAI Z, LU Y. Optimization of composition of a directly combined catalyst in dibenzothiophene oxidation for deep desulfurization[J]. Ind Eng Chem Res, 2007, 46(5): 1447-1451. doi: 10.1021/ie0611857 [17] COLLINS F M, LUCY A R, SHARP C. Oxidative desulphurization of oils via hydrogen peroxide and heteropolyanion catalysis[J]. J Mol Catal A, 1997, 117(1/3): 397-403. http://d.wanfangdata.com.cn/NSTLQK_10.1016-S1381-1169(96)00251-8.aspx [18] XI Z W, ZHOU N, SUN Y, LI K L. Reaction-controlled phase-transfer catalysis for propylene epoxdation to propylene oxide[J]. Science, 2001, 292(11): 1139-1141. https://www.researchgate.net/publication/11988678_Reaction-Controlled_Phase-Transfer_Catalysis_for_Propylene_Epoxidation_to_Propylene_Oxide [19] LU H Y, GAO J B, JIANG Z X, JING F, YANG Y X, WANG G, LI C. Ultra-deep desulfurization of diesel by selevtive oxidation with[C18H37N (CH3)3]4[H2NaPW10O36] catalyst assembled in emulsion droplets[J]. J Catal, 2006, 239(2): 369-375. doi: 10.1016/j.jcat.2006.01.025 [20] LO W H, YANG H, WEI G. One-pot desulfurization of light oils bychemical oxidation and solvent extraction with room temperature ionic liquids[J]. Green Chem, 2003, 5(9): 639-642. https://www.researchgate.net/publication/238132119_One-pot_desulfurization_of_light_oils_by_chemical_oxidation_and_solvent_extraction_with_room_temperature_ionic_liquids [21] LI C, JIANG Z X, GAO J B. Ultra-deep desulfurization of diesel: Oxidation with a recoverable catalyst assembled in emulsion[J]. Chem Eur J, 2004, 10(9): 2277-2280. doi: 10.1002/(ISSN)1521-3765 [22] LI G, SALIM C, HINODE H. Hydrothermal syntheses and crystal structures of two hybrid materials constructed from polyoxometalate clusters and metal-dipyridine complexes[J]. Solid State Sci, 2008, 39(25): 121-128. [23] TAMON H, OKAZAKI M. Influence of acidic surface oxides of activated carbon on gas adsorption characteristics[J]. Carbon, 1996, 34(6): 741-746. doi: 10.1016/0008-6223(96)00029-2 [24] RAO G R, RAJKUMAR T. Interaction of keggin anions of 12-tungstophostporic acid with CexZr1-xO2 solid solutions[J]. J Colloid Interface Sci, 2008, 324(1/2): 134-141. [25] YAMAURA T, KAMATA K, YAMAGUCHI K. Effcient sulfoxidation with hydrogen peroxide catalyzed by a divanadium-substituted phosphotungstate[J]. Catal Today, 2013, 203: 76-81. doi: 10.1016/j.cattod.2012.01.026 -





 下载:
下载: