Effect of CO2 on pyrolysis behavior of Shengli lignite
-
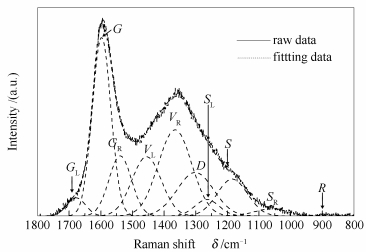
摘要: 以5-8 mm胜利褐煤为研究对象,利用固定床反应器,在400-700℃、CO2气氛下进行热解实验,研究了CO2气氛对煤热解气、液、固三相产物分布的影响,探讨了CO2对煤焦结构作用的机理。研究表明,与N2气氛相比,CO2气氛热解提高焦油和热解水产率,降低热解气和半焦产率;400和500℃时,CO2气氛下形成的半焦孔隙结构和总孔体积没有明显的变化,600和700℃时,CO2气氛下所得半焦的比表面积及孔容较N2气氛下的大,是与煤焦内部挥发分大量释放以及CO2进入孔道与活性位反应有关;煤热解过程中CO2的引入能促进煤焦中3-5环芳香结构的消耗,导致煤焦结构芳香度的提高;600和700℃时,CO2气氛下热解气中H2和CH4产率低,同时CO2能与煤焦发生气化反应显著提高CO含量。Abstract: The pyrolysis of Shengli lignite (5-8 mm) under CO2 atmosphere was investigated at the temperature from 400 to 700℃ in a fixed bed reactor, the effect of CO2 on the distribution of gas, solid and liquid phase in the product was analyzed, and the influence mechanism on the char structure under CO2 atmosphere was explored. The results indicate that the yields of tar and water from pyrolysis under CO2 atmosphere are higher than that under N2 atmosphere, with a decrease in char and gas yield. At 400℃ and 500℃, the surface area and pore volume of the char prepared under CO2 atmosphere are nearly the same as that under N2 atmosphere, while at 600 and 700℃, the surface area and pore volume of the char prepared under CO2 atmosphere are higher than that under N2 atmosphere. It maybe attributes to that CO2 can not only promote the release of volatile, but can also react with the active sites in the channels of char. The introduction of CO2 into pyrolysis can increase the aromaticity of char due to the consumption of 3-5 rings aromatic structures. The yields of H2 and CH4 obtained from pyrolysis under CO2 atmosphere are lower than that obtained under N2 atmosphere at 600 and 700℃, and the yield of CO is much higher because of the occurrence of the char-CO2 reaction.
-
Key words:
- lignite /
- pyrolysis /
- char property /
- CO2 atmosphere
-
表 1 胜利褐煤的工业分析和元素分析
Table 1 Proximate and ultimate analyses of SL coal
Proximate analysis w/% Ultimate analysis wdaf/% Mad Ad Vdaf C H O* N S 21.14 18.62 43.25 70.74 3.68 22.89 1.11 1.59 *:by difference -
[1] 赵虹, 郭飞, 杨建国.印尼褐煤的吸附特性和脱水研究[J].煤炭学报, 2008, 33(7):799-802. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB200807017.htmZHAO Hong, GUO Fei, YANG Jian-guo.Adsorption characteristic of Indonesia Lignite and dewater experiment[J]. J China Coal Soc, 2008, 33(7):799-802. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB200807017.htm [2] YU J H, ARASH T, HAN Y N, YIN F K, LI X C. A review on water in low rank coals:The existence, interaction with coal structure and effects on coal utilization[J]. Fuel Process Technol, 2013, 106(2):9-20. http://www.academia.edu/2479628/A_review_on_water_in_low_rank_coals_The_existence_interaction_with_coal_structure_and_effects_on_coal_utilization [3] ÖZTAZTA N A, YÜRÜM Y. Pyrolysis of Turkish Zonguldak bituminous coal. Part 1. Effect of mineral matter[J]. Fuel, 2000, 79(10):1221-1227. doi: 10.1016/S0016-2361(99)00255-0 [4] 甘建平, 马宝岐, 尚建选, 马晓迅, 杨占彪.煤炭分质转化理念与路线的形成和发展[J].煤化工, 2013, 41(1):3-6. http://www.cnki.com.cn/Article/CJFDTOTAL-MHGZ201301002.htmGAN Jian-ping, MA Bao-qi, SHANG Jian-xuan, MA Xiao-xun, YANG Zhan-biao.Formation and development of the coal grading conversion ideas[J]. Coal Chem Ind, 2013, 41(1):3-6. http://www.cnki.com.cn/Article/CJFDTOTAL-MHGZ201301002.htm [5] 周军, 杨哲, 吴雷, 张秋利, 兰新哲, 尚文智. CO2气氛中低变质煤微波热解研究[J].煤炭学报, 2015, 40(10):2465-2471. http://www.cnki.com.cn/Article/CJFDTotal-MTXB201510034.htmZHOU Jun, YANG Zhe, WU Lei, ZHANG Qiu-li, LAN Xin-zhe, SHANG Wen-zhi.Study on microwave pyrolysis of low rank coal under CO2 atmosphere[J]. J China Coal Soc, 2015, 40(10):2465-2471. http://www.cnki.com.cn/Article/CJFDTotal-MTXB201510034.htm [6] GAO S P, ZHAO J T, WANG Z Q, WANG J F, Fang Y T, HUANG J J. Effect of CO2 on pyrolysis behaviors of lignite[J]. Thin Solid Films, 2013, 41(3):257-264. doi: 10.1016/S1872-5813(13)60017-1 [7] 段伦博, 赵长遂, 周骛, 屈成锐, 李英杰, 陈晓平. CO2气氛对烟煤热解过程的影响[J].中国电机工程学报, 2010, 30(2):62-66. http://www.cnki.com.cn/Article/CJFDTotal-ZGDC201002013.htmDUAN Lun-bo, ZHAO Chang-sui, ZHOU Wu, QU Cheng-rui, LI Ying-jie, CHEN Xiao-ping. Effect of CO2 atmosphere on the pyrolysis process of bituminous coal[J]. Proc CSEE, 2010, 30(2):62-66. http://www.cnki.com.cn/Article/CJFDTotal-ZGDC201002013.htm [8] GUIZANI C, SANZ F J E, SALVADOR S. Effects of CO2 on biomass fast pyrolysis:Reaction rate, gas yields and char reactive properties[J]. Fuel, 2014, 116:310-320. doi: 10.1016/j.fuel.2013.07.101 [9] JAMIL K, HAYASHI J I, LI C Z. Pyrolysis of a Victorian brown coal and gasification of nascent char in CO2 atmosphere in a wire-mesh reactor[J]. Fuel, 2004, 83(7/8):833-843. https://www.researchgate.net/publication/229405521_Pyrolysis_of_a_Victorian_brown_coal_and_gasification_of_nascent_char_in_CO2_atmosphere_in_a_wire-mesh_reactor [10] WANG P F, JIN L J, LIU J H, ZHU S W, HU H Q. Analysis of coal tar derived from pyrolysis at different atmospheres[J]. Fuel, 2013, 104(2):14-21. https://www.researchgate.net/publication/239142429_Analysis_of_coal_tar_derived_from_pyrolysis_at_different_atmospheres [11] LI X J, HAYASHI J I, LI C Z. FT-Raman spectroscopic study of the evolution of char structure during the pyrolysis of a Victorian brown coal[J]. Fuel, 2006, 85(12):1700-1707. https://www.researchgate.net/publication/244067993_FT-Raman_Spectroscopic_Study_of_the_Evolution_of_Char_Structure_During_the_Prolysis_of_a_Victorian_Brown_Coal [12] LI X J, HAYASHI J I, LI C Z. Volatilisation and catalytic effects of alkali and alkaline earth metallic species during the pyrolysis and gasification of Victorian brown coal. Part Ⅶ. Raman spectroscopic study on the changes in char structure during the catalytic gasification in air[J]. Fuel, 2006, 85(10/11):1509-1517. https://www.researchgate.net/publication/244067894_Volatilisation_and_catalytic_effects_of_alkali_and_alkaline_earth_metallic_species_during_the_pyrolysis_and_gasification_of_Victorian_brown_coal_Part_VII_Raman_spectroscopic_study_on_the_changes_in_ch?_sg=2mFg_na9yQ1ZpIXH0Q-8Q9YEXs_ULs2W5ykA8krLhJe5ih0d7x0As0RRJ8aX7qQ_cPusXdffdEiaW3wlPCiNfg [13] ZHANG S, MIN Z H, TAY H L, ASADULLAH M, LI C Z. Effects of volatile-char interactions on the evolution of char structure during the gasification of Victorian brown coal in steam[J]. Fuel, 2011, 90(4):1529-1535. doi: 10.1016/j.fuel.2010.11.010 [14] NAREDI P, PISUPATI S. Effect of CO2 during coal pyrolysis and char burnout in oxy-coal combustion[J]. Energy Fuels, 2011, 25(6):2452-2459. doi: 10.1021/ef200197w [15] CAI H Y, MEGARITIS A, MESSENBÖCK R, DIX M, DUGGWELL D R, KANDIYOTI R. Pyrolysis of coal maceral concentrates under pf-combustion conditions (Ⅰ):Changes in volatile release and char combustibility as a function of rank[J]. Fuel, 1998, 77(12):1273-1282. doi: 10.1016/S0016-2361(98)00039-8 [16] 刘源, 贺新福, 杨伏生, 张亚刚, 任秀彬, 周安宁.热解温度及气氛变化对神府煤热解产物分布的影响[J].煤炭学报, 2015, 40(S2):497-504. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB2015S2028.htmLIU Yuan, HE Xin-fu, YANG Fu-sheng, ZHANG Ya-gang, REN Xiu-bin, ZHOU An-ning. Impacts of pyrolysis temperature and atmosphere on product distribution of Shenfu coal pyrolysis[J]. J China Coal Soc, 2015, 40(S2):497-504. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB2015S2028.htm [17] 丘纪华.煤粉在热分解过程中比表面积和孔隙结构的变化[J].燃料化学学报, 1994, 22(3):316-320. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX403.014.htmQIU Ji-hua. Variation of surface area and pore structure of pulverized coal during pyrolysis[J]. J Fuel Chem Technol, 1994, 22(3):316-320. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX403.014.htm [18] LORENZ H, CARREA E, TAMURA M, HAAS J. The role of char surface structure development in pulverized fuel combustion[J]. Fuel, 2000, 79(10):1161-1172. doi: 10.1016/S0016-2361(99)00259-8 [19] 周毅, 段钰锋, 陈晓平, 赵长遂, 吴新.半焦孔隙结构的影响因素[J].锅炉技术, 2005, 36(4):34-38. http://www.cnki.com.cn/Article/CJFDTOTAL-GLJS200504008.htmZHOU Yi, DUAN Yu-feng, CHEN Xiao-ping, ZHAO Chang-sui, WU Xin. Influential factors on pore structure of coal-char[J]. Boiler Technol, 2005, 36(4):34-38. http://www.cnki.com.cn/Article/CJFDTOTAL-GLJS200504008.htm [20] 白永辉. CO2作为气化剂对煤焦-H2O气化反应的影响机制[D].太原:太原理工大学, 2014.BAI Yong-hui. Influence mechanism of CO2 on char-steam gasification[D]. Taiyuan:Taiyuan University of Technology, 2014. [21] ASADULLAH M, ZHANG S, MIN Z H, YIMSIRI P, LI C Z. Effects of biomass char structure on its gasification reactivity[J]. Bioresour Technol, 2010, 101(20):7935-43. doi: 10.1016/j.biortech.2010.05.048 [22] 申峻, 王志忠.不同煤阶煤炭化过程中挥发分组成及微孔变化的研究[J].煤炭学报, 2007, 32(6):626-629. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB200706016.htmSHEN Jun, WANG Zhi-zhong. Study on variation of micro-pores ( < 100 nm) and volatile components of different rank coals during carbonization[J]. J China Coal Soc, 2007, 32(6):626-629. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB200706016.htm [23] 柳晓飞, 尤静林, 王媛媛, LU Li-ming, 解迎芳, 余立旺, 伏清.澳大利亚烟煤热解的拉曼光谱研究[J].燃料化学学报, 2014, 42(3):270-276. doi: 10.1016/S1872-5813(14)60019-0LIU Xiao-fei, YOU Jing-lin, WANG Yuan-yuan, LU Li-ming, XIE Ying-fang, YU Li-wang, FU Qing. Raman spectroscopic study on the pyrolysis of Australian bituminous coal[J]. J Fuel Chem Technol, 2014, 42(3):270-276. doi: 10.1016/S1872-5813(14)60019-0 [24] 赵丽红, 郭慧卿, 马青兰.煤热解过程中气态产物分布的研究[J].煤炭转化, 2007, 30(1):5-9. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH200701001.htmZHAO Li-hong, GUO Hui-qing, MA Qing-lan. Study on gaseous products distributions during coal pyrolysis[J]. Coal Convers, 2007, 30(1):5-9. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH200701001.htm [25] ARENILLAS A, RUBIERA F, PIS J J. Simultaneous thermogravimetric-mass spectrometric study on the pyrolysis behaviour of different rank coals[J]. J Anal Appl Pyrolysis, 1999, 50(1):31-46. doi: 10.1016/S0165-2370(99)00024-8 [26] 何选明.煤化学[M]. 2版.北京:冶金工业出版社, 2010.HE Xuan-ming. Coal Chemistry[M]. Version 2. Beijing:Metallurgical Industry Press, 2010. [27] 朱学栋, 朱子彬, 唐黎华, 张成芳.煤的热解研究Ⅰ.气氛和温度对热解的影响[J].华东理工大学学报, 1998, 24(1):37-41. http://www.cnki.com.cn/Article/CJFDTOTAL-HLDX801.007.htmZHU Xue-dong, ZHU Zi-bin, TANG Li-hua, ZHANG Cheng-fang. Fundamental study on the pyrolysis of coals[J]. J East China Univ Sci Technol, 1998, 24(1):37-41. http://www.cnki.com.cn/Article/CJFDTOTAL-HLDX801.007.htm -





 下载:
下载: