A review on the migration and transformation of heavy metals influence by alkali/alkaline earth metals during combustion
-
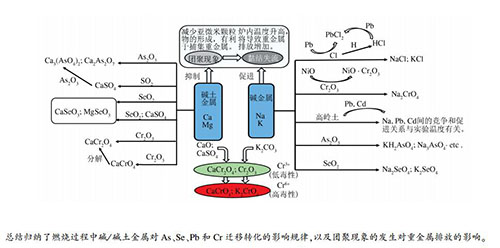
摘要: 碱/碱土金属广泛存在于各种固体燃料中,在燃烧过程中碱/碱土金属与燃料中重金属及其他矿物发生复杂的物理化学反应,从而影响重金属的迁移和转化。本研究主要介绍了碱/碱土金属对As、Se、Pb和Cr四种重金属迁移转化的影响规律,包括碱金属和碱土金属对重金属迁移转化的影响,颗粒物团聚与黏结对重金属排放的影响三个方面。碱/碱土金属能够抑制重金属的挥发:碱金属与Cl元素的结合,降低了PbCl2的生成;碱金属的存在有利于提升高岭土对Pb的吸附效率;碱/碱土金属可以与As和Se形成稳定的化合物。但同时需要注意碱/碱土金属与Cr的部分结合产物中,Cr以六价态存在,具有较高的毒性。碱/碱土金属对于团聚现象发生,分别起到了促进和抑制作用,适当含量的碱金属有利于减少重金属的释放。通过总结碱/碱土金属对重金属迁移转化的影响规律,以期为降低重金属的危害提供思路。Abstract: Alkali/alkaline earth metals(AAEMs) are widely present in various solid fuels. During the combustion process, the AAEMs undergo complex physical and chemical reactions with heavy metals and other minerals in the fuel, thereby affecting the migration and transformation of heavy metals. This paper mainly introduced the influence of AAEMs on the migration and transformation of As, Se, Pb and Cr, including the influence of alkali metals and alkaline earth metals on the migration and transformation of heavy metals, and the influence of particle agglomeration and coherence on heavy metal emissions. AAEMs can inhibit the volatilization of heavy metals. The combination of alkali metals and Cl elements reduces the production of PbCl2. The presence of alkali metals is beneficial to improve the adsorption efficiency of kaolin for Pb. AAEMs can form stable compounds with As and Se. However, at the same time, it should be noted that in the partial combination products of AAEMs and Cr, Cr exists in a hexavalent state and has high toxicity. AAEMs play a role in promoting and inhibiting the occurrence of agglomeration, respectively. An appropriate content of alkali metals is beneficial to reduce the release of heavy metals. By summarizing the influence of AAEMs on the migration and transformation of heavy metals, it is hoped to provide ideas for reducing the harm of heavy metals.
-
Key words:
- combustion /
- alkali metals /
- alkaline earth metals /
- heavy metals /
- adsorption
-
表 1 五种重金属以及氯化物和氧化物的熔点和沸点温度[20]
Table 1 Melting point and boiling point temperature of five trace metals and their chlorides and oxides[20] (with permission from Elsevier)
Metal Element /℃ Chloride /℃ Oxide /℃ melting boiling melting boiling melting boiling Cu 1083 2300 498 1366 1026 1800 Pb 328 1740 501 954 886 - Zn 419 907 283 732 >1850 - Cr 1857 2672 1150 1300 2266 - Ni 1455 2732 987 1001 1980 - -
[1] GEORGE A, SHEN B X, KANG D R, YANG J C, LUO J Z. Emission control strategies of hazardous trace elements from coal-fired power plants in China[J]. Global J Environ Sci, 2020, 93: 66-90. http://www.sciencedirect.com/science/article/pii/S1001074220300693 [2] UDAYANGA W D C, VEKSHA A, GIANNIS A, LISAK G, CHANG V W C, LIM T T. Fate and distribution of heavy metals during thermal processing of sewage sludge[J]. Fuel, 2018, 226: 721-744. http://www.researchgate.net/publication/324913298_Fate_and_distribution_of_heavy_metals_during_thermal_processing_of_sewage_sludge [3] LI W H, SUN Y J, HUANG Y M, SHIMAOKA T, WANG H W, WANG Y N, MA L, ZHANG D L. Evaluation of chemical speciation and environmental risk levels of heavy metals during varied acid corrosion conditions for raw and solidified/stabilized MSWI fly ash[J]. Waste Manag, 2019, 87: 407-416. http://www.sciencedirect.com/science/article/pii/S0956053X19301047 [4] YI H H, HAO J M, DUAN L, TANG X L, NING P, Li X H. Fine particle and trace element emissions from an anthracite coal-fired power plant equipped with a bag-house in China[J]. Fuel, 2008, 87: 10-11. http://www.sciencedirect.com/science/article/pii/S0016236107004383 [5] EPA-HQ-OAR-2009-0234, National emission standards for hazardous air pollutants from coal-and-oil fired electric utility steam generating units and standards of performance for fossil-fuel-fired electric utility, industrial-commercial-institutional, and small industrial-commercial-institutional steam generating units[S]. [6] GB 3095-2012, 环境空气质量标准[S].GB 3095-2012, Ambient air quality standard[S]. [7] ZHANG S R, JIANG X G, LV G J, ABULAITI N X, JIN Y Q, YAN J H, LIN X L, SONG H B, CAO J J. Effect of chlorine, sulfur, moisture and ash content on the partitioning of As, Cr, Cu, Mn, Ni and Pb during bituminous coal and pickling sludge co-combustion[J]. Fuel, 2019, 239: 601-610. http://www.sciencedirect.com/science/article/pii/S0016236118319471 [8] HU H Y, XU Z, LIU H, CHEN D K, LI A J, YAO H, NARUSE I. Mechanism of chromium oxidation by alkali and alkaline earth metals during municipal solid waste incineration[J]. Proc Combust Inst, 2015, 35(2): 2397-2403. http://www.sciencedirect.com/science/article/pii/S1540748914004131 [9] 徐章.城市生活垃圾焚烧飞灰中砷和硒的赋存形态研究[D].武汉: 华中科技大学, 2016.XU Zhang. The study of a rsenic and selenium occurrence in municipal solid waste incineration fly ash[D]. Wuhan: Huazhong University of Science and Technology, 2016. [10] 牟维鹏.不同化学形式硒的毒性作用机制[J].国外医学(卫生学分册), 2001, (4): 202-205+210. http://www.cqvip.com/Main/Detail.aspx?id=5404312MOU Wei-peng. Toxicity mechanism of different chemical forms of selenium[J]. Foreign Med, 2001, (4): 202-205+210. http://www.cqvip.com/Main/Detail.aspx?id=5404312 [11] 蒋志坚, 曾小强, 陈晓平, 梁财.城市污泥流化床焚烧炉飞灰中重金属迁移特性[J].热能动力工程, 2017, 32(9): 92-98+126-127. http://www.cnki.com.cn/Article/CJFDTotal-RNWS201709017.htmJIANG Zhi-jian, ZENG Xiao-qiang, CHEN Xiao-ping, LIANG Cai. Heavy metal migration characteristics of fly ash in fluidized bed incinerator for municipal sewage sludge[J]. Therm Energy Power Eng, 2017, 32(9): 92-98+126-127. http://www.cnki.com.cn/Article/CJFDTotal-RNWS201709017.htm [12] 李园, 陈娟, 张平安, 姚洪.高岭土同时吸附Na, Pb化合物的机理研究[J].工程热物理学报, 2013, 34(1): 168-172. http://www.cnki.com.cn/Article/CJFDTotal-GCRB201301043.htmLI Yuan, CHEN Juan, ZHANG Pin-gan, YAO Hong. Simultaneous a dsorption of Na and Pb compounds by kaolinite[J]. J Eng Thermophys, 2013, 34(1): 168-172. http://www.cnki.com.cn/Article/CJFDTotal-GCRB201301043.htm [13] 杨益, 王敦敦, 陈珣, 姚斌.哈密地区高碱煤掺烧结渣特性综合评价研究[J].煤炭科学技术, 2020, 48(6): 207-213. http://d.wanfangdata.com.cn/periodical/mtkxjs202006027YANG Yi, WANG Dun-dun, CHEN Xun, YAO Bin. Study on comprehensive evaluation of slagging characteristics of high-alkali coal blending combustion from Hami Area[J]. Coal Sci Technol, 2020, 48(6): 207-213. http://d.wanfangdata.com.cn/periodical/mtkxjs202006027 [14] 杨益, 陈晨, 陈珣, 王敦敦, 姚斌.掺烧哈密地区高碱煤沾污特性的试验研究[J].煤炭科学技术, 2019, 47(5): 214-219. http://www.cnki.com.cn/Article/CJFDTotal-MTKJ201905034.htmYANG Yi, CHEN Chen, CHEN Xun, WANG Dun-dun, YAO Bin. Experimental study on fouling characteristics for blending ratio of high alkali coal in Hami area[J]. Coal Sci Technol, 2019, 47(5): 214-219. http://www.cnki.com.cn/Article/CJFDTotal-MTKJ201905034.htm [15] KARIN L, DAN B, ANDERS N, ANDREI S. Fate of Cu, Cr, and As during combustion of impregnated wood with and without peat additive[J]. Environ Sci Technol, 2007, 41(18): 6534-6540. http://europepmc.org/abstract/MED/17948805 [16] 秦跃强, 陈雪莉, 陈汉鼎, 刘海峰.添加CaO对煤热解过程中砷和硫迁移转化的影响[J].燃料化学学报, 2017, 45(2): 147-156. doi: 10.3969/j.issn.0253-2409.2017.02.003QIN Yue-qiang, CHEN Xue-li, CHEN Han-ding, LIU Hai-feng. Effects of adding CaO on the release and transformation of arsenic and sulfur during coal pyrolysis[J]. J Fuel Chem Technol, 2017, 45(2): 147-156. doi: 10.3969/j.issn.0253-2409.2017.02.003 [17] 李耀德, 杨冬, 周熙宏, 董乐, 王思洋.污泥与高碱煤协同燃烧中碱金属与重金属竞争机制研究[J].动力工程学报, 2020, 40(5): 349-355+364. http://www.cqvip.com/QK/95606A/202005/7102107797.htmlLI Yao-de, YANG Dong, ZHOU Xi-hong, DONG Le, WANG Si-yang. Study on competition mechanism of alkali metals and heavy metals in collaborative combustion of sludge and alkali coal[J]. Chin J Power Eng, 2020, 40(5): 349-355+364. http://www.cqvip.com/QK/95606A/202005/7102107797.html [18] 梅艳钢, 王志青, 高松平, 郑洪岩, 张郃, 房倚天.碱金属与碱土金属在煤炭热转化过程中的影响研究进展[J].燃料化学学报, 2020, 48(4): 385-394. doi: 10.3969/j.issn.0253-2409.2020.04.001MEI Yan-gang, WANG Zhi-qing, GAO Song-ping, ZHENG Hong-yan, ZHANG Yun, FANG Yi-tian. Research progress of the influence of alkali metals and alkaline earth metals on coal thermal chemical conversion[J]. J Fuel Chem Technol, 2020, 48(4): 385-394. doi: 10.3969/j.issn.0253-2409.2020.04.001 [19] WENDT J O L, LEE S J. High-temperature sorbents for Hg, Cd, Pb, and other trace metals: Mech Appl[J]. Fuel, 2009, 89(4): 894-903. https://www.sciencedirect.com/science/article/pii/S0016236109000416 [20] LIU J Y, FU J W, NING X A, SUN S Y, WANG Y J, XIE W M, HUANG S S, ZHONG S. An experimental and thermodynamic equilibrium investigation of the Pb, Zn, Cr, Cu, Mn and Ni partitioning during sewage sludge incineration[J]. J Environ Sci, 2015, 35(9): 43-54. http://smartsearch.nstl.gov.cn/paper_detail.html?id=c0afa1c3ef544373c5e64224041a0d7f [21] WANG X Y, HUANG Y J, NIU M M, WANG Y X, LIU C Q. Effect of multi-factors interaction on trace lead equilibrium during municipal solid waste incineration[J]. J Mater Cycles Waste Manage, 2016, 18(2): 287-295. http://smartsearch.nstl.gov.cn/paper_detail.html?id=1bcbc7235b21c6d9626c60446f9b0143 [22] WANG K S, CHIANG K Y, LIN S M, TSAI C C, SUN C J. Effects of chlorides on emissions of toxic compounds in waste incineration: Study on partitioning characteristics of heavy metal[J]. Chemosphere, 1999, 38(8): 1833-1849. http://www.ncbi.nlm.nih.gov/pubmed/10101850/ [23] 余婉璇, 刁永发, 沈恒.生物质与煤混燃过程中碱金属对汞氧化影响的热力学研究[J].中国电机工程学报, 2012, 32(26): 96-102+153. http://d.wanfangdata.com.cn/Periodical/zgdjgcxb201226015YU Wan-xuan, DIAO Yong-fa, SHEN Heng. Thermodynamics study of effects of alkali metals on mercury transformation during co-combustion of biomass with coal[J]. Chin Soc Electr Eng, 2012, 32(26): 96-102+153. http://d.wanfangdata.com.cn/Periodical/zgdjgcxb201226015 [24] 姚佳彬.钠化合物对燃煤氮氧化物和汞排放的影响研究[D].北京: 华北电力大学, 2016.YAO Jia-bin. Effects of sodium compounds on nitrogen oxides and mercury releasing during coal combustion[D]. Beijing: North China Electric Power University, 2016. [25] WEI X L, SCHNELL U, HEIN K R G. Behaviour of gaseous chlorine and alkali metals during biomass thermal utilisation[J]. Fuel, 2005, 84(7): 841-848. http://www.sciencedirect.com/science/article/pii/S0016236104003667 [26] LIN C L, WEY M Y, YU W J. Emission characteristics of organic and heavy metal pollutants in fluidized bed incineration during the agglomeration/defluidization process[J]. Combust Flame, 2005, 143(3): 139-149. http://www.sciencedirect.com/science/article/pii/S0010218005001458 [27] CHEN T, LEI C, YAN B, LI L L. Analysis of heavy metals fixation and associated energy consumption during sewage sludge combustion: Bench scale and pilot test[J]. J Cleaner Prod, 2019, 229: 1243-1250. http://www.sciencedirect.com/science/article/pii/S0959652619314593 [28] WANG X Y, HUANG Y J, ZHONG Z P, YAN Y P, NIU M M, WANG Y X. Control of inhalable particulate lead emission from incinerator using kaolin in two addition modes[J]. Fuel Process Technol, 2014, 119: 228-235. http://www.sciencedirect.com/science/article/pii/S0378382013003640 [29] SI J P, LIU X W, XU M H, SHENG L, ZHOU Z J, WANG C, ZHANG Y, SEO Y C. Effect of kaolin additive on PM 2.5 reduction during pulverized coal combustion: Importance of sodium and its occurrence in coal[J]. Appl Energy, 2014, 114: 434-444. http://www.sciencedirect.com/science/article/pii/S0306261913008222 [30] YAO H, NARUSE I. Using sorbents to control heavy metals and particulate matter emission during solid fuel combustion[J]. Particuology, 2009, 7(6): 477-482. http://www.sciencedirect.com/science/article/pii/S1674200109000790 [31] GALE T K, WENDT L O L. High-temperature interactions between multiple-metals and kaolinite[J]. Combust Flame, 2003, 133(3): 383-383. http://www.sciencedirect.com/science/article/pii/S0010218002004042 [32] GALE T K, WENDT L O L. In-furnace capture of cadmium and other semi-volatile metals by sorbents[J]. Proc Combust Inst, 2005, 30(2): 2999-3007. http://www.sciencedirect.com/science/article/pii/S0082078404002498 [33] DAVIS S B, GALE T K, WENDT L O L. Competition for sodium and toxic metals capture on sorbents[J]. Aerosol Sci Technol, 2000, 32(2): 142-151. doi: 10.1080/027868200303849 [34] JAGODZINSKA K, MROCZEK K, NOWINSKA K, GOLOMBEK K, KALISZ S. The impact of additives on the retention of heavy metals in the bottom ash during RDF incineration[J]. Energy, 2019, 183: 854-868. http://www.sciencedirect.com/science/article/pii/S0360544219312940 [35] CONTRERAS M L, AROSTEGUI J M, ARMESTO L. Arsenic interactions during co-combustion processes based on thermodynamic equilibrium calculations[J]. Fuel, 2008, 88(3): 539-546. http://www.sciencedirect.com/science/article/pii/S0016236108003918 [36] ZHOU C C, LIU G J, XU Z Y, SUN H, LAM P K S. Retention mechanisms of ash compositions on toxic elements (Sb, Se and Pb) during fluidized bed combustion[J]. Fuel, 2018, 213: 98-105. http://www.sciencedirect.com/science/article/pii/S0016236117313674 [37] ZHOU C C, LIU G J, XU Z Y, SUN H, LAM P K S. Effect of ash composition on the partitioning of arsenic during fluidized bed combustion[J]. Fuel, 2017, 204: 91-97. http://www.sciencedirect.com/science/article/pii/S001623611730618X [38] 赵云.碱金属与污泥中重金属迁移的竞争机制研究[D].沈阳: 沈阳航空航天大学, 2012.ZHAO Yun. Influences of alkali metals on heavy metals migration and competition in sewa ge sludge[D]. Shenyang: Shenyang University of Aeronautics and Astronautics, 2012. [39] CHEN D K, HU H Y, XU Z, LIU H, CAO J X, SHEN J H, YAO H. Findings of proper temperatures for arsenic capture by CaO in the simulated flue gas with and without SO2[J]. Chem Eng J, 2015, 267: 201-206. http://www.sciencedirect.com/science/article/pii/S1385894715000649 [40] LI Y Z, TONG H L, ZHUO Y Q, LI Y, XU X C. Simultaneous removal of SO2 and trace As2O3 from flue gas: Mechanism, kinetics study, and effect of main gases on arsenic capture[J]. Environ Sci Technol, 2007, 41(8): 2894. doi: 10.1021/es0618494 [41] JADHAV R A, FAN L S. Capture of gas-phase arsenic oxide by lime: kinetic and mechanistic studies[J]. Environ Sci Technol, 2001, 35(4): 794-799. http://europepmc.org/abstract/MED/11349294 [42] DIAZ S M, MARTINEZ T M R. Retention of arsenic and selenium compounds using limestone in a coal gasification flue gas[J]. Environ Sci Technol, 2004, 38(3): 899-903. doi: 10.1021/es034344b [43] SONG B, SONG M, CHEN D D, CAO Y, MENG F Y, WEI Y X. Retention of arsenic in coal combustion flue gas at high temperature in the presence of CaO[J]. Fuel, 2020, 259: 116249.1-116249.7. http://www.sciencedirect.com/science/article/pii/S0016236119316035 [44] XU S R, SHUAI Q, HUANG Y J, BAO Z Y, HU S H. Se capture by a CaO-ZnO composite sorbent during the combustion of Se-rich stone coal[J]. Energy Fuels, 2013, 27: 6880-6886. doi: 10.1021/ef4013449 [45] FAN Y M, ZHUO Y Q, LOU Y, ZHU Z W, LI L L. SeO2 adsorption on CaO surface: DFT study on the adsorption of a single SeO2 molecule[J]. Appl Surf Sci, 2017, 413: 366-371. http://www.sciencedirect.com/science/article/pii/S0169433217308772 [46] FAN Y M, ZHUO Y Q, LI L L. SeO2 adsorption on CaO surface: DFT and experimental study on the adsorption of multiple SeO2 molecules[J]. Appl Surf Sci, 2017, 420: 465-471. http://adsabs.harvard.edu/abs/2017ApSS..420..465F [47] LI Y Z, TONG H L, ZHUO Y Q, CHEN C H, XU X C. Simultaneous removal of SO2 and trace SeO2 from flue gas: effect of product layer on mass transfer[J]. Environ Sci Technol, 2006, 40(13): 4306. http://www.tandfonline.com/servlet/linkout?suffix=CIT0095&dbid=8&doi=10.1080%2F00206814.2017.1362671&key=16856751 [48] LI Y Z, TONG H L, ZHUO Y Q, WANG S J, XU X C. Simultaneous removal of SO2 and trace SeO2 from flue gas: effect of SO2 on selenium capture and kinetics study[J]. Environ Sci Technol, 2006, 40(24): 7919-7924. http://www.tandfonline.com/servlet/linkout?suffix=CIT0095&dbid=8&doi=10.1080%2F00206814.2017.1362671&key=16856751 [49] KUO J H, LIN C L, WEY M Y. Effect of particle agglomeration on heavy metals adsorption by Al- and Ca-based sorbents during fluidized bed incineration[J]. Fuel Process Technol, 2011, 92(10): 2089-2098. http://www.sciencedirect.com/science/article/pii/S0378382011002347 [50] CHEN J, JIAO F C, ZHANG L, YAO H, NINOMIYA Y. Elucidating the mechanism of Cr(Ⅵ) formation upon the interaction with metal oxides during coal oxy-fuel combustion[J]. J Hazard Mater, 2013, 261: 260-268. http://europepmc.org/abstract/med/23969010 [51] HU H Y, SHI M Y, YANG Y H, LIU H, XU M, SHEN J H, YAO H. Further insight into the formation and oxidation of CaCr2O4 during solid fuel combustion[J]. Environ Sci Technol, 2018, 52(4): 2385-2391. http://www.ncbi.nlm.nih.gov/pubmed/29390188 [52] YAO H, MKILAHA I S N, NARUSE I. Screening of sorbents and capture of lead and cadmium compounds during sewage sludge combustion[J]. Fuel, 2003, 83(7): 1001-1007. http://www.sciencedirect.com/science/article/pii/S0016236103003533 [53] ZHA J R, HUANG Y J, XIA W Q, XIA Z P, LIU C Q, DONG L, LIU L Q. Effect of mineral reaction between calcium and aluminosilicate on heavy metal behavior during sludge incineration[J]. Fuel, 2018, 229: 241-247. http://www.sciencedirect.com/science/article/pii/S001623611830838X [54] ZHOU H, SUN J, MENG A H, LI Q H, ZHANG Y G. Effects of sorbents on the partitioning and speciation of Cu during municipal solid waste incineration[J]. Chin J Chem Eng, 2014, 22(11/12): 1347-1351. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cjce201411028 [55] CHEN L M, LIAO Y F, MA X Q. Heavy metals volatilization characteristics and risk evaluation of co-combusted municipal solid wastes and sewage sludge without and with calcium-based sorbents[J]. Ecotoxicol Environ Saf, 2019, 182: 109370. http://www.sciencedirect.com/science/article/pii/S0147651319306918 [56] BARTONOVA L, KLIKA Z. Effect of CaO on retention of S, Cl, Br, As, Mn, V, Cr, Ni, Cu, Zn, W and Pb in bottom ashes from fluidized-bed coal combustion power station[J]. J Environ Sci, 2014, 26(7): 1429-1436. http://d.wanfangdata.com.cn/Periodical/jes-e201407008 [57] ZHA J R, HUANG Y J, CLOUGH P T, DONG L, XU L G, LIU L Q, ZHU Z C, YU M Z. Desulfurization using limestone during sludge incineration in a fluidized bed furnace: Increased risk of particulate matter and heavy metal emissions[J]. Fuel, 2020, 273: 117614. https://www.sciencedirect.com/science/article/pii/S0016236120306098 [58] LOPES M H, ABELHA P, OLIVEIRA J F S, CABRITA I, GULYURTLU I. Heavy Metals Behavior during Monocombustion and Co-Combustion of Sewage Sludge[J]. Environ Eng Sci, 2005, 22(2): 205-220. http://www.researchgate.net/publication/245336387_Heavy_Metals_Behavior_during_Monocombustion_and_Co-Combustion_of_Sewage_Sludge [59] 郭家宏, 林坤森, 熊坤, 林秋良, 刘敬勇.碱金属对固废焚烧燃烧特性及重金属排放行为的影响研究[J].环境工程, 2018, 36(5): 132-138. http://www.cnki.com.cn/Article/CJFDTotal-HJGC201805028.htmGUO Jia-hong, LIN Kun-sen, XIONG Kun, LIN Qiu-liang, LIU Jing-yong. Effect of alkalic metal on the combustion characteristics, agglomeration/defluidization and heavy metals emission during fluidized bed municipal solid waste (MSW) incineration[J]. Environ Eng, 2018, 36(5): 132-138. http://www.cnki.com.cn/Article/CJFDTotal-HJGC201805028.htm [60] GAO Z F, LONG H M, DAI B, GAO X P. Investigation of reducing particulate matter (PM) and heavy metals pollutions by adding a novel additive from metallurgical dust (MD) during coal combustion[J]. J Hazard Mater, 2019, 373: 335-346. http://www.ncbi.nlm.nih.gov/pubmed/30928676 [61] KUO J H, LIN C L, WEY M Y. Effects of agglomeration processes on the emission characteristics of heavy metals under different waste compositions and the addition of Al and Ca inhibitors in fluidized bed incineration[J]. Energy Fuels, 2009. 23(9): 4325-4336. http://www.researchgate.net/publication/231274239_Effects_of_Agglomeration_Processes_on_the_Emission_Characteristics_of_Heavy_Metals_under_Different_Waste_Compositions_and_the_Addition_of_Al_and_Ca_Inhibitors_in_Fluidized_Bed_Incineration [62] ZHAO S L, DUAN Y F, LI Y N, LIU M, LU J H, DING Y J, GU X B, TAO J, DU M S. Emission characteristic and transformation mechanism of hazardous trace elements in a coal-fired power plant[J]. Fuel, 2018, 214: 597-606. http://forest.ckcest.cn/d/hxwx/dcad0fcff1f1d177ac4fea352ae92600.html -





 下载:
下载: