Upgrading of Zhaotong coal by pyrolysis and its effect on the gasification reactivity
-
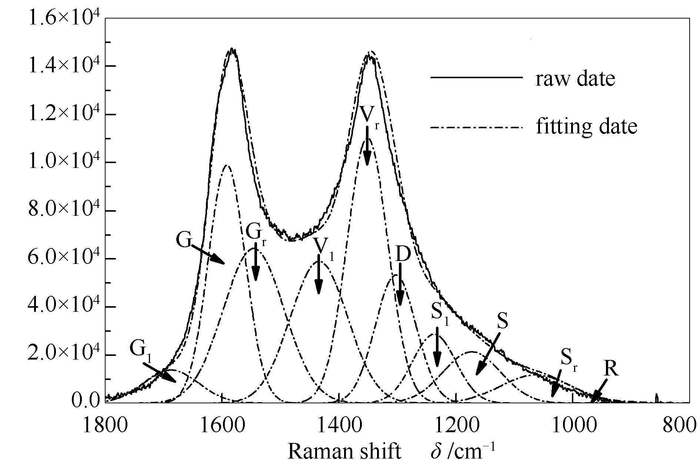
摘要: 为了对储量相对丰富的昭通褐煤进行合理有效的分级转化利用,采用固定床程序升温热解的方法研究了不同温度下煤的热解行为,借助GC-MS和拉曼光谱对所得焦油和煤焦进行了表征分析,并在850℃下对不同热解温度制得的煤焦进行了水蒸气等温气化特性评价。结果表明,热解温度为700℃时,热解气体有效组分(H2、CO、CH4)的累积物质的量占总释放量的70%,此温度下热解气低位热值增长速率最快(以500℃下热解气低位热值为基准计算,其值为90%);酚类化合物在500-700℃大量生成析出,而温度高于700℃时,酚类化合物的分解反应加剧。不同热解温度下所制煤焦的表观气化反应速率随热解温度的升高不断降低,气化产物中CO2与CO的物质的量逐渐升高,700℃热解制得的煤焦在水蒸气气氛下气化所得合成气中有效组分H2与CO的比率最高。Abstract: In order to integratedly utilize the relatively rich coal resource of Zhaotong mine, temperature-programmed pyrolysis experiments of Zhaotong lignite were performed in fixed bed reactor at different temperatures. The resultant coal tar and char were characterized by GC-MS and Raman Spectroscopy, respectively. Char-H2O isothermal gasification characteristics were evaluated in fixed bed reactor at 850℃. The results show that in pyrolysis at 700℃ the cumulative content of H2, CO and CH4 in gases accounts for about 70%, and the growth rate of low calorific value of gas is the fastest, which is 90% based on the value at 500℃. A large number of phenolic compounds are generated at 500-700℃. Above 700℃ the decomposition reactions of the phenolic compounds is intensified. With the increase of pyrolysis temperatures, the apparent reaction rate of char decreases, while the molar ratio of CO2 and CO increases. The molar ratio of H2 and CO in gasification from char pyrolyzed at 700℃ was the highest.
-
Key words:
- pyrolysis upgrading /
- release of gas /
- coal tar /
- gasification reactivity
-
表 1 实验用煤的工业分析和元素分析
Table 1 Proximate and ultimate analyses of sample used in experiment
Proximate analysis w/% Ultimate analysis wdaf /% Mad Ad Vdaf C H N St O* 7.46 15.72 55.04 66.88 5.12 1.57 0.83 25.60 *: by difference 表 2 实验用煤的灰成分分析
Table 2 Analyses of ash composition in coal sample used in experiment
Content w/% SiO2 Al2O3 Fe2O3 CaO MgO TiO2 SO3 K2O Na2O P2O5 38.13 24.84 7.17 12.77 2.06 2.50 10.71 0.85 0.83 0.18 -
[1] OZTAS N A, YURUM Y.Pyrolysis of turkish zonguldak bituminous coal (1):Effect of mineral matter[J].Fuel, 2000, 79(10):1221-1227. doi: 10.1016/S0016-2361(99)00255-0 [2] 王鹏, 文芳, 步学朋, 刘玉华, 边文, 邓一英.煤热解特性研究[J].煤炭转化, 2005, 28(1):8-13.WANG Peng, WEN Fang, BU Xue-peng, LIU Yu-hua, BIAN Wen, DENG Yi-ying.Study on the pyrolysis characteristics of coal[J].Coal Convers, 2005, 28(1):8-13. [3] 石振晶, 夏芝香, 方梦祥, 李超, 王勤辉, 骆仲泱.淮南烟煤热解及焦油析出特性[J].燃烧科学与技术, 2014, 20(1):58-64. http://www.cnki.com.cn/Article/CJFDTOTAL-RSKX201401010.htmSHI Zhen-jing, XIA Zhi-xiang, FANG Meng-xiang, LI Chao, WANG Qin-hui, LUO Zhong-yang.Pyrolysis behavior of Huainan bituminous coal and formation characteristic of tar[J].J Combust Sci Technol, 2014, 20(1):58-64. http://www.cnki.com.cn/Article/CJFDTOTAL-RSKX201401010.htm [4] ALONSO M J G, BORREGO A G, ALVAREZ D, MENENDEZ R.Pyrolysis behaviour of pulverised coals at different temperatures[J].Fuel, 1999, 78:1501-1513. doi: 10.1016/S0016-2361(99)00081-2 [5] NDAJI F E, BUTTERFIELD I M, THOMAS K M.Changes in the macromolecular structure of coals with pyrolysis temperature[J].Fuel, 1997, 76(2):169-177. doi: 10.1016/S0016-2361(96)00175-5 [6] WANG M J, TIAN J L, ROBERTS D G, CHANG L P, XIE K C.Interactions between corncob and lignite during temperature programmed co-pyrolysis[J].Fuel, 2015, 142:102-108. doi: 10.1016/j.fuel.2014.11.003 [7] 钟梅, 马凤云.不同气氛下煤连续热解产物的分配规律及产品品质分析[J].燃料化学学报, 2013, 41(12):1427-1436. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18308.shtmlZHONG Mei, MA Feng-yun.Analysis of product distribution and quality for continuous pyrolysis of coal indifferent atmospheres[J].J Fuel Chem Technol, 2013, 41(12):1427-1436. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18308.shtml [8] LI X H, MA J S, LI L L, LI B F, FENG J, TURMEL W, LI W Y.Semi-coke as solid heat carrier for low-temperature coal tar upgrading[J].Fuel Process Technol, 2016, 143:79-85. doi: 10.1016/j.fuproc.2015.11.013 [9] KRERKKAIWAN S, FUSHIMI C, TSUTSUMI A, KUCHONTHARA P.Synergetic effect during co-pyrolysis/gasification of biomass and sub-bituminous coal[J].Fuel Process Technol, 2013, 115:11-18. doi: 10.1016/j.fuproc.2013.03.044 [10] LI X J, HAYASHI J I, LI C Z.FT-Raman spectroscopic study of the evolution of char structure during the pyrolysis of a Victorian brown coal[J].Fuel, 2006, 85(12/13):1700-1707. [11] 任海君, 张永奇, 房倚天, 王洋.煤焦与生物质焦共气化反应特性研究[J].燃料化学学报, 2012, 40(2):143-148. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17875.shtmlREN Hai-jun, ZHANG Yong-qi, FANG Yi-tian, WANG Yang.Co-gasification properties of coal char and biomass char[J].J Fuel Chem Technol, 2012, 40(2):143-148. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17875.shtml [12] QI X J, GUO X, XUE L C, ZHENG C G.Effect of iron on Shenfu coal char structure and its influence on gasification reactivity[J].J Anal Appl Pyrolysis, 2014, 110:401-407. doi: 10.1016/j.jaap.2014.10.011 [13] ZHAO B F, ZHANG X D, CHEN L, SUN L Z, SI H Y, CHEN G Y.High quality fuel gas from biomass pyrolysis with calcium oxide[J].Bioresour Technol, 2014, 156:78-83. doi: 10.1016/j.biortech.2014.01.031 [14] 杜娟, 王俊宏, 崔银萍, 何秀风, 常丽萍.西部煤热解过程中气相产物的生成与释放规律[J].中国矿业大学学报, 2008, 37(5):694-698. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD200805021.htmDU Juan, WANG Jun-hong, CUI Yin-ping, HE Xiu-feng, CHANG Li-ping.Forming and releasing of gaseous products of coal during pyrolysis in Western China[J].J China Univ Min Technol, 2008, 37(5):694-698. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD200805021.htm [15] 朱学栋, 朱子彬, 韩崇家, 唐黎华.煤的热解研究:煤中官能团与热解生成物[J].华东理工大学学报, 2000, 26(1):14-17. http://www.cnki.com.cn/Article/CJFDTOTAL-HLDX200001003.htmZHU Xue-dong, ZHU Zi-bin, HAN Chong-jia, TANG Li-hua.Fundamental study of coal pyrolysis:Functional group and pyrolysis products[J].J East China Univ Sci Technol, 2000, 26(1):14-17. http://www.cnki.com.cn/Article/CJFDTOTAL-HLDX200001003.htm [16] 闫金定, 崔洪, 杨建丽, 刘振宇.热重质谱联用研究兖州煤的热解行为[J].中国矿业大学学报, 2003, 32(3):311-315. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD200303023.htmYAN Jin-ding, CUI Hong, YANG Jian-li, LIU Zhen-yu.Research on pyrolysis behavior of Yanzhou coal using TG/MS[J].J China Univ Min Technol, 2003, 32(3):311-315. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD200303023.htm [17] STEPHANIE G M, STEPHEN A W.Determination of polycyclic aromatic sulfur heterocycles in fossil fuel-related samples[J].Anal Chem, 1999, 71(1):58-69. doi: 10.1021/ac980664f [18] KONG J, ZHAO R F, BAI Y H, LI G L, ZHANG C, LI F.Study on the formation of phenols during coal flash pyrolysis using pyrolysis-GC/MS[J].Fuel Process Technol, 2014, 127:41-46. doi: 10.1016/j.fuproc.2014.06.004 [19] 谢克昌.煤的结构与反应性[M].北京:科学出版社, 2002.XIE Ke-chang.Coal Structure and its Reactivity[M].Beijing:Science Press, 2002. [20] ELMER B L, NATHAN D M, ALYSSA K S, MARY J W.An experimental study on the thermal decomposition of catechol[J].Proc Combust Inst, 2002, 29:2299-2306. doi: 10.1016/S1540-7489(02)80280-2 [21] SISKIN M, ACZEL T.Pyrolysis studies on the structure of ethers and phenols in coal[J].Fuel, 1983, 62:1321-1326. doi: 10.1016/S0016-2361(83)80017-9 [22] LI C Z.Some recent advances in the understanding of the pyrolysis and gasification behavior of Victorian brown coal[J].Fuel, 2007, 86(12/13):1664-1683. [23] YU J L, TIAN F J, CHOW M C, MCKENZIE L J, LI C Z.Effect of iron on the gasification of Victorian brown coal with steam:enhancement of hydrogen production[J].Fuel, 2006, 85:127-133. doi: 10.1016/j.fuel.2005.05.026 [24] CHAO S, HU S, HE L M, XIANG J, SUN L S, SU S, JIANG L, CHEN Q D, XU C F.The synergistic effect of Ca (OH)2 on the process of lignite steam gasification to produce hydrogen-rich gas[J].Int J Hydrogen Energy, 2014, 39:15506-15516. doi: 10.1016/j.ijhydene.2014.07.111 -





 下载:
下载: