Catalytic performance of Ag(x)/ZSM-5 catalysts for selective catalytic reduction of nitric oxide by methane
-
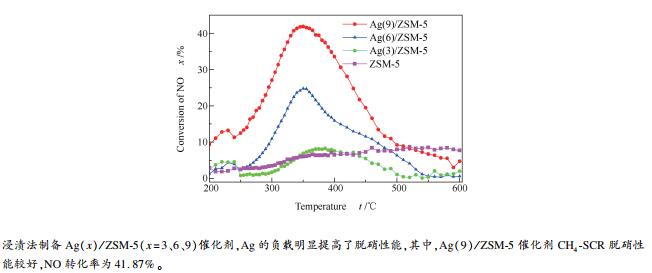
摘要: 采用浸渍法制备Ag(x)/ZSM-5(x=3、6、9)催化剂。采用XRD、SEM、NH3-TPD、Py-FTIR、XPS和NO-TPD等手段对催化剂的理化性质进行表征,在常压固定床微型反应器中评价催化剂甲烷选择性催化还原(CH4-SCR)NO催化性能,考察Ag负载量对Ag(x)/ZSM-5催化剂CH4-SCR脱硝性能的影响。结果表明,ZSM-5分子筛负载Ag,催化剂的酸性和酸量发生变化,改善了催化剂对NO的吸附脱附性能。随着Ag负载量增加,形成较大的Ag晶粒,有利于甲烷活化,Ag(x)/ZSM-5催化剂CH4-SCR脱硝活性提高。Ag(9)/ZSM-5催化剂CH4-SCR脱硝性能较好,在350℃时NO转化率为41.87%。Abstract: Ag(x)/ZSM-5 (x=3, 6, 9) catalysts for selective catalytic reduction of nitric oxide by methane (CH4-SCR) were prepared by impregnation method. The physicochemical properties of the catalysts were characterized by XRD, SEM, NH3-TPD, Py-FTIR, XPS and NO-TPD. The catalytic performance for selective catalytic reduction of nitric oxide by methane was evaluated in a fixed-bed micro-reactor under atmospheric pressure and the influence of Ag loading were investigated.The results show that the impregnation of Ag has changed the acidity and amount of acid of the catalyst and improved the adsorption and desorption performance of NO on ZSM-5 molecular sieve. With the increase of Ag loading, Ag particle size increases and results in higher methane activation as well as the denitration activity of CH4-SCR over Ag(x)/ZSM-5 catalyst. Ag(9)/ZSM-5 has better denitration activity and the NO conversion is 41.87% at 350℃.
-
Key words:
- methane /
- NO /
- Ag /
- ZSM-5 /
- selective reduction
-
表 1 Ag(x)/ZSM-5(x=0、3、6、9)催化剂中元素含量
Table 1 Elemental contents of the Ag(x)/ZSM-5 Ag(x)/ ZSM-5 (x=0, 3, 6, 9) catalysts
Catalyst Elemental composition w/% O Na Al Si Ag* ZSM-5 63.42 0.59 0.94 35.05 - Ag(3)/ZSM-5 55.34 0.24 1.01 41.03 2.44 Ag(6)/ZSM-5 57.44 0.11 1.44 36.17 5.08 Ag(9)/ZSM-5 59.97 0.07 0.69 32.47 7.30 *: base on carrier 表 2 Ag(x)/ZSM-5(x=0、3、6、9)催化剂样品的NH3-TPD测试
Table 2 NH3-TPD results of the Ag(x)/ZSM-5 (x=0, 3, 6, 9) catalysts
Catalyst Weak acid Medium acid Strong acid peak t/℃ percentage/% peak t/℃ percentage/% peak t/℃ percentage/% ZSM-5 191 57.6 280 22.1 395 20.3 Ag(3)/ZSM-5 228 53.8 303 29.0 465 17.3 Ag(6)/ZSM-5 228 51.3 300 33.7 465 15.0 Ag(9)/ZSM-5 225 50.2 295 38.3 450 11.4 表 3 Ag(x)/ZSM-5 Ag(x)/ZSM-5(x=3、6、9)催化剂的XPS表征
Table 3 XPS results of the Ag(x)/ZSM-5 (x=3, 6, 9) catalysts
Catalyst Binding energy of Ag 3d5/2 E/eV SAg0/% Binding energy of Ag 3d3/2 E/eV SAg0/% Ag+ Ag0 Ag+ Ag0 Ag(3)/ZSM-5 367.2 368.8 69 373.3 374.8 71 Ag(6)/ZSM-5 367.6 368.8 73 373.5 374.8 76 Ag(9)/ZSM-5 367.9 368.8 81 373.9 374.8 81 表 4 Ag(x)/ZSM-5(x=3、6、9)催化剂的NO-TPD测试
Table 4 NO-TPD results of the Ag(x)/ZSM-5 (x=3, 6, 9) catalysts
Catalyst α β γ η peak t/℃ percentage/% peak t/℃ percentage/% peak t/℃ percentage/% peak t/℃ percentage/% Ag(3)/ZSM-5 96 8.4 185 17.6 262 44.4 315 29.6 Ag(6)/ZSM-5 113 9.9 212 21.7 283 42.9 327 25.5 Ag(9)/ZSM-5 105 19.0 200 25.3 269 35.0 319 20.7 -
[1] LI D X, XIAO Z G, TALLAL B A, XU S H. Flue gas denitration by wet oxidation absorption methods:Current status and development[J]. Environ Eng Sci, 2018, 20(4):1-14. http://cn.bing.com/academic/profile?id=f2b838ec89dcbce2ac83252334917573&encoded=0&v=paper_preview&mkt=zh-cn [2] ZHOU T Y, YUAN Q, PAN X L, BAO X H. Growth of Cu/SSZ-13 on SiC for selective catalytic reduction of NO with NH3[J]. Chin J Catal, 2018, 39(1):71-78. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb201801010 [3] 王继封, 王慧敏, 张亚青, 张秋林, 宁平. WO3的引入对MnOx-Fe2O3催化剂上NH3-SCR反应中N2选择性的促进作用[J].燃料化学学报, 2019, 47(7):814-822. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201907006WANG Ji-feng, WANG Hui-min, ZAHNG Ya-qing, ZHANG Qiu-lin, NING Ping. Promotion effect of tungsten addition on N2 selectivity of MnOx-Fe2O3 for NH3-SCR[J]. J Fuel Chem Technol, 2019, 47(7):814-822. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201907006 [4] FREDRIK G J A, PIHL T J, TOOPS M S H H. Lean NOx reduction over Ag/alumina catalysts via ethanol-SCR using ethanol/gasoline blends[J]. Appl Catal B:Environ, 2017, 202(9):42-50. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=84a6696479f3728f83a6f7c45c35edd2 [5] GELVES J F, DORKIS L, MÁRQUEZ M A, A'LVAREZ A C, GONZA'LEZ L M, VILLA A L. Activity of an iron colombian natural zeolite as potential geo-catalyst for NH3-SCR of NOx[J]. Catal Today, 2018, 25(1):1-40. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=656a748a34bcaa9ea4645e4df8578989 [6] NTZIACHRISTOS L, PAPADIMITRIOU G, LIGTERINK N, HAUSBERGER S. Implications of diesel emissions control failures to emission factors and road transport NOx evolution[J]. Atmos Environ, 2016, 141(7):542-551. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=dfd90d8c866489deead89ba62e9e0db4 [7] EMIROĞLU A O. Investigation of NOx reduction activity of Rh/ZnO nanowires catalyst[J]. Atmos Pollut Res, 2016, 8(1):1-5. https://www.researchgate.net/publication/307523116_Investigation_of_NOx_reduction_activity_of_RhZnO_nanowires_catalyst [8] XIANG X, CAO Y, SUN L J, WU P F, CAO L, XU S T, TIAN P, LIU Z M. Improving the low-temperature hydrothermal stability of Cu-SAPO-34 by the addition of Ag for ammonia selective catalytic reduction of NOx[J]. Appl Catal A:Gen, 2018, 551:79-87. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=77168714ef8bfa57cf91d4970fa76a01 [9] KOEBEL M, ELSENER M, KLEEMANN M. Urea-SCR:A promising technique to reduce NOx emissions from automotive diesel engines[J]. Catal Today, 2000, 59(3/4):335-345. http://d.old.wanfangdata.com.cn/Periodical/nrjgc201102001 [10] ZOU C L, WU X, MENG H, DU Y, LI Z. The SO2 resistance improvement of Mn-Fe/ZSM-5 for NH3-SCR at low temperature by optimizing synthetic method[J]. ChemistrySelect, 2018, 3(46):13042-13047. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1002/slct.201801518 [11] 李元元, 黄妍, 唐南, 闫润华, 胡振宇, 肖娆, 付晴, 赵令葵, 张俊丰, 杨柳春.不同晶体结构MnO2纳米催化剂低温NH3-SCR性能研究[J].燃料化学学报, 2018, 46(5):578-584. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201805010LI Yuan-yuan, HUANG Yan, TANG Nan, YAN Run-hua, HU Zhen-yu, XIAO Rao, FU Qing, ZHAO Ling-kui, ZHANG Jun-feng, YANG Liu-chun. Promotion effect of tungsten addition on N2 selectivity of MnOx-Fe2O3 for NH3-SCR[J]. J Fuel Chem Technol, 2018, 46(5):578-584. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201805010 [12] CAO J, YAO X J, YANG F, CHEN L, FU M, TANG C J, DONG L. Improving the denitration performance and K-poisoning resistance of the V2O5-WO3/TiO2 catalyst by Ce4+ and Zr4+ co-doping[J]. Chin J Catal, 2019, 40(1):95-104. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb201901011 [13] LIU Z M, ZHANG S X, LI J H, ZHU J Z, MA L L. Novel V2O5-CeO2/TiO2 catalyst with low vanadium loading for the selective catalytic reduction of NOx by NH3[J]. Appl Catal B:Environ, 2014, 158-159:11-19. https://www.researchgate.net/publication/261717210_Novel_V2O5-CeO2TiO2_catalyst_with_low_vanadium_loading_for_the_selective_catalytic_reduction_of_NOx_by_NH3 [14] SHI X, HE H, XIE L. The effect of Fe species distribution and acidity of Fe-ZSM-5 on the hydrothermal stability and SO2 and hydrocarbons durability in NH3-SCR reaction[J]. Chin J Catal, 2015, 36(4):649-656. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb201504026 [15] SEBASTIAN P M A, DEREWINSKI Y W N M, WASHTON E D. Sub-micron Cu/SSZ-13:Synthesis and application as selective catalytic reduction (SCR) catalysts[J]. Appl Catal B:Environ, 2017, 201:461-469. http://cn.bing.com/academic/profile?id=d9c322f623909a38b4664581ff8f1233&encoded=0&v=paper_preview&mkt=zh-cn [16] SHUA Y J, HEA M, JIA J. Synergetic degradation of VOCs by vacuum ultraviolet photolysis and catalyticozonation over Mn-xCe/ZSM-5[J]. J Hazard Mater, 2019, 364:770-779. https://www.zhangqiaokeyan.com/academic-journal-foreign_other_thesis/0204112795708.html [17] YUNG M M, STARACE A K, GRIFFIN M B, WELLSB J D, PATALANOC R E, SMITHA K R, SCHAIDLE J A. Restoring ZSM-5 performance for catalytic fast pyrolysis of biomass:Effect of regeneration temperature[J]. Catal Today, 2018, 141:6-10. http://cn.bing.com/academic/profile?id=12b7ae93c5f2b2c36bdcefe8a54a45dc&encoded=0&v=paper_preview&mkt=zh-cn [18] 邓湘玲, 叶松寿, 曹志凯, 张诺伟, 郑进保, 陈秉辉. Ag/Ce0.75Zr0.25O2催化剂中Ag的负载量对碳烟燃烧活性的影响[J].化工学报, 2017, 68(8):3064-3070. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgxb201708014DENG Xiang-ling, YE Song-shou, CAO Zhi-kai, ZHANG Nuo-wei, ZHENG Jin-bao, CHEN Bing-hui. Effect of Ag loading on soot oxidation for Ag/Ce0.75Zr0.25O2 catalysts[J]. CIESC J, 2017, 68(8):3064-3070. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgxb201708014 [19] ROOZBEH J K, ANAHITA K. Fabrication of bimetallic Ag-Co nanoparticle deposited on hierarchical ZSM-5 as a selective catalyst for synthesis of propargylamine in water via multicomponent A3 coupling[J]. ChemistrySelect, 2018, 3(11):12666-12675. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1002/slct.201803011 [20] LEE K J, RAO K N, YU C Y, HONG S I, HA H P. Synthesis and characterisation of K-Ag/Al2O3 catalysts for CH4-SCR of NOx:Effect of SO2[J]. Chem Inter, 2013, 39(3):1463-1479. [21] WANG S, LUO S, LI X H, LIU W, WU X D, WENG D, LIU S. Thermally stable Ag/Al2O3 confined catalysts with high diffusion-induced oxidation activity[J]. Catal Today, 2018, 27(6):1-24. https://www.researchgate.net/publication/325954832_Thermally_stable_AgAl_2_O_3_confined_catalysts_with_high_diffusion-induced_oxidation_activity [22] 潘红艳, 刘秀娟, 易芸, 刘飞, 林倩. Ag改性ZSM-5分子筛催化甲醇制烯烃的研究[J].天然气化工, 2015, 40(6):7-12. http://d.old.wanfangdata.com.cn/Periodical/trqhg201506002PAN Hong-li, LIU Xiu-juan, YI Yu, LIU Fei, LIN Qian. Study on Ag-modified ZSM-5 zeolite catalyzed by methanol to olefins[J]. Nat Gas Chem Ind, 2015, 40(6):7-12. http://d.old.wanfangdata.com.cn/Periodical/trqhg201506002 [23] 赵素雷, 任瑞鹏, 曹青.几种金属原子在ZSM-5分子筛上生长机理的密度泛函理论研究[J].山西化工, 2015, 157(3):1-18. http://d.old.wanfangdata.com.cn/Periodical/shanxhg201503001ZHAO Su-lei, REN Rui-peng, CAO Qing. The growth of the metal atoms on the ZSM-5 zeolite:A density functional theory study[J]. Shanxi Chem Ind, 2015, 157(3):1-18. http://d.old.wanfangdata.com.cn/Periodical/shanxhg201503001 [24] CHEN X, ZHU A, AU C T, SHI C. Enhanced low-temperature activity of Ag-promoted Co-ZSM-5 for the CH4-SCR of NO[J]. Catal Lett, 2011, 141(1):207-212. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=bcdb3503415fd71e8055fc17453f83e1 [25] RAMÍREZ G R E, IZNAGAI R, SIMAKOV A, SIMAKOV M H, FAR'IAS F F, BARRAZA C. Cu-Ag/mordenite catalysts for NO reduction:Effect of silver on catalytic activity and hydrothermal stability[J]. Mater Res Bull, 2018, 97:369-378. http://cn.bing.com/academic/profile?id=6cae3cf0209bd396df395736ab8c91c8&encoded=0&v=paper_preview&mkt=zh-cn [26] MARTÍNEZ-ARIAS A, FERNÁNDEZ-GARCIÍA M, IGLESIAS-JUEZ A, ANDERSON J A, CONESA J C, SORIA J. Study of the lean NOx reduction with C3H6 in the presence of water over silver/alumina catalysts prepared from inverse microemulsions[J]. Appl Catal B:Environ, 2000, 28(1):29-41. https://www.researchgate.net/publication/222227145_Study_of_the_lean_NOx_reduction_with_C3H6_in_the_presence_of_water_over_silveralumina_catalysts_prepared_from_inverse_microemulsions [27] HAN S, CHENG J, ZHENG C K, YE Q, CHENG S Y, KANG T F, DAI H X. Effect of Si/Al ratio on catalytic performance of hydrothermally aged Cu-SSZ-13 for the NH3-SCR of NO in simulated diesel exhaust[J]. Appl Sur Sci, 2017, 419:382-392. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=b9e324b0057a802f1b98067d7a4ab264 [28] 窦喆, 张海杰, 潘燕飞, 徐秀峰. N2O在钾改性Cu-Co尖晶石型复合氧化物上的催化分解[J].燃料化学学报, 2014, 42(2):238-245. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201402017DOU Zhe, ZHANG Hai-jie, PAN Yan-fei, XU Xiu-feng. Catalytic decomposition of NO over potassium-modified Cu-Co spinel oxides[J]. J Fuel Chem Technol, 2014, 42(2):238-245. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201402017 [29] LI J, ZHU P F, ZHOU R X. Effect of the preparation method on the performance of CuO-MnOx-CeO2 catalysts for selective oxidation of CO in H2-rich streams[J]. J Power Sources, 2011, 196(22):9590-9598. doi: 10.1021/cs200418w [30] 韩蕾, 欧阳颖, 罗一斌, 达志坚.不同元素改性ZSM-5分子筛在轻烃催化裂解中的应用[J].石油学报, 2018, 34(2):419-429. http://d.old.wanfangdata.com.cn/Periodical/syxb-syjg201802025HAN Lei, OU Yang-ying, LUO Yi-bin, DA Zhi-jian. Application of modified ZSM-5 zeolite with different elements in catalytic cracking oflight hydrocarbon[J]. Acta Pet Sin, 2018, 34(2):419-429. http://d.old.wanfangdata.com.cn/Periodical/syxb-syjg201802025 [31] KOLOBOVA E, PESTRYAKOV A, MAMONTOV G, KOTOLEVICH Y, BOGDANCHIKOVA N, FARIAS M, VOSMERIKOV A, VOSMERIKOVA L, CORBERAN V C. Low-temperature CO oxidation on Ag/ZSM-5 catalysts:Influence of Si/Al ratio and redox pretreatments on formation of silver active sites[J]. Fuel, 2017, 188:121-131. http://cn.bing.com/academic/profile?id=8fee8e82e83e9569aa3b304df61f3a84&encoded=0&v=paper_preview&mkt=zh-cn [32] BABA T, SAWADA H. Conversion of methane into higher hydrocarbons in the presence of ethylene over H-ZSM-5 loaded with silver cations[J]. Phys Chem Chem Phys, 2002, 4(15):3919-3923. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a2a1d5849a6b7d74819247a0cc11b202 [33] 张相俊, 刘晓刚, 李清雍, 李岩, 魏波, 王虹, 李翠清, 宋永吉.载体对铜基催化剂NH3-SCR低温脱硝性能的影响[J].燃料化学学报, 2017, 45(2):220-226. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201702012ZHANG Xiang-jun, LIU Xiao-gang, LI Qing-yong, LI Yan, WEI Bo, WANG Hong, LI Cui-qing, SONG Yong-ji. Effect of carrier on the performance of copper based catalyst for selective catalytic reduction of NO with NH3 at low temperature[J]. J Fuel Chem Technol, 2017, 45(2):220-226. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201702012 -





 下载:
下载: