Catalytic performance of surface silicon-rich ZSM-5 zeolites in the co-production of lower olefins and p-xylene from methanol
-
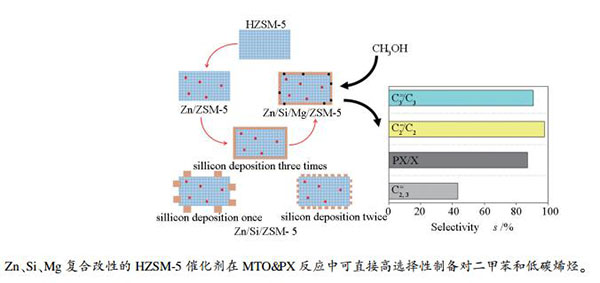
摘要: 通过表面化学改性制备表面富硅型ZSM-5分子筛,采用XRD、N2等温吸附-脱附、TEM、NH3-TPD和Py-FTIR等手段对其孔结构和酸性质进行了表征,考察其甲醇制对二甲苯联产低碳烯烃反应的催化性能。结果表明,引入Zn使催化剂中一部分强酸转变为中强酸,并增加了具有脱氢作用的Zn-L酸中心,提高乙烯和丙烯的选择性;Mg改性不仅可以调节孔道的择形性,而且增加了L酸中心的数量,有利于PX的生成;不同硅源的多次硅沉积使得SiO2可以更均匀地沉积在外表面,调变催化剂的酸性质和孔结构,从而进一步提高对二甲苯及乙烯和丙烯的选择性。最终,二甲苯中对位选择性可达到87.1%,二甲苯及乙烯和丙烯的选择性达到61%,乙烯在C2烃类的选择性高达97.8%,丙烯在C3烃类中选择性高达90.6%。Abstract: Surface silicon-rich ZSM-5 zeolites were prepared by surface chemical modification; their pore structure and acid properties were characterized by XRD, nitrogen sorption, TEM, NH3-TPD and Py-FTIR spectroscopy. The catalytic performance of modified ZSM-5 zeolites in the conversion of methanol to p-xylene and lower olefins was investigated. The results show that the introduction of Zn in ZSM-5 can change part of the strong acid sites into the medium ones and increase the Zn-Lewis acid sites with dehydrogenation capacity, which can enhance the selectivity to ethene and propene. The modification with Mg can not only adjust the pore shape selectivity, but also increase the amount of Lewis acid sites, which is beneficial to the formation of p-xylene. Through multiple silicon depositions from different silicon sources, SiO2 is uniformly deposited on the outer surface of modified ZSM-5 catalysts, which can modulate the acid properties and pore structure and thereby further improve the selectivity to p-xylene and ethene and propene. By using these modification approaches, the selectivity to p-xylene and ethene and propene reaches 61%, with 87.1% of p-xylene in the xylenes product, 97.8% of ethene in C2 hydrocarbons, and 90.6% of propene in C3 hydrocarbons.
-
Key words:
- p-xylene /
- shape-selective catalysis /
- silicon deposition /
- light olefins /
- ZSM-5
-
表 1 HZSM-5改性前后的比表面积和孔结构
Table 1 Surface area and pore structure of the original and modified HZSM-5
Catalyst Surface area A/(m2·g-1) Pore volume v/(mL·g-1) total micropore external total micropore H-ZSM-5 454 447 7 0.204 0.183 Zn/ZSM-5 390 371 19 0.192 0.157 Zn/Si/ZSM-5 303 289 14 0.183 0.152 Zn/Si/Mg/ZSM-5 282 258 24 0.173 0.151 表 2 HZSM-5改性前后的NH3-TPD表征
Table 2 NH3-TPD results of the original and modified HZSM-5
Catalyst Acidity /(mmol·g-1) Reduced amount of acid sites by modification /% total weak acid medium acid strong acid HZSM-5 1.08 0.60 0.13 0.36 Zn/ZSM-5 0.96 0.47 0.40 0.09 11.11 Zn/Si/ZSM-5 0.80 0.45 0.27 0.08 16.67 Zn/Si/Mg/ZSM-5 0.79 0.46 0.26 0.07 1.25 表 3 Zn/ZSM-5改性前后的B酸和L酸酸量
Table 3 Amounts of B and L acid sites of the modified and unmodified Zn/ZSM-5
Catalyst Acidity /(mmol·g-1) Ratio B/L B acid L acid Zn/ZSM-5 0.39 0.30 1.30 Zn/Si/ZSM-5 0.18 0.36 0.50 Zn/Si/Mg/ZSM-5 0.16 0.37 0.43 *: the concentrations of B and L acid sites were calculated from the B/L ratio and the total acid quantity measured by NH3-TPD, where the B/L ratio was obtained from the Py-FTIR spectra 表 4 HZSM-5改性前后的MTO & PX反应性能
Table 4 Catalytic performance of the original and modified HZSM-5 zeolites in the reactions of methanol to olefins (MTO) and para-xylene (PX)
Catalyst HZSM-5 Zn/ZSM-5 Zn/Si/ZSM-5 Zn/Si/Mg/ZSM-5 Methanol conversion x/% >99 >99 >99 >99 Aromatics selectivity s/% 39.65 51.29 29.90 33.42 PX/xylene /% 22.92 44.26 82.83 87.10 C2, 3= selectivity s/% 7.39 30.81 45.04 43.44 BTX selectivity s/% 36.5 41.55 22.27 25.37 Selectivity s/% CH4 4.05 3.43 7.64 7.16 C2H4 4.16 20.07 31.89 29.31 C2H6 2.45 0.89 0.97 0.67 C3H6 3.23 10.74 13.15 14.13 C3H8 32.03 1.63 1.97 1.47 C4H10 3.53 1.31 2.28 2.29 C4H8 8.52 2.11 1.77 2.49 C5H12 1.13 0.35 0.33 0.17 C5H10 0.85 5.83 7.45 4.07 C6+ hydrocarbon 0.40 2.37 2.65 4.83 Benzene 5.80 1.50 0.30 0.40 Toluene 16.10 9.60 3.70 4.60 Ethylbenzene 1.60 0.20 0.10 0.20 p-xylene 2.98 13.39 15.05 17.57 m-xylene 7.05 12.59 2.49 2.14 o-xylene 2.97 4.27 0.63 0.46 C9+ aromatics 3.15 9.74 7.63 8.04 Total 100.00 100.00 100.00 100.00 -
[1] 伍川, 王净依, 陆俊跃, 王晓东, 刘宝河, 黄培, 时钧.对二甲苯分离提纯进展[J].南京工业大学学报, 2002, 24(3):106-110. http://d.old.wanfangdata.com.cn/Periodical/njhgdxxb200203025WU Chuan, WANG Jing-yi, LU Jun-yue, WANG Xiao-dong, LIU Bao-he, HUANG Pei, SHI Jun. Research progress on separation for para-xylene[J]. J Nanjing Univ Technol, 2002, 24(3):106-110. http://d.old.wanfangdata.com.cn/Periodical/njhgdxxb200203025 [2] 冯志强. PX结晶分离技术与芳烃装置扩产改造[J].现代化工, 2015, 35(4):142-145. http://d.old.wanfangdata.com.cn/Periodical/xdhg201504037FENG Zhi-qiang. PX crystallization separation technology and reconstruction of aromatic plant[J]. Mod Chem Ind, 2015, 35(4):142-145. http://d.old.wanfangdata.com.cn/Periodical/xdhg201504037 [3] 郝西维, 刘秋芳, 刘弓, 张亚秦, 张世刚.对二甲苯生产技术开发进展及展望[J].洁净煤技术, 2016, 22(5):25-30. http://d.old.wanfangdata.com.cn/Periodical/jjmjs201605005HAO Xi-wei, LIU Qiu-fang, LIU Gong, ZHANG Ya-qin, ZHANG Shi-gang. Progress and prospect of p-xylene production technologies[J]. Clean Coal Technol, 2016, 22(5):25-30. http://d.old.wanfangdata.com.cn/Periodical/jjmjs201605005 [4] 吴巍.芳烃联合装置生产技术进展及成套技术开发[J].石油学报(石油加工), 2013, 31(2):275-281. http://d.old.wanfangdata.com.cn/Periodical/syxb-syjg201502010WU Wei. Advances and development of aromatics production technologies for an aromatics complex[J]. Acta Pet Sin(Pet Process Sect), 2013, 31(2):275-281. http://d.old.wanfangdata.com.cn/Periodical/syxb-syjg201502010 [5] 孔德金, 杨为民.芳烃生产技术进展[J].化工进展, 2011, 30(1):16-25. http://d.old.wanfangdata.com.cn/Periodical/hgjz201101002KONG De-jin, YANG Wei-min. Advance in technology for production of aromatic hydrocarbons[J]. Chem Ind Eng Prog, 2011, 30(1):16-25. http://d.old.wanfangdata.com.cn/Periodical/hgjz201101002 [6] 陈亮, 肖剑, 谢在库, 于建国.对二甲苯结晶分离技术进展[J].现代化工, 2009, 29(2):10-14. http://d.old.wanfangdata.com.cn/Periodical/xdhg200902003CHEN Liang, XIAO Jian, XIE Zai-ku, YU Jian-guo. Advances in p-xylene separation by crystalization[J]. Mod Chem Ind, 2009, 29(2):10-14. http://d.old.wanfangdata.com.cn/Periodical/xdhg200902003 [7] 范景新, 于海斌, 臧甲忠, 邢淑建.甲苯甲醇选择性烷基化技术研究进展[J].工业催化, 2013, 21(6):1-4. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gych201306001FAN Jing-xin, YU Hai-bin, ZANG Jia-zhong, XING Shu-jian. Research progress in selective alkylation of toluene and methanol[J]. Ind Catal, 2013, 21(6):1-4. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gych201306001 [8] 李昭清.甲醇甲苯烷基化制对二甲苯联产低碳烯烃催化剂的研究[D].北京: 中国石油大学, 2016. http://cdmd.cnki.com.cn/Article/CDMD-10425-1018814687.htmLI Zhao-qing. Study on catalyst for alkylation of toluene with methanol for preparing p-xylene and low-carbon olefin[D]. Beijing: China University of Petroleum, 2016. http://cdmd.cnki.com.cn/Article/CDMD-10425-1018814687.htm [9] LI J H, TONG K, XI Z W, YUAN Y, HU Z H, ZHU Z R. Highly-efficient conversion of methanol to p-xylene over shape-selective Mg-Zn-Si-HZSM-5 catalyst with fine modification of pore-opening and acidic properties[J]. Catal Sci Technol, 2016, 6:4802-4813. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cc64daacb99ffc401fba5c8a62c0bac5 [10] 关文斌, 辛玉兵, 魏立虎, 张世刚.甲醇芳构化制对二甲苯的研究进展[J].煤化工, 2019, 47(2):11-15. http://d.old.wanfangdata.com.cn/Periodical/hgsjtx201506002GUAN Wen-bin, XIN Yu-bin, WEI Li-hu, ZHANG Shi-gang. Research progress on para-xylene synthesis by aromatization of methanol[J]. Coal Chem Ind, 2019, 47(2):11-15. http://d.old.wanfangdata.com.cn/Periodical/hgsjtx201506002 [11] ZHANG G Q, ZHANG X, BAI T, CHEN T F, FAN W T. Coking kinetics and influence of reaction-regeneration on acidity, activity and deactivation of Zn/HZSM-5 catalyst during methanol aromatization[J]. J Energy Chem, 2015, 24(1):108-118. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=trqhxzz-e201501015 [12] 张志智, 秦张峰, 王国富, 王建国.近临界下HZSM-5催化的甲苯歧化反应[J].燃料化学学报, 2005, 33(1):96-100. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb200501019ZHANG Zhi-zhi, QIN Zhang-feng, WANG Guo-fu, WANG Jian-guo. Toluene disproportion over HZSM-5 under near critical coditions[J]. J Fuel Chem Technol, 2005, 33(1):96-100. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb200501019 [13] ONO Y. Transformation of lower alkanes into aromatic hydrocarbons over ZSM-5 zeolites[J]. Catal Rev:Sci Eng, 1992, 34(3):179-226. doi: 10.1080-01614949208020306/ [14] ZAIDI H A, PANT K K. Catalytic conversion of methanol togasoline range hydrocarbons[J]. Catal Today, 2004, 96(3):155-160. https://www.sciencedirect.com/science/article/pii/S0920586104003530 [15] ZHANG J G, QIAN W Z, KONG C Y, WEI F. Increasing para-xylene selectivity in making aromatics from methanol with a surface-modified Zn/P/ZSM-5 catalyst[J]. ACS Catal, 2015, 5(5):2982-2988. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8db4192569cfca328a8c6e56f8509dc0 [16] NI Y M, SUN A M, Wu X L, HAI G L, HU J L, LI T, LI G X. The preparation of nano-sized H[Zn, Al]ZSM-5 zeolite and its application in the aromatization of methanol[J]. Microporous Mesoporous Mater, 2011, 143(2/3):435-442. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6b7a3c438e41c277e8246552558c45d0 [17] 张志萍, 赵岩, 吴宏宇, 谭伟, 王祥生, 郭新闻.改性纳米HZSM-5催化剂上甲苯与甲醇的烷基化反应[J].催化学报, 2011, 32(7):1280-1286. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201107027ZHANG Zhi-ping, ZHAO Yan, WU Hong-yu, TAN Wei, WANG Xiang-sheng, GUO Xin-wen. Shape-selective alkylation of toluene with methanol over modified nano-scale HZSM-5 zeolite[J]. Chin J Catal, 2011, 32(7):1280-1286. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201107027 [18] HU Q F, HUANG X F, CUI Y, LUO T F, TANG X P, WANG T, QIAN W Z, WEI F. High yield production of C2, 3 olefins and para-xylene from methanol using a SiO2-coated FeOx/ZSM-5 catalyst[J]. RSC Adv, 2017, 7(46):28940-28944. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=ec07a98d958879c30e96d17b9ed9f27e [19] MIYAKE K, HIROTA Y, ONO K, UCHIDA Y, TANAKA S, NISHIYAMA N. Direct and selective conversion of methanol to para-xylene over Zn ion doped ZSM-5/silicalite-1 core-shell zeolite catalyst[J]. J Catal, 2016, 342:63-66. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=9007635c65e80b4343f858bbdaf3df9d [20] PAN D H, SONG X H, YANG X H, GAO L J, WEI R P, ZHANG J, XIAO G M. Efficient and selective conversion of methanol to para-xylene over stable H[Zn, Al]ZSM-5/SiO2 composite catalyst[J]. Appl Catal A:Gen, 2018, 557:15-24. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c6953925a851e66770455c5565652d6d [21] LIANG T Y, CHEN J L, QIN Z F, LI J F, WANG P F, WANG S, WANG G F, DONG M, FAN W B, WANG J G. Conversion of methanol to olefins over HZSM5 zeolite:Reaction pathway is related to the framework aluminum siting[J]. ACS Catal, 2016, 6(11):7311-7325. doi: 10.1021/acscatal.6b01771 [22] 钟炳, 罗庆云, 肖有燮, 张威.甲醇在HZSM-5上转化为烃类的催化反应机理[J].燃料化学学报, 1986, 14(1):9-16.ZHONG Bing, LUO Qing-yun, XIAO You-xie, ZHANG Wei. Reaction mechanism of methanol to hydrocarbons on HZSM-5[J]. J Fuel Chem Technol, 1986, 14(1):9-16. [23] 王锦业, 王定珠, 卢学栋, 窦秀云.阳离子改性HZSM-5沸石上低碳醇转化为芳烃[J].催化学报, 1993, 14(3):324-327. http://www.cqvip.com/QK/93027X/199303/1003389.htmlWANG Jin-ye, WANG Ding-zhu, LU Xue-dong, DOU Xiu-yun. Conversion of lower alcohols into aromatics over cation-modified HZSM-5 zeolites[J]. Chin J Catal, 1993, 14(3):324-327. http://www.cqvip.com/QK/93027X/199303/1003389.html [24] 曹劲松, 张军民, 许磊, 刘中民.甲苯甲醇烷基化制PX技术的开发优势[J].石油化工技术与经济, 2010, 26(1):8-10. http://d.old.wanfangdata.com.cn/Periodical/syhgjsjj201001003CAO Jin-song, ZHANG Jun-min, XU Lei, LIU Zhong-min. Superiorities for developing PX production process through alkylation of toluene alcohol[J]. Technol Econ Petrochem, 2010, 26(1):8-10. http://d.old.wanfangdata.com.cn/Periodical/syhgjsjj201001003 [25] ZHONG J W, HAN J F, WEI Y X, XU S T, SUN T T, GUO X W, SONG C S, LIU Z M. The template-assisted zinc ion incorporation in SAPO-34 and the enhanced ethylene selectivity in MTO reaction[J]. J Energy Chem, 2019, 32:174-181. https://www.sciencedirect.com/science/article/abs/pii/S2095495618305916 [26] ZHONG J W, HAN J F, WEI Y X, XU S T, SUN T T, GUO X W, SONG C S, LIU Z M. Enhancing ethylene selectivity in MTO reaction by incorporating metal species in the cavity of SAPO-34 catalysts[J]. Chin J Catal, 2019, 39(11):1821-1831. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201811012 [27] REN S, LIU G, WU X. Enhanced MTO performance over acid treated hierarchical SAPO-34[J]. Chin J Catal, 2017, 38:123-130. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201701015 [28] LI L B, LIN R B, KRISHNA R, LI H, XIANG S C, WU H, LI J P, ZHOU W, CHEN B L. Ethane/ethylne separation in a metal-organic framework with iron-peroxo sites[J]. Sci, 2018, 362(6413):443-446. https://science.sciencemag.org/content/362/6413/443.full [29] JIA Y M, WANG J W, ZHANG K, WEI F, LIU S B, DING C M, LIU P. Promoted effect of zinc-nickel bimetallic oxides supported on HZSM-5 catalysts in aromatization of methanol[J]. J Energy Chem, 2017, 26(3):540-548. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=trqhxzz-e201703031 [30] 王珏, 赵璧英, 谢有畅. MgO/HZSM-5中MgO分散状态和催化性能的关系[J].物理化学学报, 2001, 17(11):966-971. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=wlhxxb200111002WANG Yu, ZHAO Bi-ying, XIE You-chang. Correlations between the dispersion state of MgO and catalytic behavior of MgO/HZSM-5[J]. Acta Phys-Chim Sin, 2001, 17(11):966-971. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=wlhxxb200111002 [31] 韩丽华, 刘平, 高俊华, 周浩, 孙晓芳, 刘增厚, 张侃. Si、P、Mg改性HZSM-5提高甲苯/甲醇烷基化中对二甲苯的选择性[J].天然气化工(C1化学与化工), 2019, 44(3):1-6. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=trqhg201903001HAN Li-hua, LIU Ping, GAO Jun-hua, ZHOU Hao, SUN Xiao-fang, LIU Zeng-hou, ZHANG Kan. Si, P and Mg-modified HZSM-5 catalyst for enhancing the para-selectivity in toluene/methanol alkylation[J]. Nat Gas Ind, 2019, 44(3):1-6. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=trqhg201903001 [32] JANG H G, MIN H K, HONG S B, SEO J. Tetramethylbenzenium radical cations as major active intermediates of methanol-to-olefin conversions over phosphorous-modified HZSM-5 zeolites[J]. J Catal, 2013, 299:240-248. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=65ed9aea12e373aa8c53fc9b71e3e8a2 [33] 王森, 陈艳艳, 卫智虹, 秦张峰, 李俊汾, 董梅, 樊卫斌, 王建国.分子筛骨架结构和酸性对其甲醇制烯烃(MTO)催化性能影响研究进展[J].燃料化学学报, 2015, 43(10):1201-1214. http://www.ccspublishing.org.cn/article/id/100033419WANG Sen, CHEN Yan-yan, WEI Zhi-hong, QIN Zhang-feng, LI Jun-fen, DONG Mei, FAN Wei-bin, WANG Jian-guo. Recent research progresses in the effect of framework structure and acidity of zeolites on their catalytic performance in methanol to olefins (MTO)[J]. J Fuel Chem Technol, 2015, 43(10):1201-1214. http://www.ccspublishing.org.cn/article/id/100033419 [34] BARTHOS R, LONYI F, ONYESTYAK G, VALYON J. An IR, FR, and TPD Study on the acidity of H-ZSM-5, sulfated zirconia, and sulfated zirconia-titania using ammonia as the probe molecule[J]. J Phys Chem B, 2000, 104(31):7311-7319. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=62fd1a4ed03526f709c739556e916d12 [35] KATADA N, IGI H, KIM J H, NIWA M. Determination of the acidic properties of zeolite by theoretical analysis of temperature-programmed desorption of ammonia based on adsorption equilibrium[J]. J Phys Chem B, 1997, 101(31):5969-5977. doi: 10.1021-jp9639152/ [36] TAN W, LIU M, ZHAO Y, HOU K K, WU H Y, ZHANG A F, LIU H O, WANG Y R, SONG C S, GUO X W. Para-selective methylation of toluene with methanol over nano-sized ZSM-5 catalysts:Synergistic effects of surface modifications with SiO2, P2O5 and MgO[J]. Microporous Mesoporous Mater, 2014, 196(15):18-30. [37] WOOLERY G L, KUEHL G H, TIMKEN H C, CHESWER A W, VARTULI J C. On the nature of framework Brønsted and Lewis acid sites in ZSM-5[J]. Zeolites, 1997, 19(4):288-296. [38] TOPSOE N Y, PEDERSEN K, DEROUANE E G. Infrared and temperature-programmed desorption study of the acidic properties of ZSM-5-type zeolites[J]. J Catal, 1981, 70(1):41-52. https://www.sciencedirect.com/science/article/pii/0021951781903158 [39] 童锴, 李军辉, 奚志文, 朱志荣.金属氧化物和硅沉积改性ZnO/SiO2/ZSM-5的甲醇择形芳构化反应催化[J].燃料化学学报, 2015, 43(2):221-227. http://www.ccspublishing.org.cn/article/id/100033287TONG Kai, LI Jun-hui, XI Zhi-wen, ZHU Zhi-rong. Study of methanol shape-selective aromatization over ZnO/SiO2/ZSM-5 modified with mental oxide and silicon deposition[J]. J Fuel Chem Technol, 2015, 43(2):221-227. http://www.ccspublishing.org.cn/article/id/100033287 [40] 刘维桥, 雷卫宁, 尚通明. Zn对HZSM-5催化剂催化剂物化及甲醇芳构化反应性能的影响[J].化工进展, 2011, 30(9):1967-1971. http://d.wanfangdata.com.cn/Periodical_hgjz201109019.aspxLIU Wei-qiao, LEI Wei-ning, SHANG Tong-ming. Physicochemical and methanol aromatization property of HZSM-5 catalyst promoted by Zn[J]. Chem Ind Eng Prog, 2011, 30(9):1967-1971. http://d.wanfangdata.com.cn/Periodical_hgjz201109019.aspx [41] 赵环宇, 柳云骐, 刘平, 张秀斌, 刘晨光.液相沉积硅改性ZSM-5条件对催化甲苯歧化反应的影响[J].中国石油大学学报(自然科学版), 2006, 30(6):117-120. http://d.old.wanfangdata.com.cn/Periodical/sydxxb200606024ZHAO Huan-yu, LIU Yun-qi, LIU Ping, ZHANG Xiu-bin, LIU Chen-guang. Effects of ZSM-5 zeolite catalysts modification conditions by liquid Si deposition on toluene disproportionation reaction[J]. J China University of Petroleum(Nat Sci Edi), 2006, 30(6):117-120. http://d.old.wanfangdata.com.cn/Periodical/sydxxb200606024 [42] ZHU X L, ZHANG J Y, CHENG M, WANG G W, YU M X, LI C Y. Methanol aromatization over Mg-P-modified[Zn, Al]ZSM-5 zeolites for efficient coproduction of para-xylene and light olefins[J]. Ind Eng Chem Res, 2019, 58(42):19446-19455. doi: 10.1021/acs.iecr.9b03743 [43] 张秀斌, 李歧峰, 柳云骐, 刘晨光.磷镁改性的ZSM-5分子筛催化性能研究[J].工业催化, 2004, 12(2):40-45. http://d.old.wanfangdata.com.cn/Periodical/gych200402010ZHANG Xiu-bin, LI Qi-feng, LIU Yu-qi, LIU Chen-guang. Toluene disproportionation over ZSM-5 zeolite modified by phosphorus and magnesium[J]. Ind Catal, 2004, 12(2):40-45. http://d.old.wanfangdata.com.cn/Periodical/gych200402010 [44] LI Y, JUN H. Kinetics study of the isomerization of xylene on ZSM-5 zeolites:the effect of the modification with MgO and CaO[J]. Appl Catal A:Gen, 1996, 142(1):123-137. https://www.sciencedirect.com/science/article/pii/0926860X9600021X [45] NIU X J, GAO J, MIAO Q, DONG M, WANG G F, FAN W B, QIN Z F, WANG J G. Influence of preparation method on the performance of Zn-containing HZSM-5 catalysts in methanol-to-aromatics[J]. Microporous Mesoporous Mater, 2014, 197:252-261. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=7dd90c7a9b1564bcd09ca55038d7ea55 [46] CHANG C D, LANG W H, SMITH R L. The conversion of methanol and other O-compounds to hydrocarbons over zeolite catalysts[J]. J Catal, 1979, 56(2):169-173. https://www.sciencedirect.com/science/article/pii/0021951777901725 [47] 李玲玲, 聂小娃, 宋春山, 郭新闻. H-ZSM-5分子筛催化二甲苯异构化的反应机理[J].物理化学学报, 2013, 29(4):754-762. http://d.old.wanfangdata.com.cn/Periodical/wlhxxb201304017LI Ling-ling, NIE Xiao-wa, SONG Chun-shan, GUO Xin-wen. Isomerization mechanism of xylene catalyzed by H-ZSM-5 molecular sieve[J]. Acta Phys-Chim Sin, 2013, 29(4):754-762. http://d.old.wanfangdata.com.cn/Periodical/wlhxxb201304017 [48] VALECILLOS J, EPELDE E, ALBO J, AGUAYO A T, BILBAO J, CASTANO P. Slowing down the deactivation of H-ZSM-5 zeolite catalyst in the methanol-to-olefin (MTO) reaction by P or Zn modifications[J]. Catal Today, 2019. https://www.sciencedirect.com/science/article/pii/S0920586119304201 [49] KAEDING W W, CHU C, YOUNG L B, WEINSTEIN B, BUTTER S A. Selective alkylation of toluene with methanol to produce para-xylene[J]. J Catal, 1981, 67(1):159-174. https://www.sciencedirect.com/science/article/pii/0021951781902694 [50] YASHIMA T, SAKAGUCHI Y, NAMBA S. Selective formation of p-xylene by alkylation of toluene with methanol on ZSM-5 type zeolites[J]. Stud Surf Sci Catal, 1981, 7(Part A):739-751. doi: 10.1016-S0167-2991(09)60312-8/ [51] 朱杰, 崔宇, 陈元君, 周华群, 王垚, 魏飞.甲醇制烯烃过程研究进展[J].化工学报, 2010, 61(7):1674-1684. http://d.old.wanfangdata.com.cn/Periodical/hgxb201007010ZHU Jie, CUI Yu, CHEN Yuan-jun, ZHOU Hua-qun, WANG Yao, WEI Fei. Recent researches on process from methanol to olefins[J]. J Chem Ind Eng, 2010, 61(7):1674-1684. http://d.old.wanfangdata.com.cn/Periodical/hgxb201007010 [52] 潘红艳, 田敏, 何志艳, 花开慧, 林倩.甲醇制烯烃用ZSM-5催化剂的研究进展[J].化工进展, 2014, 33(10):2625-2633.PAN Hong-yan, TIAN Min, HE Zhi-jie, HUA Kai-hui, LIN Qian. Advances in research on modified ZSM-5 molecular sieves for conversion of methanol to olefins[J]. Chem Ind Eng Prog, 2014, 33(10):2625-2633. [53] TENG H, WANG J, REN X Q, CHEN D M. Disproportionation of toluene by modified ZSM-5 zeolite catalysts with high shape-selectivity prepared using chemical liquid deposition with tetraethyl orthosilicate[J]. Chin J Chem Eng, 2011, 19(2):292-298. http://d.old.wanfangdata.com.cn/Periodical/cjce201102018 -





 下载:
下载: