Effect of reburning temperature on physico-chemical characteristics evolution of rice husk char
-
摘要: 利用高温携带流装置,在再燃条件下获得了不同的稻壳焦。采用元素分析、SEM-EDS分析、N2吸附-脱附、ICP-AES、XRD和FT-IR等手段对稻壳焦的理化结构进行了分析与表征,研究了再燃温度对稻壳再燃过程中灰焦理化结构演化规律的影响。结果表明,在850-1 150 ℃,反应温度的升高有利于稻壳焦孔隙结构的形成,进而有效提高稻壳焦的比表面积和孔容积。稻壳中碱(土)金属元素的释放顺序为Na> Ca> Mg> K,且随温度升高,碱(土)金属元素的释放率呈现先略微升高后逐渐趋于稳定的趋势,氯的释放率逐渐增大。稻壳焦中的碱(土)金属主要以硅酸盐和硫酸盐的形式存在。稻壳焦表面含氧官能团随反应温度的升高逐渐减少。Abstract: Different kinds of rice husk (RH) char were prepared in an entrained flow reactor under reburning conditions. The physico-chemical properties of pyrolyzed RH chars were characterized by scanning electron microscopy-energy dispersive spectrometer (SEM-EDS), specific surface area and pore size analyzer, X-ray diffraction (XRD) and Fourier transform infra-red (FT-IR) spectroscopy, respectively. The effects of temperature on evolution of physico-chemical structures of RH chars were discussed. The results indicate that the specific surface area and pore volume of RH chars increase significantly from 850 to 1 150 ℃. The order of release of alkaline and alkaline earth metals (AAEMs) bound in RH is Na > Ca > Mg > K. With increasing reaction temperature, release fractions of AAEMs generally increase firstly and then being stable, and the chlorine release fraction increases gradually. AAEMs in RH chars are mainly in the form of silicate and sulfate. The surface oxygen-containing functional groups in RH chars decrease significantly with increasing temperature.
-
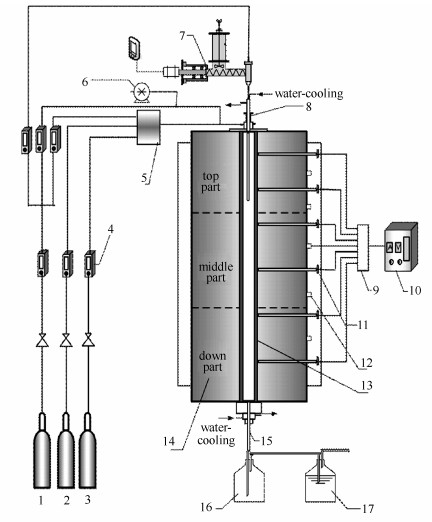
图 1 携带流反应装置示意图
1: N2;2: CO2;3: O2;4: mass flowmeter; 5: mixing container; 6: syringe pump; 7: mini-screw feeder;
8: water-cooling injector; 9: switcher; 10: controller; 11: silicon carbide rod; 12: thermocouples;
13: quartz tube; 14: drop tube furnace(DTF); 15: sampling probe; 16: ash collector; 17: flue gas purificationFigure 1 Experimental setup of the entrained flow reactor
表 1 稻壳的工业分析和元素分析
Table 1 Proximate, ultimate analysis and low heating value of rice husk

表 2 再燃区入口的模拟烟气组分
Table 2 Simulated flue gas component

表 3 稻壳焦的孔隙结构
Table 3 Pore structure parameters of RH chars

表 4 稻壳原料和稻壳焦的元素含量
Table 4 Element contents of RH raw and RH-char

-
[1] YANG H, XU Z, FAN M, BLAND A E, JUDKINS R R. Adsorbents for capturing mercury in coal-fired boiler flue gas[J]. J Hazard Mater, 2007, 146(1/2): 1-11. http://www.ncbi.nlm.nih.gov/pubmed/17544578 [2] SMOOT L D, HILL S C, XU H. NOx control through reburning[J]. Prog Energy Combust Sci, 1998, 24(4): 385-408. http://www.ingentaconnect.com/content/els/03601285/1998/00000024/00000005/art00022 [3] 周强, 段钰锋, 洪亚光, 朱纯, 佘敏, 韦红旗.模拟烟气活性炭喷射脱汞实验研究[J].中国电机工程学报, 2013, 33(35): 36-43. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201335006.htmZHOU Qiang, DUAN Yu-feng, HONG Ya-guang, ZHU Chun, SHE Min, WEI Hong-qi. Experimental study on mercury capture using activated carbon injection in simulated flue gas[J]. Proceedings of the CSEE, 2013, 33(35): 36-43. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201335006.htm [4] LU P, WANG Y, HUANG Z, LU F, LIU Y. Study on NO reduction and its heterogeneous mechanism through biomass reburning in an entrained flow reactor[J].Energy Fuels, 2011, 25(7): 2956-2962. doi: 10.1021/ef2002553 [5] DEMIRBAS A. Combustion characteristics of different biomass fuels[J]. Prog Energy Combust Sci, 2004, 30(2): 219-230. doi: 10.1016/j.pecs.2003.10.004 [6] 费华, 李培生, 孙金丛, 张莹, 罗凯, 张红婴.修正离散随机孔模型应用于两种秸秆生物质焦CO2气化的动力学研究[J].中国电机工程学报, 2016, 36(9): 2459-2464. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=zgdc201609018&dbname=CJFD&dbcode=CJFQFEI Hua, LI Pei-sheng, SUN Jin-cong, ZHANG Ying, LUO Kai, ZHANG Hong-ying. Kinetics of two straw biomass chars gasification with CO2 based a modified discrete random pore model [J]. Proceedings of the CSEE, 2016, 36(9): 2459-2464. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=zgdc201609018&dbname=CJFD&dbcode=CJFQ [7] BIAGINI E, CIONI M, TOGNOTTI L. Development and characterization of a lab-scale entrained flow reactor for testing biomass fuels [J]. Fuel, 2005, 84(12/13): 1524-1534. http://www.sciencedirect.com/science/article/pii/S0016236105000499 [8] 栾积毅. 生物质再燃过程的试验研究及数值模拟[D]. 哈尔滨: 哈尔滨工业大学, 2009. http://cdmd.cnki.com.cn/Article/CDMD-10213-2009292283.htmLUAN Ji-yi. Experimental studies and numerical simulation on reburning process of biomass[D]. Harbin: Harbin Institute of Technology, 2009. http://cdmd.cnki.com.cn/Article/CDMD-10213-2009292283.htm [9] 陈应泉, 朱波, 王贤华, 胥广富, 杨海平, 陈汉平. 生物质热解过程中焦炭物化结构演变特性[J]. 太阳能学报, 2012, 33(8): 1267-1272. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=tylx201208004&dbname=CJFD&dbcode=CJFQCHEN Ying-quan, ZHU Bo, WANG Xian-hua, XU Guang-fu, YANG Hai-ping. Evolutoion of char's physical and chemical structure during biomass pyrolysis[J]. Acta Energy Sol Sin, 2012(8): 1267-1272. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=tylx201208004&dbname=CJFD&dbcode=CJFQ [10] 孟晓晓, 孙锐, 袁皓, 周伟, 任霄汉, 张瑞寒.不同热解温度下玉米秸秆中碱金属K和Na的释放及半焦中赋存特性[J].化工学报, 2017, 68(4): 1600-1667. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ201704040.htmMENG Xiao-xiao, SUN Rui, YUAN Hao, ZHOU Wei, REN Xiao-han, ZHANG Rui-han. Effect of different pyrolysis temperature on alkali metal K and Na emission and existence in semi-char[J]. CIESC J, 2017, 68(4): 1600-1667. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ201704040.htm [11] 肖瑞瑞, 陈雪莉, 王辅臣, 于广锁.不同生物质灰的理化特性[J].太阳能学报, 2011, 32(3): 364-369. http://www.cnki.com.cn/Article/CJFDTOTAL-TYLX201103013.htmXIAO Rui-rui, CHEN Xue-li, WANG Fu-chen, YU Guang-suo. The physicochemical properties on different biomass ash[J]. Acta Energy Sol Sin, 2011, 32(3): 364-369. http://www.cnki.com.cn/Article/CJFDTOTAL-TYLX201103013.htm [12] 杜胜磊, 陈汉平, 杨海平, 胥广富, 王贤华, 张世红.生物质热解过程中F和Cl的迁徙行为研究[J].中国电机工程学报, 2010, 30(14): 115-120. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201014020.htmDU Sheng-lei, CHEN Han-ping, YANG Hai-ping, XU Guang-fu, WANG Xian-hua, ZHANG Shi-hong. Transformation behavior of F and Cl during biomass pyrolysi[J]. Proceedings of the CSEE, 2010, 30(14): 115-120. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201014020.htm [13] 吴鹏, 余春江, 柏继松, 李廉明, 黄芳.生物质热解氯的析出机制研究[J].中国电机工程学报, 2013, 33(11): 75-81. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201311011.htmWU Peng, YU Chun-jiang, BAI Ji-song, LI Lian-ming, HUANG Fang. Mechanism study of chlorine release during biomass pyrolysis[J]. Proceedings of the CSEE, 2013, 33(11): 75-81. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201311011.htm [14] 杨文, 纪晓瑜, 董凯, 宋兴飞, 别如山.生物质燃烧过程中Cl及碱金属K、Na的析出特性[J].太阳能学报, 2015, 36(12): 3072-3075. doi: 10.3969/j.issn.0254-0096.2015.12.034YANG Wen, JI Xiao-yu, DONG Kai, SONG Xing-fei, BIE Ru-shan. Releasing characteristics of chlorine and alkali metals(K and Na)in process of biomass combustions[J]. Acta Energy Sol Sin, 2015, 36(12): 3072-3075 doi: 10.3969/j.issn.0254-0096.2015.12.034 [15] 冯冬冬. 生物质热解焦炭理化特性的研究[D]. 哈尔滨: 哈尔滨工业大学, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10213-1014081700.htmFENG Dong-dong. Rsearch on Physical and Chemical Characteristics of Biomass Pyrolysis Char[D]. Harbin: Harbin Institute of Technology, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10213-1014081700.htm [16] 尹建军, 段钰锋, 王运军, 王卉, 冒咏秋, 韦红旗.生物质焦的表征及其吸附烟气中汞的研究[J].燃料化学学报, 2012, 40(4): 390-396. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17916.shtmlYIN Jian-jun, DUAN Yu-feng, WANG Yun-jun, WANG Hui, MAO Yong-qiu, WEI Hong-qi. Characterization of biomass char and its mercury adsorption performance [J]. J Fuel Chem Technol, 2012, 40(4): 390-396. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17916.shtml [17] JY/T 015-1996, 感耦等离子体原子发射光谱方法通则[S].JY/T 015-1996, General rules for inductively coupled plasma-atomic emission spectrometry[S]. [18] 周军, 张海, 吕俊复, 岳光溪.高温下热解温度对煤焦孔隙结构的研究[J].燃料化学学报, 2007, 35(2): 155-159. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17121.shtmlZHOU Jun, ZHANG Hai, LU Jun-fu, YUE Guang-xi. Effect of pyrolysis temperature on porous structure of anthracite chars produced at high temperatures[J]. J Fuel Chem Technol, 2007, 35(2): 155-159. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17121.shtml [19] 费华, 石金明, 李元林, 刘洋.鄱阳湖水生生物质热解过程中碱金属析出特征[J].有色金属科学与工程, 2017, 8(1): 139-144. http://www.cnki.com.cn/Article/CJFDTOTAL-JXYS201701024.htmFEI Hua, SHI Jin-ming, LI Yuan-lin, LIU Yang. Precipitation characteristics of alkali metal of aquatic biomass in Poyang Lake during pyrolysis[J]. Nonferrous Met Sci Eng, 2017, 8(1): 139-144. http://www.cnki.com.cn/Article/CJFDTOTAL-JXYS201701024.htm [20] 杜胜磊, 杨海平, 钱柯贞, 姚丁丁, 王贤华, 陈汉平.生物质热解过程中碱及碱土金属迁徙规律研究[J].中国电机工程学报, 2013, 33(26): 48-53. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201326009.htmDU Sheng-lei, YANG Hai-ping, QIAN Ke-zhen, YAO Ding-ding, WANG Xian-hua, CHEN Han-ping. Releasing behavior of alkali and alkaline earth metals during biomass pyrolysis[J]. Proceedings of the CSEE, 2013, 33(26): 48-53. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201326009.htm [21] OKUNO T, SONOYAMA N, HAYASHI J, LI C Z, SATHE C, CHIBA T. Primary release of alkali and alkaline earth metallic species during the pyrolysis of pulverized biomass[J]. Energy Fuels, 2005, 19(5): 2164-2171. doi: 10.1021/ef050002a [22] KNUDSEN J N, JENSEN P A, LIN W G, FRANDSEN F J, JOHANSEN K D. Sulfur transformations during thermal conversion of herbaceous biomass[J]. Energy Fuels, 2004, 18(3): 810-819. doi: 10.1021/ef034085b [23] 江龙, 胡松, 宋尧, 黄丹, 向军, 陈刚, 陈文. 生物质快速热解特性研究[J]. 太阳能学报, 2011, 32(12): 1735-1740. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=tylx201112003&dbname=CJFD&dbcode=CJFQJIANG Long, HU Song, SONG Yao, YANG Tao, HUANG Dan, XIANG Jun, CHEN Gang, CHEN Wen. Study on Characteristics of biomass [J]. Acta Energy Sol Sin, 2011, 32(12): 1735-1740. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=tylx201112003&dbname=CJFD&dbcode=CJFQ [24] LI G T, ZHU W Y, ZHU L F, CHAI X Q. Effect of pyrolytic temperature on the adsorptive removal of p-benzoquinone, tetracycline, and polyvinyl alcohol by the biochars from sugarcane bagasse[J]. Korean J Chem Eng, 2016, 33(7): 2215-2221. doi: 10.1007/s11814-016-0067-9 [25] DEMIRBAS A. Mechanisms of liquefaction and pyrolysis reactions of biomass[J]. Energy Convers Manage, 2000, 41(6): 633-646. doi: 10.1016/S0196-8904(99)00130-2 -





 下载:
下载: