Deactivation and regeneration of commercial SCR catalysts used in coal fired power plant
-
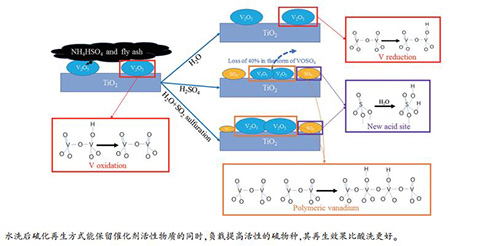
摘要: 对某燃煤电站脱硝系统已运行24000 h的SCR催化剂进行活性测试,利用XRD、BET、XRF、FT-IR和XPS等表征手段分析发现,催化剂出现微、中孔堵塞及活性物质氧化等问题,导致催化剂失活。采用水洗和酸洗方法清除催化剂孔道中的堵塞物,发现水洗能还原部分V物质而酸洗能恢复催化剂表面的活性位并均匀负载具有提高活性位酸性作用的硫物种。对水洗后的催化剂进行SO2硫化处理,发现相比酸洗Brønsted和Lewis酸性位强度提高的同时Lewis酸性位密度也提高。再生处理后,酸洗在250 ℃以上活性恢复到新鲜催化剂水平,450 ℃硫化催化剂活性在380 ℃达到新鲜催化剂活性的104.6%。Abstract: The activity of SCR catalyst used in a coal-fired power plant for 24000 h was tested. With the help of XRD, BET, XRF, FT-IR and XPS, it is found that the catalyst has some problems such as micropore and mesopore plugging and the oxidation of active substances, which leads to the deactivation of the catalyst. Water washing and H2SO4 washing were used to remove plugs in the pores of the catalyst. The result shows that water washing can resume part of vanadium species and H2SO4 washing can renew the active sites on the surface of catalyst and uniformly load the sulfur species which can increase the acidity of active site. The SO2 sulphation treatment was performed on the catalyst after water washing. It is indicated that the Brønsted acid and the Lewis acid sites have higher strength and the Lewis acid has higher density than that by H2SO4 washing. After the regeneration treatment, H2SO4 washed catalyst activity is restored to the level of fresh catalyst above 250 ℃. And the activity of SO2 sulphation catalyst at 450 ℃ reaches 104.6% of the fresh catalyst activity at 380 ℃.
-
Key words:
- SCR catalysts /
- denitrification /
- deactivation /
- regeneration /
- sulphation
-
表 1 失活前后催化剂的XRF分析
Table 1 Chemical composition of the catalysts
Element Fresh catalyst w/% Deactivated catalyst w/% Ti 87.35 77.89 V 1.61 1.51 W 4.84 4.64 Si 3.50 10.55 S 1.01 2.00 Al 0.83 1.31 Fe 0.15 0.24 K 0.09 0.14 Ca 0.62 1.71 表 2 催化剂的XPS分析
Table 2 XPS analysis of catalyst sample
Fresh Deactivated Water washing H2SO4 washing S350 S400 S450 V/(kg·m-3) 2.47 2.36 2.32 1.41 2.21 2.31 2.22 S/(kg·m-3) 2.24 4.75 2.35 6.17 2.96 3.11 4.42 V5+ /% 57.3 75.5 64.2 76.1 56.2 68.5 64.9 V4+ /% 42.7 24.5 35.8 23.9 43.8 31.5 35.1 -
[1] 中华人民共和国环境保护部.中国环境统计年报. 2015[M].中国环境出版社, 2016.Ministry of environmental protection of People's Republic of China. China Environmental Statistics Annual Report. 2015[M]. China Environmental Press, 2016. [2] XIE X, LU J, HUMS E, HUANG Q, LU Z. Study on the deactivation of V2O5-WO3/TiO2 selective catalytic reduction catalysts through transient kinetics[J]. Energy Fuels, 2015, 29(6):3890-3896. doi: 10.1021/acs.energyfuels.5b01034 [3] 唐昊, 陆强, 杨江毅, 李慧, 李文艳, 杨勇平.废弃SCR催化剂的循环再利用及表征分析研究[J].燃料化学学报, 2018, 46(2):233-242. doi: 10.3969/j.issn.0253-2409.2018.02.014TANG Hao, LU Qiang, YANG Jiang-yi, LI Hui, LI Wen-yan, YANG Yong-ping. Research on recycling and characterization analysis of the waste SCR catalyst[J]. J Fuel Chem Technol, 2018, 46(2):233-242. doi: 10.3969/j.issn.0253-2409.2018.02.014 [4] 吴卫红, 吴华, 罗佳, 蒋啸. SCR烟气脱硝催化剂再生研究进展[J].应用化工, 2013, 42(7):1304-1307. http://d.old.wanfangdata.com.cn/Periodical/sxhg201307039WU Wei-hong, WU Hua, LUO Jia, JIANG Xiao. Research progress on the regeneration of SCR catalysts for flue gas denitrification[J]. Appl Chem Ind, 2013, 42(7):1304-1307. http://d.old.wanfangdata.com.cn/Periodical/sxhg201307039 [5] 于艳科, 何炽, 陈进生, 孟小然.电厂烟气脱硝催化剂V2O5-WO3/TiO2失活机理研究[J].燃料化学学报, 2012, 40(11):1359-1365. doi: 10.3969/j.issn.0253-2409.2012.11.013YU Yan-ke, HE Chi, CHEN Jin-sheng, MENG Xiao-ran. Deactivation mechanism of de NOx catalyst (V2O5-WO3/TiO2) used in coal fired power plant[J]. J Fuel Chem Technol, 2012, 40(11):1359-1365. doi: 10.3969/j.issn.0253-2409.2012.11.013 [6] LEE J B, KIM S K, DONG W K, KIM K H, CHUN S N, HUR K B, SANG M J. Effect of H2SO4 concentration in washing solution on regeneration of commercial selective catalytic reduction catalyst[J]. Korean J Chem Eng, 2012, 29(2):270-276. doi: 10.1007/s11814-011-0156-8 [7] 李如冰, 吴玉锋, 章启军, 陈希, 刘彬.关于商用SCR(V2O5-WO3/TiO2)催化剂的再生和回收研究概述[J].现代化工, 2017, (3):29-33. http://xueshu.baidu.com/s?wd=paperuri%3A%281b90db19dabbe29f213b3072de9c499a%29&filter=sc_long_sign&tn=SE_xueshusource_2kduw22v&sc_vurl=http%3A%2F%2Fkns.cnki.net%2FKCMS%2Fdetail%2Fdetail.aspx%3Ffilename%3DXDHG201703007%26dbname%3DCJFD%26dbcode%3DCJFQ&ie=utf-8&sc_us=6345934236335212081LI Ru-bing, WU Yu-feng, ZHANG Qi-jun, CHEN Xi, LIU Bin. A comprehensive review of the regeneration and recovery of commercial SCR catalyst (V2O5-WO3/TiO2)[J]. Mod Chem Ind, 2017, (3):29-33. http://xueshu.baidu.com/s?wd=paperuri%3A%281b90db19dabbe29f213b3072de9c499a%29&filter=sc_long_sign&tn=SE_xueshusource_2kduw22v&sc_vurl=http%3A%2F%2Fkns.cnki.net%2FKCMS%2Fdetail%2Fdetail.aspx%3Ffilename%3DXDHG201703007%26dbname%3DCJFD%26dbcode%3DCJFQ&ie=utf-8&sc_us=6345934236335212081 [8] LI J, TANG X, GAO F, YI H, ZHAO S. Studies on the calcium poisoning and regeneration of commercial De-NOx SCR catalyst[J]. Chem Pap, 2017, 71(10):1921-1928. doi: 10.1007/s11696-017-0186-8 [9] 喻小伟, 周瑜, 刘帅, 宋超. SCR脱硝催化剂失活原因分析及再生处理[J].热力发电, 2014, 43(2):109-113. doi: 10.3969/j.issn.1002-3364.2014.02.109YU Xiao-wei, ZHOU Yu, LIU Shuai, SONG Chao. Reason analysis for deactivation of commercial SCR de-NOx catalyst and its regeneration[J]. Therm Power Gener, 2014, 43(2):109-113. doi: 10.3969/j.issn.1002-3364.2014.02.109 [10] GAO F, TANG X, YI H, ZHAO S, ZHANG T, LI D, MA D. The poisoning and regeneration effect of alkali metals deposed over commercial V2O5-WO3/TiO2 catalysts on SCR of NO by NH3[J]. Sci Bull, 2014, 59(31):3966. doi: 10.1007/s11434-014-0496-y [11] 高凤雨, 唐晓龙, 易红宏, 赵顺征, 李东, 马玎, 张佟佟.商用SCR催化剂的钠中毒及再生[J].中南大学学报(自然科学版), 2015, 46(6):2382-2390. http://d.old.wanfangdata.com.cn/Periodical/zngydxxb201506052GAO Feng-yu, TANG Xiao-long, YI Hong-hong, ZHAO Shun-zheng, LI Dong, MA Ding, ZHANG Tong-tong. Sodium poisoning mechanism and regeneration of commercial De-NOx SCR catalysts[J]. J Cent South Univ Technol, 2015, 46(6):2382-2390. http://d.old.wanfangdata.com.cn/Periodical/zngydxxb201506052 [12] TANABE K, MISONO M, ONO Y, HATTORI H. New solid acids and bases:Their catalytic properties[J]. Stud Surf Sci Catal, 1989, 51:1-365. doi: 10.1016/S0167-2991(08)61044-7 [13] LIETTI L, RAMIS G, BERTI F, TOLEDO G, ROBBA D, BUSCA G, FORZATTI P. Chemical, structural and mechanistic aspects on NOx SCR over commercial and model oxide catalysts[J]. Catal Today, 1998, 42(1/2):101-116. http://cn.bing.com/academic/profile?id=27e37e99e3c84a868ba72eb8271fd01f&encoded=0&v=paper_preview&mkt=zh-cn [14] KHODAYARI R, ODENBRAND C U I. Regeneration of commercial TiO2-V2O5-WO3 SCR catalysts used in bio fuel plants[J]. Appl Catal B:Environ, 2001, 30(1/2):87-99. [15] 沈家铨, 张建华, 邹宜金, 于艳科, 陈进生, 王金秀, 江长水.波纹板式SCR催化剂失活机理及再生研究[J].电力科技与环保, 2016, (3):8-11. doi: 10.3969/j.issn.1674-8069.2016.03.003SHEN Jia-quan, ZHANG Jian-hua, ZOU Yi-jin, YU Yan-ke, CHEN Jin-sheng, WANG Jin-xiu, JIANG Chang-shui. Mechanism of deactivation and regeneration for corrugated type SCR catalysts[J]. Electron Power Environ Protect, 2016, (3):8-11. doi: 10.3969/j.issn.1674-8069.2016.03.003 [16] 张沛, 吴思明, 方拓拓, 陈艳萍, 施耀, 何奕. 660MW燃煤电厂商用SCR催化剂的失活与再生[J].高校化学工程学报, 2017, 31(5):1186-1192. doi: 10.3969/j.issn.1003-9015.2017.05.023ZHANG Pei, WU Si-ming, FANG Tuo-tuo, CHEN Yan-ping, SHI Yao, HE Yi. Deactivation and regeneration of commercial SCR catalysts used in a 660 MW coal-fired power plant[J]. J Chem Eng Chin Univ, 2017, 31(5):1186-1192. doi: 10.3969/j.issn.1003-9015.2017.05.023 [17] NICOSIA D, CZEKAJ I, KRÖCHER O. Chemical deactivation of V2O5/WO3-TiO2 SCR catalysts by additives and impurities from fuels, lubrication oils and urea solution:Part Ⅱ. Characterization study of the effect of alkali and alkaline earth metals[J]. Appl Catal B:Environ, 2008, 77(3/4):228-236. [18] 张道军, 马子然, 孙琦, 徐文强, 李永龙, 王宝冬, 竹涛, 林德海, 季广辉, 马静.硫酸氢铵在钒基选择性催化还原催化剂表面的生成、作用及防治[J].化工进展, 2018, (7):2635-2643. http://d.old.wanfangdata.com.cn/Periodical/hgjz201807021ZHANG Dao-jun, MA Zi-ran, SUN Qi, XU Wen-qiang, LI Yong-long, WANG Bao-dong, ZHU Tao, LIN De-hai, JI Guang-hui, MA Jing. Formation mechanism, effects and prevention of NH4HSO4 formed on the surface of V2O5 based catalysts[J]. Chem Ind Eng Prog, 2018, (7):2635-2643. http://d.old.wanfangdata.com.cn/Periodical/hgjz201807021 [19] KLING Å, ANDERSSON C, MYRINGER Å, ESKILSSON D, JÄRÅS S G. Alkali deactivation of high-dust SCR catalysts used for NOx reduction exposed to flue gas from 100 MW-scale biofuel and peat fired boilers:Influence of flue gas composition[J]. Appl Catal B:Environ, 2007, 69(3/4):240-251. [20] LIU F, ASAKURA K, HE H, SHAN W, SHI X, ZHANG C. Influence of sulfation on iron titanate catalyst for the selective catalytic reduction of NOx with NH3[J]. Appl Catal B:Environ, 2011, 103(3):369-377. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=JJ0220751336 [21] SHANG X, HU G, CHI H, ZHAO J, ZHANG F, XU Y, ZHANG Y, LI J, CHEN J. Regeneration of full-scale commercial honeycomb monolith catalyst (V2O5-WO3/TiO2) used in coal-fired power plant[J]. J Ind Eng Chem, 2012, 18(1):513-519. doi: 10.1016/j.jiec.2011.11.070 [22] CHEN L, LI J, GE M. Promotional effect of Ce-doped V2O5-WO3/TiO2 with low vanadium loadings for selective catalytic reduction of NOx by NH3[J]. J Phys Chem C, 2009, 113(50):21177-21184. doi: 10.1021/jp907109e [23] 段瑞瑞. V~(4+)/V~(5+)比值调变影响因素及其V~(4+)和V~(5+)转化的氧化还原速率与SCR脱硝活性[D].哈尔滨: 工程大学, 2014.DUAN Rui-rui. Influence on modulation of the V4+/5+ ratio and rate of V4+ and V5+ redox and SCR DeNOx activity[D]. Harbin: Engineering University, 2014. [24] 程华. SCR烟气脱硝催化剂失活原因与再生技术的研究[D].广州: 华南理工大学, 2013.CHENG Hua. Study of the deactivation causes and the regeneration methods for commercial V2O5-WO3/TiO2 SCR catalyst[D]. Guangzhou: South China University of Technology, 2013. [25] 马建蓉, 黄张根, 刘振宇, 郭士杰.再生方法对V2O5/AC催化剂同时脱硫脱硝活性的影响[J].催化学报, 2005, 26(6):463-469. doi: 10.3321/j.issn:0253-9837.2005.06.009MA Jian-rong, HUANG Zhang-gen, LIU Zhen-yu, GUO Shi-jie. Effect of regeneration method on activity for simultaneous removal of SO2 and nO over V2O5/AC catalyst-sorbent[J]. Chin J Catal, 2005, 26(6):463-469. doi: 10.3321/j.issn:0253-9837.2005.06.009 [26] 姜烨, 高翔, 吴卫红, 张涌新.选择性催化还原脱硝催化剂失活研究综述[J].中国电机工程学报, 2013, 33(14):18-31. http://d.old.wanfangdata.com.cn/Periodical/zgdjgcxb201314003JIANG Ye, GAO Xiang, WU Wei-hong, ZHANG Yong-xin. Review of the deactivation of selective catalytic reduction DeNOx catalysts[J]. Proc CSEE, 2013, 33(14):18-31. http://d.old.wanfangdata.com.cn/Periodical/zgdjgcxb201314003 [27] PENG Y, LI J, SHI W, XU J, HAO J. Design strategies for development of SCR catalyst:Improvement of alkali poisoning resistance and novel regeneration method[J]. Environ Sci Technol, 2012, 46(22):12623. doi: 10.1021/es302857a [28] BUSCA G, LIETTI L, RAMIS G, BERTI F. Chemical and mechanistic aspects of the selective catalytic reduction of NOx by ammonia over oxide catalysts:A review[J]. Appl Catal B:Environ, 1998, 18(1/2):1-36. http://cn.bing.com/academic/profile?id=9278a98e78dac11b96e4812210c6e554&encoded=0&v=paper_preview&mkt=zh-cn [29] KHODAYARI R, ODENBRAND C U I. Regeneration of commercial SCR catalysts by washing and sulphation:Effect of sulphate groups on the activity[J]. Appl Catal B:Environ, 2001, 33(4):277-291. doi: 10.1016/S0926-3373(01)00193-X -





 下载:
下载: