Changes of Zhundong coal properties by hydrothermal upgrading and its impacts on CO2 gasification
-
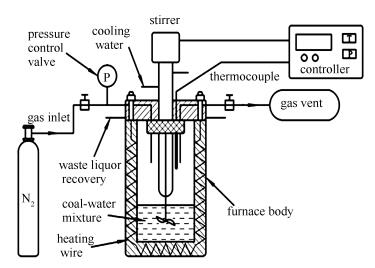
摘要: 选取准东高钠煤为研究对象,在高压反应釜中进行了不同温度(150-350℃)下的水热提质处理实验,依托电感耦合等离子体质谱(ICP-MS)、氮气等温吸附模型(BET)和X射线衍射(XRD)等技术,分析了煤样的煤质特性变化以及对CO2气化的综合影响规律。结果表明,水热提质处理后煤样成熟度增加,煤阶上升,钠的脱除效果显著,在300-350℃时脱除率均大于95%。煤焦化学结构致密、有序、稳定;煤样CO2气化曲线向高温区偏移,气化活性指数呈下降趋势且300-350℃时下降明显。水热提质过程中煤样气化反应活性受到煤阶、碱金属钠含量、物理孔隙结构、化学微晶结构等多个因素的综合影响。Abstract: The Zhundong high sodium coal was hydrothermally upgraded at 150-350℃ in an autoclave. The inductively coupled plasma mass spectrometer (ICP-MS), nitrogen adsorption isotherm (BET) and X-ray diffraction (XRD) were used to investigate the changes of coal properties and the impacts on the CO2 gasification characteristics. The results indicate that the coal quality is increased after the hydrothermal upgrading. The removal effect of sodium is obvious, reaching to more than 95% at 300-350℃. The pore structure of the chars changes significantly, the specific surface area and total pore volume increase initially at 150-300℃ and then decrease at 300-350℃. The crystalline structure tends to be aromatic and graphitized, and the chemical structure becomes dense, orderly and stable. Additionally, the gasification reactivity exhibits a decreasing trend, especially for the chars treated at 300-350℃. The CO2 gasification reactivity during the process of hydrothermal upgrading is comprehensively influenced by different factors containing coal rank, sodium content, physical pore structure and chemical microcrystalline structure.
-
Key words:
- Zhundong coal /
- hydrothermal upgrading /
- coal properties /
- sodium removal /
- CO2 gasification
-
表 1 煤样的工业分析、元素分析
Table 1 Proximate and ultimate analysis of samples
Sample Proximate analysis w/% Ultimate analysis wdaf/% Qnet, d
/ (MJ·kg-1)Atomic ratio (daf) Mad Ad Vd FCd C H O* N S H/C O/C Raw 10.95 5.61 31.90 62.49 73.95 3.97 20.90 0.54 0.64 25.52 0.644 0.212 H150 9.02 5.15 31.86 62.99 74.42 3.92 20.77 0.56 0.40 25.59 0.632 0.209 H200 5.56 5.43 31.15 63.42 74.52 3.90 20.62 0.58 0.38 25.60 0.628 0.208 H250 4.82 5.03 29.86 65.11 74.92 3.80 20.38 0.58 0.32 25.76 0.608 0.204 H300 4.24 5.22 28.48 66.31 76.31 3.79 19.02 0.60 0.29 26.28 0.595 0.187 H350 2.41 5.38 25.36 69.26 78.47 3.73 16.98 0.59 0.23 27.08 0.570 0.162 *:by difference 表 2 焦样的XRD参数
Table 2 XRD parameters of coal chars
Sample 2 θ002/(°) 2 θ100/(°) d002/nm Lc/nm La/nm Raw 24.93 43.89 0.357 1.556 1.897 H150 25.04 43.79 0.355 1.557 2.299 H200 25.17 43.95 0.354 1.705 2.376 H250 25.26 43.84 0.352 1.850 2.382 H300 25.34 44.06 0.351 1.905 2.389 H350 25.58 43.44 0.348 2.243 2.411 -
[1] 严陆光, 夏训诚, 吕绍勤, 吴甲春, 林闽, 黄常纲.大力推进新疆大规模综合能源基地的发展[J].电工电能新技术, 2011, 30(1):1-7. http://www.cnki.com.cn/Article/CJFDTOTAL-DGDN201101001.htmYAN Lu-guang, XIA Xun-cheng, LÜ Shao-qin, WU Jia-chun, LIN Min, HUANG Chang-gang. Great promotion of development of large scale integrative energy base in Xinjiang[J]. Adv Technol Electri Eng Energy, 2011, 30(1):1-7. http://www.cnki.com.cn/Article/CJFDTOTAL-DGDN201101001.htm [2] 王忠萍.新疆洁净煤技术现状和前景[J].洁净煤技术, 2009, 15(4):9-13. http://www.cnki.com.cn/Article/CJFDTOTAL-JJMS200904003.htmWANG Zhong-ping. The current situation and prospects of Xinjiang clean coal technology[J]. Clean Coal Technol, 2009, 15(4):9-13. http://www.cnki.com.cn/Article/CJFDTOTAL-JJMS200904003.htm [3] 陈川, 张守玉, 刘大海, 郭熙, 董爱霞, 熊绍武, 施大钟, 吕俊复.新疆高钠煤中钠的赋存形态及其对燃烧过程的影响[J].燃料化学学报, 2013, 41(7):832-838. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18218.shtmlCHEN Chuan, ZHANG Shou-yu, LIU Da-hai, GUO Xi, DONG Ai-xia, XIONG Shao-wu, SHI Da-zhong, LÜ Jun-fu. Existence form of sodium in high sodium coals from Xinjiang and its effect on combustion process[J]. J Fuel Chem Technol, 2013, 41(7):832-838. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18218.shtml [4] 陶玉洁, 张彦威, 周俊虎, 景雪晖, 李涛, 刘建忠, 岑可法.准东煤在燃烧过程中的矿物演变过程及Na、Ca释放规律[J].中国电机工程学报, 2015, 35(5):1169-1175. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201505018.htmTAO Yu-jie, ZHANG Yan-wei, ZHOU Jun-hu, JING Xue-hui, LI Tao, LIU Jian-zhong, CEN Ke-fa. Mineral conversion regularity and release behavior of Na, Ca during Zhundong coal's combustion[J]. Proc CSEE, 2015, 35(5):1169-1175. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201505018.htm [5] 刘敬, 王智化, 项飞鹏, 黄镇宇, 刘建忠, 周俊虎, 岑可法.准东煤中碱金属的赋存形式及其在燃烧过程中的迁移规律实验研究[J].燃料化学学报, 2014, 42(3):316-322. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18373.shtmlLIU Jing, WANG Zhi-hua, XIANG Fei-peng, HUANG Zhen-yu, LIU Jian-zhong, ZHOU Jun-hu, CEN Ke-fa. Modes of occurrence and transformation of alkali metals in Zhundong coal during combustion[J]. J Fuel Chem Technol, 2014, 42(3):316-322. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18373.shtml [6] Favas G, Jackson W R. Hydrothermal dewatering of lower rank coals. 1. Effects of process conditions on the properties of dried product[J]. Fuel, 2003, 82(1):53-57. doi: 10.1016/S0016-2361(02)00192-8 [7] Timpe R C, Mann M D, Pavlish J H, Louie P K K. Organic sulfur and hap removal from coal using hydrothermal treatment[J]. Fuel Process Technol, 2001, 73(2):127-141. doi: 10.1016/S0378-3820(01)00201-6 [8] 冯小飞, 张成, 张小培, 黎盛鸣, 葛江, 陈刚.水热提质对褐煤理化结构及复吸水特性的影响[J].燃料化学学报, 2016, 44(1):23-29. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18757.shtmlFENG Xiao-fei, ZHANG Cheng, ZHANG Xiao-pei, LI Sheng-ming, GE Jiang, CHEN Gang. Influence of hydrothermal upgrading on physical and chemical structures and moisture readsorption characteristics of a lignite[J]. J Fuel Chem Technol, 2016, 44(1):23-29. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18757.shtml [9] 张书, 陈艳巨, 刘丹, 王雷, 米建新, 王永刚.水热处理对胜利褐煤孔隙结构和表面性质的影响[J].煤炭科学技术, 2013, 41(6):117-121. http://www.cnki.com.cn/Article/CJFDTOTAL-MTKJ201306030.htmZHANG Shu, CHEN Yan-ju, LIU Dan, WANG Lei, MI Jian-xin, WANG Yong-gang. Hydrothermal treatment affected to pore structure and surface property of shengli lignite[J]. Coal Sci Technol, 2013, 41(6):117-121. http://www.cnki.com.cn/Article/CJFDTOTAL-MTKJ201306030.htm [10] Molina A, Mondragon F. Reactivity of coal gasification with steam and CO2[J]. Fuel, 1998, 77(15):1831-1839. doi: 10.1016/S0016-2361(98)00123-9 [11] Krevelen D W. Coal-typology, Chemistry, Physics, Constitution[M]. Elsevier Science & Technology, 1961. [12] 刘扬, 张成, 王澜, 张小培, 冯小飞, 陈刚.高碱煤中钠基化合物对灰熔融特性的影响[J].燃烧科学与技术, 2017, 03:268-273. http://www.cnki.com.cn/Article/CJFDTOTAL-RSKX201703012.htmLIU Yang, ZHANG Cheng, WANG Lan, ZHANG Xiao-pei, FENG Xiao-fei, CHEN Gang. Influence of Sodium-Based Compound on Ash Melting Characteristics of High Alkali Coal[J]. J Combust Sci Technol, 2017, 03:268-273. http://www.cnki.com.cn/Article/CJFDTOTAL-RSKX201703012.htm [13] Li G, Li S, Huang Q, Yao Q. Fine particulate formation and ash deposition during pulverized coal combustion of high-sodium lignite in a down-fired furnace[J]. Fuel, 2015, 143:430-437. doi: 10.1016/j.fuel.2014.11.067 [14] 葛立超, 张彦威, 应芝, 王智化, 周俊虎, 岑可法.水热处理对我国典型褐煤气化特性的影响[J].中国电机工程学报, 2013, 33(32):14-20. http://www.cnki.com.cn/Article/CJFDTOTAL-KEJI201406199.htmGE Li-chao, ZHANG Yan-wei, YING Zhi, WANG Zhi-hua, ZHOU Jun-hu, CEN Ke-fa. Influence of the hydrothermal dewatering on the gasification characteristics of typical chinese lignite[J]. Proc CSEE, 2013, 33(32):14-20. http://www.cnki.com.cn/Article/CJFDTOTAL-KEJI201406199.htm [15] 刘鹏, 周扬, 鲁锡兰, 王岚岚, 潘铁英, 张德祥.先锋褐煤在水热处理过程中的结构演绎[J].燃料化学学报, 2016, 44(2):129-137. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18771.shtmlLIU Peng, ZHOU Yang, LU Xi-lan, WANG Lan-lan, PAN Tie-ying, ZHANG De-xiang. Structural evolution of Xianfeng lignite during hydrothermal treatment[J]. J Fuel Chem Technol, 2016, 44(2):129-137. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18771.shtml [16] Feng B, Bhatia S K. Variation of the pore structure of coal chars during gasification[J]. Carbon, 2003, 41(3):507-523. doi: 10.1016/S0008-6223(02)00357-3 [17] Ogunsola O I. Thermal upgrading effect on oxygen distribution in lignite[J]. Fuel Process Technol, 1993, 34(1):73-81. doi: 10.1016/0378-3820(93)90062-9 [18] 赵卫东. 低阶煤水热改性制浆的微观机理及燃烧特性研究[D]. 杭州: 浙江大学, 2009.ZHAO Wei-dong. Micromechanism and combustion characteristics of low-rank coal water slurry upgraded by hot water treatments[D]. Hangzhou:Zhejiang University, 2009. [19] Lu L, Sahajwalla V, Kong C, Harris D. Quantitative X-ray diffraction analysis and its application to various coals[J]. Carbon, 2001, 39(12):1821-1833. doi: 10.1016/S0008-6223(00)00318-3 [20] Sonibare O O, Haeger T, Foley S F. Structural characterization of Nigerian coals by X-ray diffraction, Raman and FTIR spectroscopy[J]. Energy, 2010, 35(12):5347-5353. doi: 10.1016/j.energy.2010.07.025 [21] 樊文克, 崔童敏, 李宏俊, 常清华, 郭庆华, 于广锁, 王辅臣.碱金属碱土金属对神府煤焦气化活性的影响[J].燃料化学学报, 2016, 44(8):897-903. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18873.shtmlFAN Wen-ke, CUI Tong-min, LI Hong-jun, CHANG Qing-hua, GUO Qing-hua, YU Guang-suo, WANG Fu-chen. Effect of AAEM on gasification reactivity of Shenfu char[J]. J Fuel Chem Technol, 2016, 44(8):897-903. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18873.shtml [22] Yen T F, Erdman J G, Pollack S S. Investigation of the structure of petroleum asphaltenes by X-ray diffraction[J]. Anal Chem, 1961, 33(11):1587-1594. doi: 10.1021/ac60179a039 [23] Takarada T, Tamai Y, Tomita A. Reactivities of 34 coals under steam gasification[J]. Fuel, 1985, 64(10):1438-1442. doi: 10.1016/0016-2361(85)90347-3 [24] 谢克昌.煤的结构与反应性[M].北京, 科学出版社, 2002.XIE Ke-chang. Coal structure and its reactivity[M]. Beijing:Science Press, 2002. -





 下载:
下载: