-
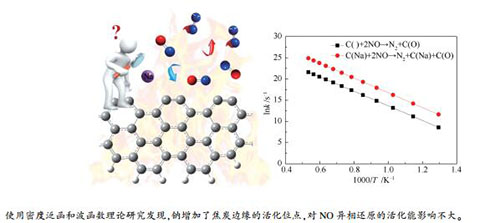
摘要: 基于密度泛函理论研究了钠对焦炭非均相还原NO的微观作用机理。计算结果表明,Na原子可以在焦炭边缘游离,最倾向于吸附在焦炭边缘穴位,释放出174.2 kJ/mol的能量。波函数分析显示,Na原子以强静电吸引的方式与边缘碳原子结合,电子由Na转移到焦炭上。NO以O临近Na原子的方式吸附在焦炭边缘最稳定。Na促进第一个NO分子的吸附,但对第二个NO分子的吸附影响不大。内禀反应坐标计算与Mayer键级分析表明非均相还原通道中Na与O之间经历"结合-分离",通过"氧化-还原"的方式影响反应的进行。N2分子的化学解吸附步是非均相还原的速控步。采用正则变分过渡态理论进行动力学分析,发现Na对速控步的活化能影响不大,但会增加焦炭上活性位点的数量,加快反应的进行。Abstract: A thorough theoretical exploration of microscopic mechanism for effect of sodium (Na) on nitric oxide (NO) heterogeneous reduction by char was performed based on density functional theory with consideration of London dispersion interaction. Calculation results show that the Na atom could migrate at edge of char and prefers to be incorporated into a five-atom ring forming a pentagon with 174.2 kJ/mol released. A strong electrostatic attraction between the Na atom and carbon atoms at the edge is found by reduced density gradient analysis. Electrons transfer from the Na atom to char, resulting in electron rearrangement on char. It is the most stable mode for adsorption of the first NO molecule when O atom in NO molecule is adjacent to the Na atom. The doping of Na could promote adsorption of the first NO molecule, but has little effect on that of the second NO molecule. The intrinsic reaction coordinate calculations and Mayer bond order analyses suggest that the Na atom affects heterogeneous reduction through "oxidized-reduced" cycle via "combination-separation" with the O atom. Desorption of N2 molecule is the rate-determining step in the whole reaction channel. The canonical variational theory was used for kinetic analyses, considering the tunneling effect along the reaction coordinate with Wigner method. It is found that the reaction is accelerated by doping Na atom. Although the addition of Na would not significantly reduce activation energy of the rate-determining step, but would increase activation sites at the edge of char.
-
Key words:
- char /
- nitric oxide /
- heterogeneous reduction /
- sodium /
- density functional theory
-
表 1 拟合所得反应动力学参数
Table 1 Fitted kinetic parameters of Arrhenius expressions
Reaction A/s-1 Ea
/(kJ·mol-1)C()+2NO→N2+C(O) 2.37×1013 148.5 C(Na)+2NO→N2+C(Na)+C(O) 7.04×1014 139.5 -
[1] 王志轩, 刘志强.我国煤电大气污染物控制现状及展望[J].中国工程科学, 2015, 17(9):56-62. http://d.old.wanfangdata.com.cn/Periodical/zggckx201509010WANG Zhi-xuan, LIU Zhi-qiang. Current situation and prospect of control on air pollutants from coal-fired power in China[J]. Eng Sci, 2015, 17(9):56-62. http://d.old.wanfangdata.com.cn/Periodical/zggckx201509010 [2] CHANG S Y, ZHUO J K, MENG S, QIN S Y, YAO Q. Clean coal technologies in China:Current status and future perspectives[J]. Eng, 2016, 2:447-459. https://www.sciencedirect.com/science/article/pii/S2095809917300814 [3] GLARBORG P, MILLER J A, RUSCIC B, KLIPPENSTEIN S J. Modeling nitrogen chemistry in combustion[J]. Prog Energy Combust Sci, 2018, 67:31-68. doi: 10.1016-0360-1285(89)90017-8/ [4] SMOOT L D, HILL S C, XU H. NOx control through reburning[J]. Prog Energy Combust Sci, 1998, 24(5):385-408. http://d.old.wanfangdata.com.cn/Periodical/dndxxb200006014 [5] 刘艳华, 张晓燕, 刘银河, 车得福.再燃煤粉的NO还原特性研究[J].燃料化学学报, 2007, 35(5):523-527. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract17222.shtmlLIU Yan-hua, ZHANG Xiao-yan, LIU Yin-he, CHE De-fu. NO reduction behavior of coal powder used for reburning[J]. J Fuel Chem Technol, 2007, 35(5):523-527. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract17222.shtml [6] 钟北京, 施卫伟, 傅维标.煤粉再燃过程中NO异相还原机理的重要性[J].燃烧科学与技术, 2002, 8(1):6-8. http://d.old.wanfangdata.com.cn/Periodical/rskxyjs200201002ZHONG Bei-jing, SHI Wei-wei, FU Wei-biao. Importance of heterogeneous mechanisms of NO reduction during reburning with pulverized char[J]. J Combust Sci Technol, 2002, 8(1):6-8. http://d.old.wanfangdata.com.cn/Periodical/rskxyjs200201002 [7] 张超群, 姜秀民, 黄庠永, 刘建国.煤焦吸附NO特性与红外光谱分析[J].化工学报, 2007, 58(3):581-586. http://d.old.wanfangdata.com.cn/Periodical/hgxb200703008ZHANG Chao-qun, JIANG Xiu-min, HUANG Xiang-yong, LIU Jian-guo. Characteristics of adsorption of NO gas on coal char and FTIR analysis[J]. CIESC J, 2007, 58(3):581-586. http://d.old.wanfangdata.com.cn/Periodical/hgxb200703008 [8] GUO F, Wu R C, BAXTER L L, HECKER W C. Models to predict kinetics of NOx reduction by chars as a function of coal rank[J]. Energy Fuels, 2019, 33:5498-5504. https://www.researchgate.net/publication/332772724_Models_To_Predict_Kinetics_of_NO_x_Reduction_by_Chars_as_a_Function_of_Coal_Rank [9] 王永桥, 陆飞, 刘永生, 卢平.生物质再燃脱硝及异相还原研究[J].中国电机工程学报, 2010, 30(26):101-106. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgdjgcxb201026018WANG Yong-qiao, LU Fei, LIU Yong-sheng, LU Ping. Study on NOx reduction and its heterogeneous mechanism during biomass reburning[J]. Proc CSEE, 2010, 30(26):101-106. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgdjgcxb201026018 [10] 赵宗彬, 李文, 李保庆.矿物质对煤焦燃烧过程中NO释放规律的影响[J].化工学报, 2003, 54(1):100-106. http://d.old.wanfangdata.com.cn/Periodical/hgxb200301025ZHAO Zong-bin, LI Wen, LI Bao-qing. Effect of mineral matter on release of NO during coal char combustion[J]. CIESC J, 2003, 54(1):100-106. http://d.old.wanfangdata.com.cn/Periodical/hgxb200301025 [11] ZHONG B J, TANG H. Catalytic NO reduction at high temperature by de-ashed chars with catalysts[J]. Combust Flame, 2007, 149:234-243. https://www.sciencedirect.com/science/article/abs/pii/S001021800600109X [12] WANG Z H, ZHOU J H, WEN Z C, LIU J Z, CEN K F. Effect of mineral matter on NO reduction in coal reburning process[J]. Energy Fuels, 2007, 21(4):2038-2043. doi: 10.1021/ef0604902 [13] 信晶, 尹书剑, 孙保民, 朱恒毅, 罗肖, 黄强, 肖海平.掺杂金属化合物强化焦炭-NO反应的析因试验研究[J].煤炭学报, 2015, 40(5):1174-1180.XIN Jing, YIN Shu-jian, SUN Bao-min, ZHU Heng-yi, LUO Xiao, HUANG Qiang, XIAO Hai-ping. Factorial experimental study of Analysis of the char-NO reaction intensified by doped metallic compounds[J]. J China Coal Soc, 2015, 40(5):1174-1180. [14] ZHANG J W, SUN S Z, ZHAO Y J, HU X D, XU G W, QIN Y K. Effects of inherent metals on NO reduction by coal char[J]. Energy Fuels, 2011, 25:5605-5610. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=653cb4f525e02a6e9b4086f0d6c84915 [15] 周昊, 刘瑞鹏, 刘子豪, 程明, 岑可法.碱金属对焦炭燃烧过程中NOx释放的影响[J].煤炭学报, 2015, 40(5):1160-1164. http://d.old.wanfangdata.com.cn/Periodical/mtxb201505028ZHOU Hao, LIU Rui-peng, LIU Zi-hao, CHENG Ming, CEN Ke-fa. Influence of alkali metal on the evolution of NOx during coke combustion[J]. J China Coal Soc, 2015, 40(5):1160-1164. http://d.old.wanfangdata.com.cn/Periodical/mtxb201505028 [16] SØRENSEN C O, JOHNSSON J E, JENSEN A. Reduction of NO over wheat straw char[J]. Energy Fuels, 2001, 15:1359-1368. doi: 10.1021/ef000223a [17] WU X Y, SONG Q, ZHAO H B, YAO Q. Catalytic mechanism of inherent potassium on the char-NO reaction[J]. Energy Fuels, 2015, 29(11):7566-7571. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8df78f24ab76897926b973f6d9ef876c [18] 吕俊复, 柯希玮, 蔡润夏, 张缦, 吴玉新, 杨海瑞, 张海.循环流化床燃烧条件下焦炭表面NOx还原机理研究进展[J].煤炭转化, 2018, 41(1):1-12. (LÜ http://d.old.wanfangdata.com.cn/Periodical/mtzh201801001LV Jun-fu, KE Xi-wei, CAI Run-xia, ZHANG Man, WU Yu-xin, YANG Hai-rui, ZHANG Hai. Research progress on the kinetics of NOx reduction over chars in fluidized bed combustion[J]. Coal Convers, 2018, 41(1):1-12. http://d.old.wanfangdata.com.cn/Periodical/mtzh201801001 [19] 周星宇, 曾凡桂, 相建华, 邓小鹏, 相兴华.马脊梁镜煤有机质大分子模型构建及分子模拟[J].化工学报, 2020, 71(4):1802-1811. http://d.old.wanfangdata.com.cn/Periodical/hgxb202004040ZHOU Xing-yu, ZENG Fan-gui, XIANG Jian-hua, DENG Xiao-peng, XAING Xing-hua. Macromolecular model construction and molecular simulation of organic matter in Majiliang vitrain[J]. CIESC J, 2020, 71(4):1802-1811. http://d.old.wanfangdata.com.cn/Periodical/hgxb202004040 [20] ZHENG M, PAN Y, WANG Z, LI X X, GUO L. Capturing the dynamic profiles of products in Hailaer brown coal pyrolysis with reactive molecular simulations and experiments[J]. Fuel, 2020, 268:117290. https://www.sciencedirect.com/science/article/pii/S0016236120302854 [21] 杨慧芳, 关海莲, 李平, 夏英, 王凤, 徐文静, 白红存, 郭庆杰.煤颗粒燃烧过程氧化机理及有机氮转化的分子模拟:以宁东红石湾煤为例[J].化工学报, 2020, 71(2):799-810. http://d.old.wanfangdata.com.cn/Periodical/hgxb202002037YANG Hui-fang, GUAN Hai-lian, LI Ping, XIA Ying, WANG Feng, XU Wen-jing, BAI Hong-cun, GUO Qing-jie. Molecular modeling of oxidation mechanism and organic nitrogen conversion in coal particle combustion:A case study on HSW coal of Ningdong[J]. CIESC J, 2020, 71(2):799-810. http://d.old.wanfangdata.com.cn/Periodical/hgxb202002037 [22] 许紫阳, 岳爽, 王春波, 刘瑞琪.焦炭催化CO还原NO的反应机理研究[J].燃料化学学报, 2020, 48(3):266-274. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract29530.shtmlXV Zi-yang, YUE Shuang, WANG Chun-bo, LIU Rui-qi. Reaction mechanism of NO reduction with CO catalyzed by char[J]. J Fuel Chem Technol, 2020, 48(3):266-274. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract29530.shtml [23] 王新伟, 马强, 韩超杰, 林日亿.四氢噻吩与硫酸镁热化学还原生成H2S机制[J].中国石油大学学报(自然科学版), 2020, 44(1):156-162.WANG Xin-wei, MA Qiang, HAN Chao-jie, LIN Ri-yi. Study on formation mechanism of H2S by thermochemical sulfate reduction of thiolane and magnesium sulphate[J]. J China Univ Pet, 2020, 44(1):156-162. [24] ZHAO D, LIU H, SUN C, XU L, CAO Q. DFT study of the catalytic effect of Na on the gasification of carbon-CO2[J]. Combust Flame, 2018, 197:471-86. https://www.sciencedirect.com/science/article/pii/S0010218018303924 [25] 陈玉, 张福丽, 姚辉超, 刘植昌, 崔佳, 徐春明.催化水煤气变换反应的计算模拟进展[J].化工进展, 2012, 31(10):2221-2227. http://d.old.wanfangdata.com.cn/Periodical/hgjz201210020CHEN Yu, ZHANG Fu-li, YAO Hui-chao, LIU Zhi-chang, CUI Jia, XU Chun-ming. Progress of theoretical simulation of catalytic water-gas-shift reaction[J]. Chem Ind Eng Prog, 2012, 31(10):2221-2227. http://d.old.wanfangdata.com.cn/Periodical/hgjz201210020 [26] PERRY S T, HAMBLY E M, FLETCHER T H, SOLUM M S, PUGMIRE R J. Solid-state 13C NMR characterization of matched tars and chars from rapid coal devolatilization[J]. Proc Combust Inst, 2000, 28:2313-2319. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=CC028008586 [27] SHENG C. Char structure characterised by Raman spectroscopy and its correlations with combustion reactivity[J]. Fuel, 2007, 86:2316-2324. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=bba4ebe3243ddae2f8939ee65f85eeec [28] 王宝俊, 章丽娜, 凌丽霞, 章日光.煤分子结构对煤层气吸附与扩散行为的影响[J].化工学报, 2016, 67(6):2548-2557. http://d.old.wanfangdata.com.cn/Periodical/hgxb201606048WANG Bao-jun, ZHANG Li-na, LING Li-xia, ZHANG Ri-guang. Effects of coal molecular structure on adsorption and diffusion behaviors of coalbed methane[J]. CIESC J, 2016, 67(6):2548-2557. http://d.old.wanfangdata.com.cn/Periodical/hgxb201606048 [29] 赵鹏飞, 郭欣, 郑楚光.活性炭及氯改性活性炭吸附单质汞的机制研究[J].中国电机工程学报, 2010, 30(23):40-44. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgdjgcxb201023007ZHAO Peng-fei, GUO Xin, ZHENG Chu-guang. Investigation the mechanism of elemental mercury binding on activated carbon and chlorine-embedded activated carbon[J]. Proc CSEE, 2010, 30(23):40-44. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgdjgcxb201023007 [30] 张秀霞, 谢苗, 伍慧喜, 吕晓雪, 林日亿, 周志军.钙对焦炭非均相还原NO的微观作用机理:DFT研究[J].燃料化学学报, 2020, 48(2):163-171. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb202002005ZHANG Xiu-xia, XIE Miao, WU Hui-xi, LÜ Xiao-xue, LIN Ri-yi, ZHOU Zhi-jun. Microscopic effect mechanism of Ca on NO heterogeneous reduction by char:A DFT study[J]. J Fuel Chem Technol, 2020, 48(2):163-171. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb202002005 [31] 陈萍, 顾明言, 汪嘉伦, 卢坤, 林郁郁.含氮煤焦还原NO反应路径研究[J].燃料化学学报, 2019, 47(3):279-286. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201903004CHEN Ping, GU Ming-yan, WANG Jia-lun, LU Kun, LIN Yu-yu. Reaction pathways for the reduction of NO by nitrogen-containing char[J]. J Fuel Chem Technol, 2019, 47(3):279-286. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201903004 [32] 高正阳, 刘晓硕, 李昂, 马传志, 李祥, 杨建蒙.电厂烟气中SO2对活性炭吸附单质铅(Pb0)的影响机理[J].环境科学学报, 2019, 39(11):3732-3739. http://d.old.wanfangdata.com.cn/Periodical/hjkxxb201911019GAO Zheng-yang, LIU Xiao-shuo, LI Ang, MA Chuan-zhi, LI Xiang, YANG Jian-meng. The effect of SO2 on adsorption of element lead toward activated carbon in coal-fired power plants[J]. Acta Sci Circums, 2019, 39(11):3732-3739. http://d.old.wanfangdata.com.cn/Periodical/hjkxxb201911019 [33] ZHANG X X, WU H X, XIE M, LÜ X X, ZHOU Z J, LIN R Y. Wave function and molecular reactivity study of char with different edges and the chemisorption properties of nitric oxide[J]. J Energy Inst, 2020, 93(4):1519-1526. [34] HE K, ROBERTSON A W, FAN Y, ALLEN C H, LIN Y C, SUENAGA K, KIRKLAND A I, WARNER J H. Temperature dependence of the reconstruction of zigzag edges in graphene[J]. ACS Nano, 2015, 9(5):4786-95. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=3e9569acdb6bf8862043748ef16aa4a7 [35] CHEN N, YANG R T. Ab initio molecular orbital calculation on graphite:Selection of molecular system and model chemistry[J]. Carbon, 1998, 36:1061-1070. [36] STEPHENS P, DEVLIN F, CHABALOWSKI C, FRISCH M. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields[J]. J Phys Chem, 1994, 98:11623-11627. [37] 吕泽康, 龙慎伟, 李冠兵, 牛胜利, 路春美, 韩奎华, 王永征.生物质锅炉氯腐蚀的密度泛函理论研究[J].化工学报, 2019, 70(11):4370-4376. (LÜ http://d.old.wanfangdata.com.cn/Periodical/hgxb201911029Ze-kang, LONG Shen-wei, LI Guan-bing, NIU Sheng-li, LU Chun-mei, HAN Kui-hua, WANG Yong-zheng. Density functional theory study on chlorine corrosion of biomass furnace[J]. CIESC J, 2019, 70(11):4370-4376. http://d.old.wanfangdata.com.cn/Periodical/hgxb201911029 [38] GONZALEZ C, SCHLEGEL H B. Reaction path following in mass-weighted internal coordinates[J]. J Phys Chem, 1990, 94:5523-5527. doi: 10.1021-j100377a021/ [39] MERRICK J P, MORAN D, RADOM L. An evaluation of harmonic vibrational frequency scale factors[J]. J Phys Chem A, 2007, 111:11683-11700. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=375cee870c136dd6b808c4026f00bcc2 [40] BURSCH M, CALDEWEYHER E, HANSEN A, NEUGEBAUER H, EHLERT S, GRIMME S. Understanding and quantifying london dispersion effects in organometallic complexes[J]. Acc Chem Res, 2019, 52:258-266. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=ac9493810573a477fd99eee14491c472 [41] GOERIGK L, HANSEN A, BAUER C, EHRLICH S, NAJIBI A, GRIMME S. A look at the density functional theory zoo with the advanced GMTKN55 database for general main group thermochemistry, kinetics and noncovalent interactions[J]. Phys Chem Chem Phys, 2017, 19:32184-32215. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f24b01273034e327ab1e701bc128a2b1 [42] FRISCH M J, TRUCKS G W, SCHLEGEL H B, et al. Gaussian 09[CP]. Revision D.01; Gaussian, Inc., Wallingford CT, 2009. [43] 张守玉, 陈川, 施大钟, 吕俊复, 王健, 董爱霞, 熊绍武.高钠煤燃烧利用现状[J].中国电机工程学报, 2013, 33(5): 1-12.ZHANG Shou-yu, CHEN Chuan, SHI Da-zhong, LÜ Jun-fu, WANG Jian, DONG Ai-xia. Situation of combustion utilization of high sodium coal[J]. Proc CSEE, 2013, 33(5): 1-12. [44] 宋维健, 宋国良, 齐晓宾, 吕清刚.准东高钠煤气化过程中Na的迁移转化规律[J].煤炭学报, 2016, 41(2):490-496. http://d.old.wanfangdata.com.cn/Periodical/mtxb201602030SONG Wei-jian, SONG Guo-liang, QI Xiao-bin, LÜ Qing-gang. Sodium transformation law of Zhundong coal during gasification[J]. J China Coal Soc, 2016, 41(2):490-496. http://d.old.wanfangdata.com.cn/Periodical/mtxb201602030 [45] 魏砾宏, 崔保崇, 陈勇, 杨天华, 郭良振.高碱煤钠赋存形态及其燃烧过程中迁移转化的研究进展[J].燃料化学学报, 2019, 47(8):897-906. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201908001WEI Li-hong, CUI Bao-chong, CHEN Yong, YANG Tian-hua, GUO Liang-zhen. Occurrence of sodium in high alkali coal and its transformation during combustion[J]. J Fuel Chem Technol, 2019, 47(8):897-906. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201908001 [46] 刘磊, 金晶, 林郁郁, 侯封校.钙元素对焦炭表面NO吸附行为的影响:密度泛函理论研究[J].燃料化学学报, 2015, 43(12):1414-1419. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201512002LIU Lei, JIN Jing, LIN Yu-yu, HOU Feng-xiao. Effect of calcium on the absorption of NO on char surface:A density functional theory study[J]. J Fuel Chem Technol, 2015, 43(12):1414-1419. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201512002 [47] 刘吉, 陆强, 蒋晓燕, 胡斌, 董长青, 杨勇平.碱金属离子对吡咯热解生成NOx前驱物HCN机理的影响[J].煤炭学报, 2018, 43(9):2633-2638. http://d.old.wanfangdata.com.cn/Periodical/mtxb201809032LIU Ji, LU Qiang, JIANG Xiao-yan, HU Bin, DONG Chang-qing, YANG Yong-ping. Effect of alkali metal ions on the formation mechanism of HCN as NOx precursor during pyrrole pyrolysis[J]. J China Coal Soc, 2018, 43(9):2633-2638. http://d.old.wanfangdata.com.cn/Periodical/mtxb201809032 [48] JIAO A Y, JIANG X M, LIU J X. Density functional theory investigation on the catalytic reduction of NO by CO on the char surface:the effect of iron[J]. Environ Sci Technol, 2020, 54:2422-2428. [49] ZHANG X X, XIE M, WU H X, LÜ X X, Zhou Z J. DFT study of the effect of Ca on NO heterogeneous reduction by char[J]. Fuel, 2020, 265:116995. [50] MPOURNPAKIS G, FROUDAKIS G. Why alkali metals preferably bind on structural defects of carbon nanotubes:A theoretical study by first principle[J]. J Chem Phys, 2006, 125:204707. [51] FAROKH N A H, ROMAN T, HUSSAIN T, SEARLES D J. Computational study on the adsorption of sodium and calcium on edge-functionalized graphene nanoribbons[J]. J Phys Chem C, 2019, 123:14895-14908. [52] LU T, CHEN F W. Multiwfn:A multifunctional wavefunction analyzer[J]. J Comput Chem, 2012, 33:580-592. http://d.old.wanfangdata.com.cn/Periodical/sdsfdxxb-zr201205006 [53] 邓军, 李亚清, 张玉涛, 杨超萍, 张静, 史学强.羟基(-OH)对煤自燃侧链活性基团氧化反应特性的影响[J].煤炭学报, 2020, 45(1):232-240. http://d.old.wanfangdata.com.cn/Periodical/mtxb202001024DENG Jun, LI Ya-qing, ZHANG Yu-tao, YANG Chao-ping, ZHANG Jing, SHI Xue-qiang. Effects of hydroxyl on oxidation characteristics of side chain active groups in coal[J]. J China Coal Soc, 2020, 45(1):232-240. http://d.old.wanfangdata.com.cn/Periodical/mtxb202001024 [54] JOHNSON E R, KEINAN S, MORI-SÁNCHEZ P, CONTRERAS-GARCÍA J, COHEN A J, YANG W T. Revealing noncovalent interactions[J]. JACS, 2010, 132:6498-6506. [55] 张秀霞, 周志军, 周俊虎, 刘建忠, 岑可法.煤粉再燃中煤焦异相还原NO机理的量化研究[J].燃烧科学与技术, 2011, 17(2):155-159. http://d.old.wanfangdata.com.cn/Periodical/rskxyjs201102010ZHANG Xiu-xia, ZHOU Zhi-jun, ZHOU Jun-hu, LIU Jian-zhong, CEN Ke-fa. A quantum chemistry study of heterogeneous reduction mechanisms of NO on the surface of char during pulverized coal reburning[J]. J Combust Sci Technol, 2011, 17(2):155-159. http://d.old.wanfangdata.com.cn/Periodical/rskxyjs201102010 [56] ZHAO T, SONG W L, FAN C G, LI S G, GLARBORG P, YAO X Q. Density functional theory study of the role of a carbon-oxygen single bond group in the NO-char reaction[J]. Energy Fuels, 2018, 32:7734-7744. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c13c170337c18cc196d87913c4ba3806 [57] MAYER I. Charge, Bond order and valence in the ab initio SCF theory[J]. Chem Phys Lett, 1983, 97:270. [58] GARRETT B C, TRUHLAR D G. Accuracy of tunneling corrections to transition state theory for thermal rate constants of atom transfer reactions[J]. J Phys Chem, 1979, 83(1):200-203. -





 下载:
下载: