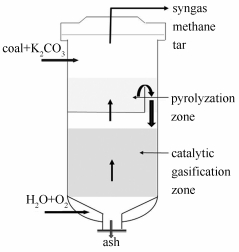
Effects of different atmospheres on sulfur transformation during K2CO3 catalytic pressurized pyrolysis of coal
-
摘要: 在加压热解装置上,考察了碳酸钾及热解气氛对煤热解过程中硫分布及其形态的影响。结果表明,碳酸钾通过捕获H2S增加了半焦硫含量,同时可将煤焦表面活化,导致煤中有机质与黄铁矿分解产生的活泼硫结合形成新的有机硫。氢气能促进煤中硫的脱除,但是碳酸钾存在下热解释放的硫一部分以K2S的形式固定于半焦中。水蒸气可显著促进煤中黄铁矿的分解,同时可与煤焦中的K2S反应,降低半焦中的硫含量。两段床催化气化炉中,碳酸钾催化剂经热解后不影响其对煤焦的催化性能。Abstract: The effects of K2CO3 and atmospheres on sulfur distribution and forms during pressurized coal pyrolysis were investigated in a lab-scale reactor. The results show that K2CO3 increases the sulfur remained in char by capturing H2S during coal pyrolysis. The surface of the coal char is activated in the presence of K2CO3, and then the nascent sulfur releasing from the pyrite is captured by the organic matrix in coal to form new organic sulfur. Hydrogen shows positive effects on removal of pyrite and organic sulfur, and the released H2S is partly fixed in char in the form of K2S when K2CO3 was added. The decomposition of pyrite is enhanced in H2O atmosphere. Meanwhile, K2S in char can not exist due to effect of water vapour, so the sulfur remained in char is notably reduced. The catalytic property of K2CO3 on char gasification is not changed after pyrolysis in two-stage catalytic coal gasifier.
-
图 2 煤加压热解装置示意图
Figure 2 Schematic diagram of the pressurized coal pyrolysis equipment
1: hopper; 2: electric furnace; 3: fixed-bed reactor; 4: thermocouple; 5: flange; 6: steam generator; 7: water storage tank; 8: high-pressure water pump; 9: mass flowmeter; 10: the primary cooler; 11: gas-liquid separator; 12: ethanediol; 13: circulating pump; 14: n-hexane second-stage cooler; 15: quenching trap; 16: micrometering valve; 17: wet type gas flowmeter; 18: gas bag
表 1 煤质分析
Table 1 Analysis of coals
Sample Proximate analysis wd/% Ultimate analysis wd/% Sulfur forms wd/% A V FC C H N St Sp Ss So BLG 14.26 31.10 54.64 66.24 3.99 1.04 0.63 0.38 0.04 0.22 ND 17.42 32.41 50.17 64.14 4.25 0.75 2.36 1.18 0.42 0.67 BLG: Buliangou coal;ND: Ningdong coal;Sp: pyrite sulfur;Ss: sulfatic sulfur;So: orgnic sulfur 表 2 灰成分分析
Table 2 Analysis of coal ash
Sample Content w/% Al2O3 CaO Fe2O3 K2O MgO Na2O P2O5 SO3 TiO2 SiO2 BLG 53.10 2.37 1.89 1.44 0.55 0.34 0.54 4.21 2.51 26.65 ND 15.58 12.01 9.40 1.91 2.96 2.17 0.08 13.57 0.71 38.52 表 3 碳酸钾及气氛对热解的影响
Table 3 Effect of K2CO3 and atmospheres on pyrolysis
Sample# Char yield wd/% Tar yield wd/% BLG-R-N 81.96 5.81 BLG-C-N 79.52 5.55 BLG-R-N (640 ℃) 76.89 5.43 BLG-C-N (640 ℃) 73.67 5.37 BLG-R-H 79.90 7.26 BLG-C-N 75.86 6.84 BLG-R-W 81.05 6.15 BLG-C-W 76.31 6.01 BLG-R-H+W 75.04 7.62 ND-C-N 79.24 4.81 ND-C-H 74.35 7.73 ND-C-W 75.73 5.75 ND-C-H+W 74.74 7.26 #: sample code A-B-C means that the sample was obtained by pyrolysis of coal A (BLG=Buliangou coal, ND=Ningdong coal) in atmosphere C (N=N2, H=H2, W=H2O, H+W=H2+ H2O) with the addition of B (R=raw coal, C=K2CO3) 表 4 不连沟煤热解半焦中的形态硫的分布(3.5 MPa)
Table 4 Distribution of sulfur forms in char of BLG coal (3.5 MPa)
Sample Atmospheres Temperature t/℃ Sp/St(%) So/St(%) Ssulfide/St(%) BLG-coal - - 59.38 34.38 - BLG-char N2 530 26.12 27.30 2.93 BLG+10%K2CO3-char N2 530 24.88 35.54 13.62 BLG-char N2 640 12.97 32.56 4.93 BLG+10%K2CO3-char N2 640 15.05 38.65 22.22 St: the total sulfur of feed stock (same below) 表 5 煤热解半焦中的形态硫分布(3.5 MPa-530 ℃)
Table 5 Distribution of sulfur forms in char of coals (3.5 MPa-530 ℃)
Atmospheres Sp/St(%) So/St(%) Ssulfide/St(%) BLG-char N2 26.12 27.30 2.93 BLG-char 70%N2+30%H2 20.97 14.01 0.47 BLG+10%K2CO3-char N2 24.88 35.54 13.62 BLG+10%K2CO3-char 70%N2+30%H2 21.98 15.39 30.70 ND+10%K2CO3-char N2 31.11 36.34 11.65 ND+10%K2CO3-char 70%N2+30%H2 11.32 16.59 37.12 表 6 煤热解半焦中的形态硫分布(3.5 MPa-530 ℃)
Table 6 Distribution of sulfur forms in char of coals (3.5 MPa-530 ℃)
Sample Atmospheres Sp/St(%) So/St(%) Ssulfide/St(%) BLG-char N2 26.12 27.30 2.93 BLG+10%K2CO3-char N2 24.88 35.54 13.62 BLG-char 70%N2+30%H2O 13.15 27.49 3.41 BLG+10%K2CO3-char 70%N2+30%H2O 19.12 27.62 0.31 ND+10%K2CO3-char N2 31.11 36.34 11.65 ND+10%K2CO3-char 70%N2+30%H2O 13.41 24.41 11.24 表 7 10%K2CO3-煤热解半焦中的形态硫的分布
Table 7 Distribution of sulfur forms in char of 10%K2CO3-coals
Sample Atmospheres Sp/St(%) So/St(%) Ssulfide/St(%) BLG+10%K2CO3-char N2+30%H2 19.12 27.62 0.31 BLG+10%K2CO3-char N2+21%H2+30%H2O 14.96 19.23 7.93 ND+10%K2CO3-char N2+30%H2 13.41 24.41 11.24 ND+10%K2CO3-char N2+21%H2+30%H2O 10.19 17.83 14.54 表 8 10%K2CO3-宁东煤热解半焦中的硫化物
Table 8 Sulfide in char during pyrolysis of 10%K2CO3-ND coal
Sample Atmospheres Ssulfide/St(%) K2S/St(%) FeS/St(%) ND+10%K2CO3-char N2 11.65 2.15 9.50 ND+10%K2CO3-char 70%N2+30%H2 37.12 28.37 8.75 -
[1] 2015年我国天然气消费增速创10年最低[EB/OL]. http://news.xinhuanet.com/energy/2016-02/12/c_1118022736.htm. [2] 毕继诚, 李克忠, 崔鑫, 祖静茹, 孙志强, 毛燕东, 康守国.一种联产合成气和煤焦油的方法:中国, CN102604683 A[P]. 2012-07-25.BI Ji-cheng, LI Ke-zhong, CUI Xin, ZU Jing-ru, SUN Zhi-qiang, MAO Yan-dogn, KANG Shou-guo. Method for co-production of synthetic gas and coal tar:CN, 102399595A[P]. 2012-04-04. [3] CHEN H K, LI B Q, YANG J L, ZHANG B J. Transformation of sulfur during pyrolysis and hydro-pyrolysis of coal[J]. Fuel, 1998, 77(6):487-493. doi: 10.1016/S0016-2361(97)00275-5 [4] HUANG F, ZHANG L Q, YI B J, XIA Z J, ZHENG C G. Effect of H2O on pyrite transformation behavior during oxy-fuel combustion[J]. Fuel Process Technol, 2015, 131:458-465. doi: 10.1016/j.fuproc.2014.12.027 [5] LEVY J H, WHITE T J. The reaction of pyrite with water vapor[J]. Fuel, 1988, 67:1336-1339. doi: 10.1016/0016-2361(88)90114-7 [6] SUGAWARA K, ABO K, SUGAWARA T, NISHYAMA Y, SHOLES M A. Dynamic behaviour of sulfur forms in rapid pyrolysis of coals with alkali treatment[J]. Fuel, 1995, 74:1823-1829. doi: 10.1016/0016-2361(95)80014-9 [7] LIU Q R, HU H Q, ZHU S W, ZHOU Q, LI W Y, WEI X Y, XIE K C. Desulfurization of coal by pyrolysis and hydro-pyrolysis with addition of KOH/NaOH[J]. Energy Fuels, 2005, 19(4):1673-1678. doi: 10.1021/ef0497053 [8] 袁申富.煤加氢气化的气化特性实验研究[D].太原:中国科学院山西煤炭化学研究所, 2015.YUAN Shen-fu. Experimental study on gasification characteristics of coal hydrogasification[D]. Taiyuan:Institute of Coal Chemistry, 2015. [9] MOULDER J F, STICKLE W F, EOBOL P E. Handbook of X-ray Photoelectron Spectroscopy[M]. Eden Prairie:Perkin Elmer Corporation, 1992. [10] CLEYLE P J, CALEY W F, STEWART I, WHITEWAY S G. Decomposition of pyrite and trapping of sulfur in a coal matrix during pyrolysis of coal[J]. Fuel, 1984, 63:1579-1582. doi: 10.1016/0016-2361(84)90230-8 [11] LBARRA J V, PALLCIOS J M, MOLINER R, BONET A. Evidence of reciprocal organic matter-pyrite interactions affecting sulfur removal during coal pyrolysis[J]. Fuel, 1994, 73(7):1046-1050. doi: 10.1016/0016-2361(94)90235-6 [12] ČERNIČ-SIMIĆ S. A study of factors that influence the behaviour of coal sulfur during carbonization[J]. Fuel, 1962, 41:141-151. [13] HUHN F, KLEIN J, JÜNTGEN H. Investigations on the alkali-catalyzed steam gasification of coal:Kinetics and interactions of alkali catalyst with carbon[J]. Fuel, 1983, 62(2):196-199. doi: 10.1016/0016-2361(83)90197-7 [14] MIMS C A, PABST J K. Role of surface salt complexes in alkali-catalyzed carbon gasification[J]. Fuel, 1983, 62:176-179. doi: 10.1016/0016-2361(83)90193-X [15] SNAPE E, MITCHELL S C, GARRCIA R, ISMALL K, BARTLE K D. Determination of organic sulfur forms in coals and coal derives by high pressure temperature-programmed reduction[J]. Fuel, 1993, 72(5):703-704. https://web.anl.gov/PCS/acsfuel/preprint%20archive/Files/40_3_CHICAGO_08-95_0461.pdf [16] GARCIA R, MOINELO S, LAFFERTY C, SNAPE C E. Pyrolytic desulfurization of some high-sulfur coals[J]. Energy Fuels, 1991, 5(4):582-586. doi: 10.1021/ef00028a009 [17] XU W C, KUMAGAI M. Sulfur transformation during rapid hydro-pyrolysis of coal under high pressure by using a continuous free fall pyrolyzer[J]. Fuel, 2003, 82:245-254. doi: 10.1016/S0016-2361(02)00290-9 [18] MINKOVA V, RAZVIGOROVA M, GORANOVA M, LJUTZKANOV L, ANGELOVA G. Effect of water vapour on the pyrolysis of solid fuels 1. Effect of water vapour during the pyrolysis of solid fuels on the yield and composition of the liquid products[J]. Fuel, 1991, 70(6):713-719. doi: 10.1016/0016-2361(91)90067-K [19] LANG R J. Anion effects in alkali-catalyzed steam gasification[J]. Fuel, 1986, 65(10):1324-1329. doi: 10.1016/0016-2361(86)90097-9 -





 下载:
下载: