Synthesis of tetragonal sulfated zirconia via a novel route for biodiesel production
-
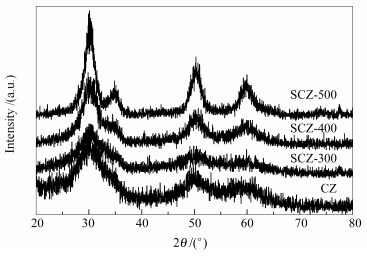
摘要: 将过硫酸铵浸渍于直接合成的纳米氧化锆晶体表面, 经300-500℃高温焙烧处理获得硫化氧化锆催化剂.采用X射线衍射 (XRD)、氮气吸附-脱附、氨气程序升温脱附 (NH3-TPD)、傅里叶红外光谱 (FT-IR)、扫描电镜 (SEM) 和能谱仪 (EDS) 等分析手段对催化剂的结构性能进行了表征.结果表明, 所有的催化剂均展现出纯四方相结构和高的结晶度.其中, 经500℃热处理获得的催化剂拥有最高的硫含量和酸性位, 将其应用于大豆油与甲醇酯交换反应合成生物柴油, 获得了脂肪酸甲酯收率高达84.6%的催化效果, 进一步表明该催化剂表面存在优越的超强酸位.Abstract: The sulfated zirconia was prepared by directly impregnating ammonium persulphate on the crystalline zirconia and followed by the calcination temperatures of 300-500℃. The structural properties of the catalysts were characterized by X-ray diffraction (XRD), N2 adsorption-desorption, ammonia temperature programmed desorption (NH3-TPD), Fourier transform infrared spectroscopy (FT-IR) and a scanning electron microscope (SEM) equipped with an energy dispersive spectroscope (EDS). The experiment results demonstrated that the catalysts presented the tetragonal structure of zirconia and high crystallinity. The catalyst calcined at 500℃ possessed the highest sulfur content and acidic sites in the catalysts. The catalyst exhibited high catalytic activity in transesterification of soybean oil with methanol. The yield of biodiesel achieved 84.6% because of the preferable super-acid sites on the surface of the catalyst.
-
Key words:
- tetragonal sulfated zirconia /
- impregnating /
- crystalline zirconia /
- transesterification /
- biodiesel
-
Table 1 Textrual properties of the CZ and SCZ samples
Sample Calcination temperature t/℃ ABET /(m2·g-1) CZ - 450 SCZ-300 300 141 SCZ-400 400 156 SCZ-500 500 66 Table 2 Acid properties of the different SCZ samples
Sample Peak temperature t/℃ Area under NH3-TPD peaks (a.u./mg) Total NH3-TPD peak area (a.u./mg) SCZ-300 234 553 0.08 0.09 0.17 SCZ-400 309 601 0.05 0.29 0.34 SCZ-500 309 602 0.62 0.53 1.15 Table 3 Catalytic results obtained over the SCZ catalysts
Sample Reaction temperature t/℃ Reaction time t/h FAME yield w/% SCZ-300 150 6 1.8 SCZ-400 150 6 7.1 SCZ-500 150 6 84.6 -
[1] ZALI A, SHOKROLAHI A. Nano-sulfated zircionia as an efficient, recyclable and environmentally benign catalyst for one-pot three component synthesis of amidoalkyl naphthols[J]. Chin Chem Lett, 2012, 23(3):269-272. doi: 10.1016/j.cclet.2011.12.002 [2] GARG S, SONI K, KUMARAN G M, BAL R, GORA-MARED K, GUPTA J K, SHARMA L D, DHAR G M. Acidity and catalytic activities of sulfated zirconia inside SBA-15[J]. Catal Today, 2009, 141(1/2):125-129. https://www.researchgate.net/publication/223352370_Acidity_and_catalytic_activities_of_sulfated_zirconia_inside_SBA-15 [3] ZHAO J, YUE Y H, HUA W M, HE H Y, GAO Z. Catalytic activities and properties of sulfated zirconia supported on mesostructured γ-Al2O3[J]. Appl Catal A:Gen, 2008, 336(1/2):133-139. doi: 10.1016/j.apcata.2007.08.014 [4] ESSAYEM N, MARTIN V, RIONDEL A, VEDRINE J C. Esterification of acrylic acid with but-1-ene over sulfated Fe-and Mn- promoted zirconia[J]. Appl Catal A:Gen, 2007, 326(1):74-81. doi: 10.1016/j.apcata.2007.03.038 [5] HESHMATPOUR F, AGHAKHANPOUR R B. Synthesis and characterization of superfine pure tetragonal nanocrystalline sulfated zirconia powder by a non-alkoxide sol-gel route[J]. Adv Powder Technol, 2012, 23(1):80-87. doi: 10.1016/j.apt.2010.12.012 [6] MORENO J A, PONCELET G. Isomerization of n-Butane over sulfated Al-and Ga-promoted zirconium oxide catalysts. influence of promoter and preparation method[J]. J Catal, 2001, 203(2):453-465. doi: 10.1006/jcat.2001.3324 [7] BAUTISTA P, FARALDOS M, YATES M, BAHAMONDE A. Influence of sulphate doping on Pd/zirconia based catalysts for the selective catalytic reduction of nitrogen oxides with methane[J]. Appl Catal B:Environ, 2007, 71(3/4):254-261. doi: 10.1016/j.apcatb.2006.08.020 [8] SAITO M, ITOH M, IWAMOTO J. Low-temperature hydrogen-selective catalytic reduction of NOx on Pt/sulfated-ZrO2 catalysts under excess oxygen conditions[J]. Chem Lett, 2008, 37(12):1210-1211. doi: 10.1246/cl.2008.1210 [9] REDDY B M, REDDY G K, RAO K N. Influence of alumina and titania on the structure and catalytic properties of sulfated zirconia:Beckmann rearrangement[J]. J Mol Catal A:Chem, 2009, 306(1/2):62-68. https://www.researchgate.net/publication/244279092_Influence_of_alumina_and_titania_on_the_structure_and_catalytic_properties_of_sulfated_zirconia_Beckmann_rearrangement [10] PIETROGIACOMI D, MAGLIANO A, SANNINO D, CAMPA M C, CIAMBELLI P, INDOVINA V. In situ sulphated CuOx/ZrO2 and CuOx/sulphated-ZrO2 as catalysts for the reduction of NOx with NH3 in the presence of excess O2[J]. Appl Catal B:Environ, 2005, 60(1/2):83-92. doi: 10.1016/j.apcatb.2005.02.025 [11] SIGNORETTO M, MELADA S, PINNA F, POLIZZI S, CERRAT O C, MORTERRA C. Ga2O3-promoted sulfated zirconia systems:Morphological, structural and redox properties[J]. Microporous Mesoporous Mater, 2005, 81(1/3):19-29. doi: 10.1016/j.micromeso.2005.01.009 [12] SONG Y Q, TIAN J, YE Y R, JIN Y Q, ZHOU X L, WANG J A, XU L Y. Effects of calcinations temperature and water-washing treatment on n-hexane hydroisomerization behavior of Pt-promoted sulfated zirconia based catalysts[J]. Catal Today, 2013, 212(8):108-114. doi: 10.1016/j.cattod.2012.07.024 [13] GAO S, CHEN X B, WANG H Q, MO J S, WU Z B, LIU Y, WENG X L. Ceria supported on sulfated zirconia as a superacid catalyst for selective catalytic reduction of NO with NH3[J]. J Colloid Interface Sci, 2013, 394(1):515-521. https://www.researchgate.net/publication/235396064_Ceria_supported_on_sulfated_zirconia_as_a_superacid_catalyst_for_selective_catalytic_reduction_of_NO_with_NH3 [14] LI X B, NAGAOKA K, OLINDO R, LERCHER J A. Synthesis of highly active sulfated zirconia by sulfation with SO3[J]. J Catal, 2006, 238(1):39-45. doi: 10.1016/j.jcat.2005.11.039 [15] SHI G L, YU F, WANG Y, LI R F. Synthesis of growth-controlled ZrO2 nanocrystals via vapor phase hydrolysis[J]. Ceram Int, 2014, 40(8):13083-13088. doi: 10.1016/j.ceramint.2014.05.006 [16] YADAV G D, AJGAONKAR N P, VARMA A. Preparation of highly superacidic sulfated zirconia via combustion synthesis and its application in pechmann condensation of resorcinol with ethyl acetoacetate[J]. J Catal, 2012, 292(4):99-110. https://www.researchgate.net/publication/256737766_Preparation_of_highly_superacidic_sulfated_zirconia_via_combustion_synthesis_and_its_application_in_Pechmann_condensation_of_resorcinol_with_ethyl_acetoacetate [17] VLASOV E A, MYAKIN S V, SYCHOV M M, AHO A, POSTNOV A Y, MALTSEVA N V, DOLGASHEV A O, OMAROV S O, MURZIN D Y. On synthesis and characterization of sulfated alumina-zirconia catalysts for isobutene alkylation[J]. Catal Lett, 2015, 145(9):1651-1659. doi: 10.1007/s10562-015-1575-7 [18] BERNAL H G, CAERO L C, FINOCCHIO E, BUSCA G. An FT-IR study of the adsorption and reactivity of tert-butyl hydroperoxide over oxide catalysts[J]. Appl Catal A:Gen, 2009, 369(1):27-35. doi: 10.1016/j.apcata.2009.08.026 [19] CORMA A, FORNES V, JUAN R M I, BUSCA G. Influence of preparation conditions on the structure and catalytic properties of SO42-/ZrO2 superacid catalysts[J]. Appl Catal A:Gen, 1994, 116(1/2):151-163. doi: 10.1002/chin.199451023 [20] YADAV G D, MURKUTE A D. Preparation of a novel catalyst UDCaT-5:enhancement in activity of acid-treated zirconia effect of treatment with chlorosulfonic acid vis-à-vis sulfuric acid[J]. J Catal, 2004, 224(1):218-223. doi: 10.1016/j.jcat.2004.02.021 [21] PETCHMALA A, LAOSIRIPOJANA N, JONGSOMJIT B, GOTO M, PANPRANOT J, MEKASUWAN-DUMRONG O. Transesterification of palm oil and esterification of palm fatty acid in near-and super-critical methanol with SO4-ZrO2 catalysts[J]. Fuel, 2010, 89(9):2387-2392. doi: 10.1016/j.fuel.2010.04.010 [22] SHI G L, YU F, WANG Y, PAN D H, WANG H G, LI R F. A novel one-pot synthesis of sulfated zirconia catalyst with high activity for biodiesel production from the transesterification of soybean oil[J]. Renew Energy, 2016, 92(A1/A6):22-29. -





 下载:
下载: