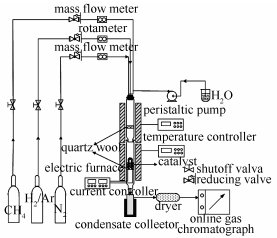
Research on electro-catalytic steam reforming of methane with modified Ni/γ-Al2O3 catalysts
-
摘要: 提出了电催化作用下甲烷水蒸气催化重整的新工艺。基于工业常规Ni基催化剂,采用等体积浸渍法,以Ni为活性组分,γ-Al2O3为载体,MgO、CaO为助剂,制备了Ni/γ-Al2O3、Ni-MgO/γ-Al2O3和Ni-CaO/γ-Al2O3催化剂,考察了电流强度、重整温度、水蒸气与甲烷的物质的量比(水碳比,S/C)对不同催化剂的CH4转化率、H2产率、CO选择性和催化剂稳定性的影响。结果表明,电催化工艺有着良好的普适性,电流的引入能够提升CH4转化率、增加H2产率,尤其在低温下电流的促进作用显著。在三种催化剂中,Ni-CaO/γ-Al2O3催化效果最佳,在电流为4.5 A、S/C为3、重整温度为700℃时,CH4转化率就高达95%以上。稳定性测试表明,电流的通入还能显著提高催化剂的稳定性,延缓催化剂的积炭失活。通过对催化剂的分析表征,发现电流的通入提升了催化剂中NiO的还原程度,同时抑制了反应过程中NiCx向石墨炭的转化,从而可延缓催化剂因积炭覆盖反应活性位点而造成的失活。Abstract: A novel electro-catalytic technique for catalytic methane steam reforming was developed. Based on the conventional industrial Ni-based catalyst, aseries of catalysts, including Ni/γ-Al2O3, Ni-MgO/γ-Al2O3 and Ni-CaO/γ-Al2O3, were prepared using incipient wetness impregnation method with Ni as the active component, γ-Al2O3 as the carrier, and MgO or CaO as the promoter. Experiments were performed to investigate the effects of electric current intensity, reforming temperature, and molar ratio of water vapor to methane (water/carbon ratio, S/C) on CH4 conversion, H2 yield, CO selectivity and catalyst stability. The results indicated that the electro-catalytic technique had good adaptability, and the introduction of electric current could improve the CH4 conversion and increase the H2 yield. Such effects were more intensive at lower reforming temperatures. Among the three catalysts, Ni-CaO/γ-Al2O3 catalyst exhibited the best catalytic efficiency, with the CH4 conversion over 95% under conditions of 4.5 A, S/C of 3, and 700℃. Stability tests of the catalysts showed that the electric current could improve the stability of catalysts and delay the deactivation caused by coke deposition. The characterization results proved that the presence of electric current enhanced the reduction degree of NiO in the catalyst and inhibited NiCx conversion to graphite carbon, resulting the delay of catalyst deactivation caused by carbon deposition over reactive sites.
-
表 1 稳定性测试过程中催化效率的下降
Table 1 The decline in catalytic efficiency during stability test
Catalyst t /℃ I /A Decline in efficiency/% 10Ni/γ-Al2O3 700 0 9.6 700 4.5 5.6 10Ni-3MgO/γ-Al2O3 700 0 7.1 700 4.5 3.3 10Ni-3CaO/γ-Al2O3 650 0 4.1 650 4.5 1.6 表 2 稳定性测试后催化剂的含碳量
Table 2 Carbon contents of the catalysts after stability tests
Catalyst I /A C w/% Decrease /% 10Ni/γ-Al2O3 0 0.89 20.2 4.5 0.71 10Ni-3MgO/γ-Al2O3 0 0.39 15.4 4.5 0.33 10Ni-3CaO/γ-Al2O3 0 0.52 28.8 4.5 0.37 表 3 催化剂的微观结构性质
Table 3 Microstructure properties of the catalysts
Catalyst Condition BET surface
A/(m2·g-1)Pore volume
v/(cm3·g-1)Average pore
diameter d/nmNi0
particle size *d/nm10Ni/γ-Al2O3 fresh catalyst 118.9 0.64 21.6 (NiO) 0 A reformed 104.6 0.57 21.9 18.6 4.5A reformed 106.1 0.56 21.1 13.5 10Ni-3MgO/γ-Al2O3 fresh catalyst 114.3 0.60 21.0 (NiO) 0 A reformed 105.2 0.56 21.3 11.5 4.5 A reformed 107.8 0.53 19.9 8.9 10Ni-3CaO/ γ-Al2O3 fresh catalyst 107.8 0.60 22.1 (NiO) 0 A reformed 98.2 0.52 21.4 12.4 4.5 A reformed 103.0 0.53 20.7 11.0 *: the average particle size of Ni is calculated using Scherrer formula based on the XRD diffraction peaks 表 4 Ni0所占百分比(Ni0/(Ni0+Ni2+))
Table 4 Percentage of Ni0 (Ni0/(Ni0+Ni2+))
Catalyst I (0 A) /% I (4.5 A) /% 10Ni/γ-Al2O3 23.8 24.0 10Ni-3MgO/γ-Al2O3 17.3 35.8 10Ni-3CaO/γ-Al2O3 25.1 29.0 表 5 NiCx和石墨炭所占百分比
Table 5 Percentages of NiCx and graphite carbon
Catalyst NiCx /% Graphite carbon /% I (0A) I (4.5A) I (0A) I (4.5A) 10Ni/
γ-Al2O314.3 20.8 69.1 62.2 10Ni-3MgO/
γ-Al2O358.4 65.7 27.6 22.4 10Ni-3CaO/
γ-Al2O347.9 54.0 40.0 35.2 -
[1] LULIANELLI A, LIGUORI S, WILCOX J, BASILE A. Advances on methane steam reforming to produce hydrogen through membrane reactors technology:A review[J]. Catal Rev, 2016, 13:1-35. doi: 10.1080/01614940.2015.1099882 [2] 王东旭, 肖显斌, 李文艳.乙酸蒸汽催化重整制氢的研究进展[J].化工进展, 2017, 36(5):1658-1662. http://www.cnki.com.cn/Article/CJFDTotal-HGJZ201705016.htmWANG Dong-xu, XIAO Xian-bin, LI Wen-yan. A review of literatures on catalytic steam reforming of acetic acid for hydrogen production[J]. Chem Ind Eng Prog, 2017, 36(5):1658-1662. http://www.cnki.com.cn/Article/CJFDTotal-HGJZ201705016.htm [3] 孙杰, 孙春文, 李吉刚, 周添, 董中朝, 陈立泉.甲烷水蒸气重整反应研究进展[J].中国工程科学, 2013, 15(2):98-106. http://or.nsfc.gov.cn/bitstream/00001903-5/292015/1/1000006899498.pdfSUN Jie, SUN Chun-wen, LI Ji-gang, ZHOU Tian, DONG Zhong-chao, CHEN Li-quan. Research on the steam reforming of methane[J]. Chin Eng Sci, 2013, 15(2):98-106. http://or.nsfc.gov.cn/bitstream/00001903-5/292015/1/1000006899498.pdf [4] 陈曦. 镍基催化剂制备及在甲烷水蒸气重整反应中的应用[D]. 辽宁: 大连理工大学, 2014.CHEN Xi. Preparation of Ni-based catalysts and application in steam reforming of methane[D]. Liaoning: Dalian University of Technology, 2014. [5] ARKATOVA L A. The deposition of coke during carbon dioxide reforming of methane over intermetallides[J]. Catal Today, 2010, 157:170-176. doi: 10.1016/j.cattod.2010.03.003 [6] IGLESIAS I, BARONETTI G, MARINO F. Nickel-based doped ceria-supported catalysts for steam reforming of methane at mild conditions[J]. Energy Sources, 2017, 39(2):129-133. doi: 10.1080/15567036.2016.1214639 [7] LIAN J, FANG X Z, LIU W M, HUANG Q, SUN Q K, WANG H M, WANG X, ZHOU W F. Ni Supported on LaFeO3 perovskites for methane steam reforming:On the promotional effects of plasma treatment in H2-Ar atmosphere[J]. Top Catal, 2017, DOI: 10.1007/s11244-017-0748-6. [8] 赵云莉, 吕永康, 常丽萍, 鲍卫仁.助剂MgO、CaO对甲烷水蒸气重整Ni/γ-Al2O3催化性能的影响[J].燃料化学学报, 2010, 38(2):218-222. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract17566.shtmlZHAO Yun-li, LÜ Yong-kang, CHANG Li-ping, BAO Wei-ren. Effects of MgO and CaO on properties of Ni/γ-Al2O3 catalyst for the reforming of methane and steam[J]. J Fuel Chem Technol, 2010, 38(2):218-222. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract17566.shtml [9] ALI S, AL-MARRI M, G. ABDELMONEIM A, KUMAR A, M. KHADER M. Catalytic evaluation of nickel nanoparticles in methane steam reforming[J]. Int J Hydrogen Energy, 2016, 41:22876-22885. doi: 10.1016/j.ijhydene.2016.08.200 [10] CHEN Y Q, YUAN L X, YE T Q, QIU S B, ZHU X F, TORIMOTO Y, YAMAMOTO M, LI Q X. Effects of current upon hydrogen production from electrochemical catalytic reforming of acetic acid[J]. Int J Hydrogen Energy, 2009, 34(4):1760-1770. doi: 10.1016/j.ijhydene.2008.12.044 [11] YE T Q, YUAN L X, CHEN Y Q, KAN T, TU J, ZHU X F, TORIMOTO Y, YAMAMOTO M, LI Q X. High efficient production of hydrogen from bio-oil using low-temperature electrochemical catalytic reforming approach over NiCuZn-Al2O3 catalyst[J]. Catal Lett, 2009, 127(3/4):323-333. http://cat.inist.fr/?aModele=afficheN&cpsidt=17144011 [12] YUAN L X, CHEN Y Q, SONG C F, YE T Q, GUO Q X, ZHU X F, TORIMOTO Y, LI Q X. Electrochemical catalytic reforming of oxygenated-organic compounds:A highly efficient method for production of hydrogen from bio-oil[J]. Chem Commun, 2008, 41:5215-5217. https://www.ncbi.nlm.nih.gov/pubmed/18956073 [13] KAN T, XIONG J X, LI X L, YE T Q, YUAN L X, TORIMOTO Y, YAMAMOTO M, LI Q X. High efficient production of hydrogen from crude bio-oil via an integrative process between gasification and current-enhanced catalytic steam reforming[J]. Int J Hydrogen Energy, 2010, 35(2):518-532. doi: 10.1016/j.ijhydene.2009.11.010 [14] 陶君. 镍基催化剂催化转化生物质焦油典型组分的研究[D]. 北京: 华北电力大学, 2015.TAO Jun. Research on the catalytic conversion of biomass tar model compounds using Ni-based catalysts[D]. Beijing: North China Electric Power University, 2015. [15] YUAN L X, YE T Q, GUO Q X, TORIMOTO Y, YAMAMOTO M, LI Q X. Hydrogen production from the current-enhanced reforming and decomposition of ethanol[J]. Energy Fuels, 2009, 23(6):3103-3112. doi: 10.1021/ef801131a [16] HU X, LU G X. Investigation of steam reforming of acetic acid to hydrogen over Ni-Co metal catalyst[J]. J Mol Catal A:Chen, 2007, 261(1):43-48. doi: 10.1016/j.molcata.2006.07.066 [17] HU X, LU G X. Syngas production by CO2 reforming of ethanol over Ni/Al2O3 catalyst[J]. Catal Commun, 2009, 10(13):1633-1637. doi: 10.1016/j.catcom.2009.04.030 [18] 姜洪涛, 华炜, 计建炳.甲烷重整制合成气镍催化剂积炭研究[J].化学进展, 2013, 25(5):859-868. http://manu56.magtech.com.cn/progchem/CN/abstract/abstract11074.shtmlJIANG Hong-tao, HUA Wei, JI Jian-bing. Study of coke deposition on Ni catalysts for methane reforming to syngas[J]. Prog Chem, 2013, 25(5):859-868. http://manu56.magtech.com.cn/progchem/CN/abstract/abstract11074.shtml [19] 方修忠. 高效抗积炭Ni基甲烷重整制氢催化剂的制备和性能研究[D]. 江西: 南昌大学, 2016.FANG Xiu-zhong. The preparation of highly active and coke resistant Ni-based catalysts for methane reforming for hydrogen production[D]. Jiangxi: Nanchang University, 2016. [20] PAKHARE D, SPIVEY J. A review of dry (CO2) reforming of methane over noble metal catalysts[J]. Chem Soc Rev, 2014, 21:7813-7837. http://www.ncbi.nlm.nih.gov/pubmed/24504089 [21] MA Z, JIANG Q Z, WANG X, ZHANG W G, MA Z F. CO2 reforming of dimethyl ether over Ni/γ-Al2O3 catalyst[J]. Catal Commun, 2012, 17:49-53. doi: 10.1016/j.catcom.2011.10.014 [22] LUISETTO I, SARNO C, FELICIS D D, BASOLI F, BATTOCCHIO C, TUTI S, LICOCCIA S, BARTOLOMEO E D. Ni supported on γ-Al2O3 promoted by Ru for the dry reforming of methane in packed and monolithic reactors[J]. Fuel Process Technol, 2017, 158:130-140. doi: 10.1016/j.fuproc.2016.12.015 [23] 赵云莉. 甲烷重整制氢镍基催化剂制备及活性评价研究[D]. 太原: 太原理工大学, 2009.ZHAO Yun-li. Study of methane catalytic reforming to hydrogen on nickel-based catalysts[D]. Taiyuan: Taiyuan University of Technology, 2009. [24] 袁丽霞. 电催化水蒸气重整生物油及乙醇制氢的基础应用研究[D]. 安徽: 中国科学技术大学, 2008.YUAN Li-xia. Basic application research on the hydrogen production from bio-oil and ethanol by electrochemical catalytic steam reforming[D]. Anhui: University of Science and Technology of China, 2008. [25] 李春义, 余长春, 沈师孔. Ni/Al2O3催化剂上CH4部分氧化制合成气反应积炭的原因[J].催化学报, 2001, 22:377-382. doi: 10.3321/j.issn:0253-9837.2001.04.016LI Chun-yi, YU Chang-chun, SHEN Shi-kong. Ni/Al2O3 catalyst for partial oxidation of CH4 to syngas[J]. Chin J Catal, 2001, 22:377-382 doi: 10.3321/j.issn:0253-9837.2001.04.016 [26] WU H J, PANTALEO G, PAROLA V L, VENEZIA A M, COLLARD X, APRILE C, LIOTTA L. Bi-and trimetallic Ni catalysts over Al2O3 and Al2O3-MOx (M=Ce or Mg) oxides for methane dry reforming:Au and Pt additive effects[J]. Appl Catal B:Environ, 2014, 156-157:350-361. doi: 10.1016/j.apcatb.2014.03.018 [27] HOFFER B W, LANGEVELD A D, JANESSENS J P, BONNÈ R L C, LOK C M, MOOLIJIN J A. Stability of highly dispersed Ni/Al2O3 catalysts:Effects of pretreatment[J]. J Catal, 2000, 192:432-440. doi: 10.1006/jcat.2000.2867 [28] MIRYAM G C, CRISTINA J G, BEATRIZ D R, JOSE I G, Rubén L F. Effect of Ni/Al molar ratio on the performance of substoichiometric NiAl2O4 spinel-based catalysts for partial oxidation of methane[J]. Appl Catal B:Environ, 2017, 209:128-138. doi: 10.1016/j.apcatb.2017.02.063 [29] NESBITT H W, D. LEGRANG, BANCOF G M. Interpretation of Ni 2p XPS spectra of Ni conductors and Ni insulators[J]. Phys Chem Minerals, 2000, 27:357-366. doi: 10.1007/s002690050265 [30] 李春义, 余长春, 沈师孔. Ni/Al2O3催化剂上CH4部分氧化制合成气反应积炭的原因[J].催化学报, 2001, 22:377-382. doi: 10.3321/j.issn:0253-9837.2001.04.016LI Chun-yi, YU Chang-chun, SHEN Shi-kong. Ni/Al2O3 catalyst for partial oxidation of CH4 to syngas[J]. Chin J Catal, 2001, 22:377-382 doi: 10.3321/j.issn:0253-9837.2001.04.016 [30] GUCZI L, STEFLER G, GESZTI O, SAJO I, PASZTI Z, TOMPOS A, SCHAY Z. Methane dry reforming with CO2:A study on surface carbon species[J]. Appl Catal A:Gen, 2010, 375:236-246. doi: 10.1016/j.apcata.2009.12.040 -





 下载:
下载: