Effect of temperature and water vapor on the form and evolution characteristics of nitrogen in coal char
-
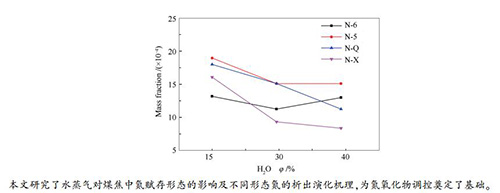
摘要: 研究水蒸气对不同赋存形态氮在热解过程中析出及转化规律有利于精细化调控氮氧化物的生成。利用X射线光电子能谱(XPS)仪分析了典型烟煤及其煤焦中氮的赋存形态,研究了温度、水蒸气浓度等因素对煤焦炭氮析出及转化特性的影响。结果表明,温度的升高以及一定范围内水蒸气浓度的增加有利于焦炭氮的析出,水蒸气浓度达到30%时,焦炭氮析出达到峰值;水蒸气的存在有利于煤焦中N-5和N-6等含氮结构物质从煤焦中析出,同时抑制N-Q和N-X等含氮结构物质的裂解。这是由于水蒸气的气化作用有利于芳香结构的分解,但同时高水蒸气浓度带来的H基团会与N-6结合从而形成N-Q,而由高水蒸气浓度带来的OH基团会促进N-6的氧化而生成N-X。Abstract: Studying the evolution and transformation of different forms of nitrogen in pyrolysis under water vapor atmosphere is conducive to control formation of nitrogen oxides. X-ray photoelectron spectroscopy (XPS) was used to examine forms of nitrogen in typical bituminous coal and its char, and effects of temperature, water vapor on evolution and conversion characteristics of coal nitrogen were investigated. The results indicate that raising temperature and concentration of water vapor are conducive to evolution of char nitrogen. When concentration of water vapor reaches 30%, the char nitrogen evolution reaches a peak. The presence of water vapor facilitates the evolution of N-5 and N-6 from coal char and suppresses that of N-Q and N-X, because the gasification effect of water vapor is beneficial to breaking aromatic structure, but the conversion of N-6 to N-Q and N-X is promoted as a result of H and OH groups brought by the high concentration of water vapor.
-
Key words:
- char /
- nitrogen content /
- vapor /
- nitrogen forms /
- XPS
-
表 1 原煤的元素分析与工业分析
Table 1 Proximate and ultimate analyses of raw coal
Proximate analysis wad/% Ultimate analysis wad/% M A V FC C H O* N S 1.75 41.09 13.37 44.21 40.94 2.56 11.94 0.72 1.00 *: calculated by difference 表 2 实验工况表
Table 2 Experimental condition
No. Temperature t/℃ Ar φ/% H2O φ/% 1 800 100 0 2 1000 100 0 3 800 85 15 5 1000 70 30 6 1000 60 40 表 3 各形态氮N 1s结合能
Table 3 N 1s binding energy of various forms of nitrogen
Nitrogen forms Symbol Binding energy E/eV Pyridine N-6 398.7 ±0.4 Pyrrole N-5 400.5 ±0.3 Protonated pyridine,quaternary nitrogen N-Q 401.4 ±0.1 Nitrogen oxide N-X 403.5 ±0.5 表 4 样品焦产率及各形态氮的相对含量
Table 4 Sample char yield and relative content of various forms of nitrogen
No. Char yield w/% Total nitrogen content w/% Relative content w/% N-6 N-5 N-Q N-X Raw coal - 1.42 14.96 31.46 27.57 26.01 1 90.3 1.00 26.70 56.11 6.79 10.40 2 84.1 0.88 23.45 70.85 0.01 5.70 3 87.4 0.98 31.94 36.91 15.34 15.81 4 82.3 0.66 19.69 28.52 27.62 24.18 5 81.0 0.54 25.85 28.25 30.05 15.85 6 80.9 0.54 30.96 28.45 25.46 15.13 -
[1] 车得福.煤氮热变迁与氮氧化物生成[M].西安:西安交通大学出版社, 2013.CHE De-fu. Thermal Coal-N Transformation and Nitrogen Oxide Generation[M]. Xi'an:Xi'an Jiaotong University Press, 2013. [2] KAMBARA S, TAKARADA T, TOYOSHIMA M, KATO K. Relation between functional forms of coal nitrogen and NOx emissions from pulverized coal combustion[J]. Fuel, 1995, 74(9):1247-1253. doi: 10.1016/0016-2361(95)00090-R [3] KAMBARA S, TAKARADA T, YAMAMOTO Y, KATO K. Relation between functional forms of coal nitrogen and formation of nitrogen oxide (NOx) precursors during rapid pyrolysis[J]. Energy Fuels, 1993, 7(6):1013-1020. doi: 10.1021/ef00042a045 [4] NELSON P F, BUCKLEY A N, KELLY M D. Functional forms of nitrogen in coals and the release of coal nitrogen as NOx precursors (HCN and NH3)[J]. Symp Combust, 1992, 24(1):1259-1267. doi: 10.1016-S0082-0784(06)80148-7/ [5] NELSON P F, KELLY M D, WORNAT M J. Conversion of fuel nitrogen in coal volatiles to NOx precursors under rapid heating conditions[J]. Fuel, 1991, 70(3):403-407. doi: 10.1016-0016-2361(91)90130-3/ [6] LI C Z, NELSON P F, LEDESMA E B, MACKIE J C. An experimental study of the release of nitrogen from coals pyrolyzed in fluidized-bed reactors[J].Symp Combust, 1996, 26(2):3205-3211. http://www.sciencedirect.com/science/article/pii/S0082078496801664 [7] 姚明宇, 刘艳华, 车得福.氧对宜宾煤中燃料氮迁移特性的影响[J].燃烧科学与技术, 2004, 10(4):336-340. doi: 10.3321/j.issn:1006-8740.2004.04.010YAO Ming-yu, LIU Yan-hua, CHE De-fu. Investigation on effects of oxygen on the transformation of Yibin coal nitrogen functionality[J]. Combust Sci Technol, 2004, 10(4):336-340. doi: 10.3321/j.issn:1006-8740.2004.04.010 [8] ZHANG Y, ZHANG J, SHENG C, CHEN J, LIU Y, ZHAO L, XIE F. X-ray photoelectron spectroscopy (XPS) investigation of nitrogen functionalities during coal char combustion in O2/CO2 and O2/Ar atmospheres[J]. Energy Fuels, 2010, 25(1):240-245. doi: 10.1021/ef101134a [9] WÓJTOWICZ M A, PELS J R, MOULIJN J A. The fate of nitrogen functionalities in coal during pyrolysis and combustion[J]. Fuel, 1995, 74(4):507-516. doi: 10.1016-0016-2361(95)98352-F/ [10] FRIEBEL J, KÖPSEL R F W. The fate of nitrogen during pyrolysis of German low rank coals-A parameter study[J]. Fuel, 1999, 78(8):923-932. doi: 10.1016/S0016-2361(99)00008-3 [11] 游卓.富氧燃烧过程中的NOx控制及其系统效率研究[D].杭州: 浙江大学, 2013. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y2507767YOU Zhuo. NOx control and efficiency optimization of oxy-fuel combustion system[D]. Hangzhou: Zhejiang University, 2013. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y2507767 [12] 王贲.富氧燃烧方式下典型煤种的NO释放特性及其化学反应动力学模拟[D].武汉: 华中科技大学, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10487-1013014586.htmWANG Ben. The release characteristics and chemical kinetics simulation of typical coals in oxy-fuel combustion[D]. Wuhan: Huazhong University of Science and Technology, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10487-1013014586.htm [13] ZOU C, HE Y, SONG Y, HAN Q, LIU Y, GUO F, ZHENG C. The characteristics and mechanism of the NO formation during oxy-steam combustion[J]. Fuel, 2015, 158:874-883. doi: 10.1016/j.fuel.2015.06.034 [14] 黄庠永, 黄澈, 李小川, 孙景宝, 刘烨明.高浓度CO2气氛中煤氮官能团转化过程研究[J].华北电力大学学报(自然科学版), 2016, 43(6):97-101. doi: 10.3969/j.ISSN.1007-2691.2016.06.15HUANG Xiang-yong, HUANG Che, LI Xiao-chuan, SUN Jing-bao, LIU Ye-ming. Conversion characteristics of fuel nitrogen functionalities under atmosphere of high concentration of CO2[J]. J North China Electric Power Univ(Nat Sci Ed), 2016, 43(6):97-101. doi: 10.3969/j.ISSN.1007-2691.2016.06.15 [15] 王宗华.热解、气化过程中燃料N的形态转化及迁移规律研究[D].武汉: 华中科技大学, 2011. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=D186015WANG Zong-hua. Research on form transformation and releasing regulation of fuel-N during pyrolysis and gasification[D]. Wuhan: Huazhong University of Science and Technology, 2011. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=D186015 [16] 徐俊, 孙志君, 熊哲, 唐浩, 刘佳薇, 苏胜, 胡松, 汪一, 许凯, 向军.水蒸气对CO2/H2O气氛下煤焦结构的影响研究[J].工程热物理学报, 2017, (11):2467-2471. http://www.cnki.com.cn/Article/CJFDTOTAL-GCRB201711032.htmXU Jun, SUN Zhi-jun, XIONG Zhe, TANG Hao, LIU Jia-wei, SU Sheng, HU Song, WANG Yi, XU Kai, XIANG Jun. Effects of steam on the char structures under CO2/H2O atmospheres[J]. J Eng Thermophys-Rus, 2017, (11):2467-2471. http://www.cnki.com.cn/Article/CJFDTOTAL-GCRB201711032.htm [17] XU J, SU S, SUN Z, SI N, QING M, LIU L, HU S, WANG Y, TANG H. Effects of H2O gasification reaction on the characteristics of chars under oxy-fuel combustion conditions with wet recycle[J]. Energy Fuels, 2016, 30(11):9071-9079. doi: 10.1021/acs.energyfuels.6b01725 [18] KELEMEN S R, GORBATY M L, KWIATEK P J. Quantification of nitrogen forms in Argonne premium coals[J]. Energy Fuels, 1994, 8(4):896-906. doi: 10.1021/ef00046a013 [19] 李梅, 杨俊和, 张启锋, 常海洲, 孙慧.用XPS研究新西兰高硫煤热解过程中氮, 硫官能团的转变规律[J].燃料化学学报, 2013, 41(11):1287-1293. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18287.shtmlLI Mei, YANG Jun-he, ZHANG Qi-feng, CHANG Hai-zhou, SUN Hui. XPS study on transformation of N and S functional groups during pyrolysis of high sulfur New Zealand coal[J]. J Fuel Chem Technol, 2013, 41(11):1287-1293. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18287.shtml [20] 朱宏斌, 倪燕慧, 唐黎华, 郑志胜, 朱子彬.烟煤快速加氢热解的研究Ⅵ.煤和半焦中氮化学形态剖析[J].燃料化学学报, 2001, 29(2):124-128. doi: 10.3969/j.issn.0253-2409.2001.02.007ZHU Hong-bin, NI Yan-hui, TANG Li-hua, ZHENG Zhi-sheng, ZHU Zi-bin. Research of coal flash hydropyrolysis Ⅵ.chemical type analysis of nitrogen in coal and semi-coke[J]. J Fuel Chem Technol, 2001, 29(2):124-128 doi: 10.3969/j.issn.0253-2409.2001.02.007 -





 下载:
下载: