Influence of O2 on the formation of As2O3 by homogeneous reaction with As and AsO in the coal-fired flue gas
-
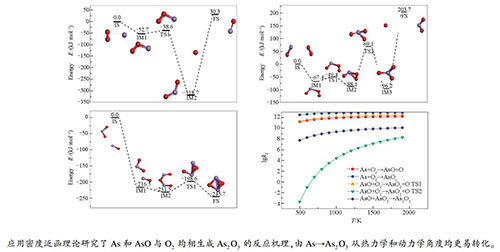
摘要: 应用量子化学密度泛函理论研究了燃煤烟气中As和AsO与O2均相生成As2O3的反应机理。首先计算确定了各反应物、中间体、过渡态和产物的结构和能量,然后运用热力学和动力学方法对As2O3均相生成过程进行分析。结果表明,由As和AsO与O2均相生成As2O3的最大反应能垒分别为32.9和157.2 kJ/mol,在烟气中由As转化为As2O3更为容易进行。在500-1900 K下,各反应的正逆反应速率常数均随温度的提高而增大,但不同反应过程受温度影响的程度不同。As与O2反应生成AsO和AsO2的两个反应过程的平衡常数在所研究的温度范围内均大于105,能完全反应,可以认为是单向反应。AsO与O2反应生成AsO2的过程平衡常数在所研究的温度范围内小于105,反应不完全,转化率低。AsO与AsO2生成As2O3(D3H)构型的平衡常数极低,反应难以进行,而生成As2O3(GAUCHE)构型反应能垒低,可自发进行。Abstract: The reaction mechanism for the formation of As2O3 by the homogeneous reaction of O2 with As and AsO in the coal-fired flue gas was investigated by the quantum chemical density functional theory. The structure and energy of each reactant, intermediate, transition state and product were determined and the thermodynamic and kinetic analysis was carried out to explore the reaction mechanism. The results show that the maximum reaction energy barriers for the formation of As2O3 from As and AsO are 32.9 and 157.2 kJ/mol, respectively. The forward and reverse reaction coefficients all increase with an increase of the reaction temperature in the range of 500-1900 K, although the influence extent of temperature varies with different reactions. For the oxidation of As, the equilibrium constants of two reactions are always greater than 105, indicating that the oxidation of As can be carried out completely and regarded as an irreversible reaction. In contrast, for the oxidation of AsO, the equilibrium constants are always less than 105, indicating that the oxidation of AsO is an incomplete reaction. The equilibrium constant of the As2O3(D3H) configuration is extremely low; however, the formation of the As2O3(GAUCHE) configuration is a spontaneous process.
-
Key words:
- arsenic in flue gas /
- coal combustion /
- homogeneous oxidation /
- reaction mechanism /
- thermodynamics /
- kinetics
-
表 1 键长、键角的计算值及参考值
Table 1 Calculated and referenced bond lengths and bond angles
Species Bond length(r/nm) and angle(θ/(°)) Calculated value Referenced value O2 r(O-O) 1.225 1.239[18] AsO r(As-O) 1.668 1.624[19, 20] AsO2 r(As-O) 1.682 1.775[21] θ(As-O) 126.212 94.655[21] As2O3 (GAUCHE) r(O-As) 1.654 1.610[17] r(As-O) 1.862 1.794[17] θ(O-As-O) 107.1 106.3[17] θ(As-O-As) 128.2 133.8[17] As2O3 (D3H) r(As-O) 1.897 1.836[17] r(As-As) 2.455 2.375[17] θ(As-O-As) 80.6 80.6[18] 表 2 各个反应在不同温度下的平衡常数
Table 2 Equilibrium constants for each reaction at different temperatures
Reaction 500 K 700 K 900 K 1100 K 1300 K 1500 K 1700 K 1900 K (1) 1.20×1027 1.40×1019 5.47×1014 8.55×1011 9.75×109 3.67×108 2.98×107 4.11×106 (2) 2.88×1019 9.10×1013 8.00×1010 9.10×108 4.10×107 4.22×106 7.42×105 1.88×105 (3)TS1 1.63×102 3.94×101 1.79×101 1.08×101 7.64 5.92 4.87 4.17 (3)TS2 7.20×10-1 3.85×10-1 2.74×10-1 2.21×10-1 1.91×10-1 1.72×10-1 1.59×10-1 1.49×10-1 (4) 2.47×10-2 7.94×10-3 4.24×10-3 2.85×10-3 2.17×10-3 1.77×10-3 1.52×10-3 1.35×10-3 -
[1] WANG C, LIU H, ZHANG Y, ZOU C, ANTHONY E J. Review of arsenic behavior during coal combustion:Volatilization, transformation, emission and removal technologies[J]. Prog Energy Combust Sci, 2018, 68:1-28. doi: 10.1016/j.pecs.2018.04.001 [2] 周文颖.周生贤在重金属污染综合防治"十二五"规划视频工作会议上强调坚决打好重金属污染防治攻坚战切实维护人民群众利益和社会稳定[J].环境保护, 2011, (4):12-13. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hjbh201104003ZHOU Wen-ying. Zhou Sheng-xian stressed at the video work conference on the "Twelfth Five-Year Plan" for comprehensive prevention and control of heavy metal pollution, resolutely cracking down on heavy metal pollution prevention and control, and effectively safeguarding the interests of the people and social stability[J]. Environ Protect, 2011, (4):12-13. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hjbh201104003 [3] 环境保护部.砷污染防治技术政策[EB/OL]. http://www.mee.gov.cn/gkml/hbb/bgg/201512/t20151228_320552.htm, 2019-10-07.Ministry of Environmental Protection. Arsenic pollution prevention and control technology policy[EB/OL]. http://www.mee.gov.cn/gkml/hbb/bgg/201512/t20151228_320552.htm, 2019-10-07. [4] 中华人民共和国生态环境部.国家卫生健康委员会关于发布《有毒有害大气污染物名录(2018年)》的公告[EB/OL]. http://www.mee.gov.cn/xxgk2018/xxgk/xxgk01/201901/t20190131_691779.html, 2019-01-25.Ministry of Ecology and Environment. National Health and Wellness Committee on the publication of the Poisonous and Harmful Air Pollutants List (2018)[EB/OL]. http://www.mee.gov.cn/xxgk2018/xxgk/xxgk01/201901/t20190131_691779.html, 2019-01-25. [5] SHEN F, LIU J, ZHANG Z, DAI J. On-line analysis and kinetic behavior of arsenic release during coal combustion and pyrolysis[J]. Environment Sci Technol, 2015, 49(22):13716-13723. doi: 10.1021/acs.est.5b03626 [6] WINTER R M, MALLEPALLI R R, HELLEM K P, SZYDLO S W. Determination of As, Cd, Cr, and Pb species formed in a combustion environment[J]. Combust Sci Technol, 1994, 101(1/6):45-58. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1080/00102209408951865 [7] GERMANI M S, ZOLLER W H. Vapor-phase concentrations of arsenic, selenium, bromine, iodine, and mercury in the stack of a coal-fired power plant[J]. Environ Sci Technol, 1988, 22(9):1079-1085. doi: 10.1021/es00174a013 [8] HIRSCH M E, STERLING R O, HUGGINS F E, HELBLE J J. Speciation of combustion-derived particulate phase arsenic[J]. Environ Eng Sci, 2000, 17(6):315-327. doi: 10.1089/ees.2000.17.315 [9] CONTRERAS M L, AROSTEGUI J M, ARMESTO L. Arsenic interactions during co-combustion processes based on thermodynamic equilibrium calculations[J]. Fuel, 2009, 88(3):539-546. doi: 10.1016/j.fuel.2008.09.028 [10] 刘迎晖, 郑楚光, 游小清, 郭欣.燃煤过程中易挥发有毒痕量元素的相互作用[J].燃烧科学与技术, 2001, 7(4):243-247. doi: 10.3321/j.issn:1006-8740.2001.04.007LIU Ying-hui, ZHENG Chu-guang, YOU Xiao-qing, GUO Xin. Interaction between most volatile toxic trace elements during coal combustion[J]. J Combust Sci Technol, 2001, 7(4):243-247. doi: 10.3321/j.issn:1006-8740.2001.04.007 [11] 刘慧敏, 王春波, 黄星智, 张月, 孙鑫.富氧燃烧方式下煤中砷的挥发行为[J].化工学报, 2015, 66(12):5079-5087. http://d.old.wanfangdata.com.cn/Periodical/hgxb201512047LIU Hui-min, WANG Chun-bo, HUANG Xing-zhi, ZHANG Yue, SUN Xin. Volatilization of arsenic in coal during oxy-fuel combustion[J]. CIESC J, 2015, 66(12):5079-5087. http://d.old.wanfangdata.com.cn/Periodical/hgxb201512047 [12] DU X, TANG J, GAO X, CHEN Y, RAN J, ZHANG L. Molecular transformations of arsenic species in the flue gas of typical power plants:A density functional theory study[J]. Energy Fuels, 2016, 30(5):4209-4214. doi: 10.1021/acs.energyfuels.5b03029 [13] 刘晶, 郑楚光, 邱建荣.燃烧烟气汞反应的量子化学计算方法研究[J].工程热物理学报, 2007, V28(3):519-521. doi: 10.3321/j.issn:0253-231X.2007.03.050LIU Jing, ZHENG Chu-guang, QIU Jian-rong. Studies on quantum chemistry calculation method of mercury reactions in combustion flue gas[J]. J Eng Thermophys, 2007, V28(3):519-521. doi: 10.3321/j.issn:0253-231X.2007.03.050 [14] AWUAH J B, DZADE N Y, TIA R, ADEI E, KWAKYE-AWUAH B, CATLOW C R A. Density functional theory study of arsenic immobilization by Al(Ⅲ)-modified zeolite clinoptilolite[J]. Phys Chem Chem Phys, 2016, 18(16):11297-11305. doi: 10.1039/C6CP00190D [15] ZHANG H, LIU J, SHEN J, JIANG X. Thermodynamic and kinetic evaluation of the reaction between NO (nitric oxide) and char(N) (char bound nitrogen) in coal combustion[J]. Energy, 2015, 82:312-321. doi: 10.1016/j.energy.2015.01.040 [16] ALI M A, RAJAKUMAR B. Thermodynamic and kinetic studies of hydroxyl radical reaction with bromine oxide using density functional theory[J]. Comput Theor Chem, 2011, 964(1/3):283-290. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=744210536dd8e51a33da2b7a1c9fbe58 [17] MONAHAN-PENDERGAST M T, PRZYBYLEK M, LINDBLAD M, WILCOX J. Theoretical predictions of arsenic and selenium species under atmospheric conditions[J]. Atmos Environ, 2008, 42(10):2349-2357. doi: 10.1016/j.atmosenv.2007.12.028 [18] HUBER K, HERZBERG G. Constants of diatomic molecules in NIST chemistry webbook, NIST standard reference database number 69, ed[J]. PJ Linstrom and WG Mallard, 2001. [19] EVENSON K M, WELLS J S, RADFORD H E. Infrared resonance of oh with the H2O laser:A Galactic maser pump?[J]. Phys Rev Lett, 1970, 25(4):199-202. doi: 10.1103/PhysRevLett.25.199 [20] MIZUSHIMA M A. Molecular parameters of OH free radical[J]. Phys Rev A, 1972, 5(1):143-157. doi: 10.1103/PhysRevA.5.143 [21] MONAHAN-PENDERGAST M T, PRZYBYLEK M, LINDBLAD M, WILCOX J. Theoretical predictions of arsenic and selenium species under atmospheric conditions[J]. Atmos Environ, 2008, 42(10):2349-2357. doi: 10.1016/j.atmosenv.2007.12.028 [22] 王泉海, 邱建荣, 温存, 孔凡海, 熊全军, 吴辉.氧燃烧方式下痕量元素形态转化的试验和模拟研究[J].工程热物理学报, 2006, 27(s2):199-202. http://d.old.wanfangdata.com.cn/Periodical/gcrwlxb2006z2052WANG Quan-hai, QIU Jian-rong, WEN Cun, KONG Fan-hai, XIONG Quan-jun, WU Hui.A experimental and simulative study on the morphological transformation of the trace element under oxygen-combustion atmosphere[J]. J Eng Thermophys, 2006, 27(s2):199-202. http://d.old.wanfangdata.com.cn/Periodical/gcrwlxb2006z2052 [23] 汤吉昀.燃煤烟气中砷的存在形式及其在分子筛中的吸附规律研究[D].重庆: 重庆大学, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10611-1017838675.htmTANG Ji-yun. The existing form of arsenic species in coal-fired flue gas and its adsorption by zeolite[D]. Chongqing: Chongqing University, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10611-1017838675.htm [24] 刘晶, 王满辉, 郑楚光, 徐明厚, 李来才, 徐杰英.煤燃烧中汞与含氯气体的反应机理研究[J].工程热物理学报, 2003, V24(1):161-164. doi: 10.3321/j.issn:0253-231X.2003.01.049LIU Jing, WANG Man-hui, ZHENG Chu-guang, XU Ming-hou, LI Lai-cai, XU Jie-ying. Reaction mechanism of mercury and gases during coal combustion[J]. J Eng Thermophys, 2003, V24(1):161-164. doi: 10.3321/j.issn:0253-231X.2003.01.049 -





 下载:
下载: