Theoretical study on enhancing the monolayer MoS2 photocatalytic water splitting with alloying and stress
-
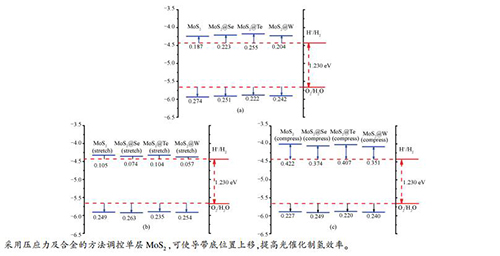
摘要: 基于密度泛函的第一性能原理计算方法研究了单层MoS2分别与MoSe2、MoTe2、WS2进行合金化, 以及加入2%应力条件下, 对光催化裂解水性能的影响。计算结果表明, 单层MoS2通过与MoSe2、MoTe2、MoWS2进行合金化, 并施加压应力两种手段进行调控, 可使带隙变大的同时, 提高CBM(conduction band minimum)带边位置, 从而提高光催化分解水的效率。通过能带和态密度的计算表明, 合金元素原子形成的不是孤立能级而是能带, 对载流子寿命影响小。Abstract: Based on the density functional theory, the photocatalytic water splitting reaction has been studied over the monolayer MoS2 alloying with MoSe2, MoTe2 and WS2 under stress condition. The calculated results show that the monolayer MoS2 alloyed with MoSe2, MoTe2 and MoWS2 under the compressive stress condition can increase the band gap and improve the position of CBM (conduction band minimum) band edge to enhance the efficiency of photocatalytic water splitting. The calculated energy band and density of states show that the alloy elements form energy band instead of isolated energy level, which has little effect on carrier life.
-
Key words:
- single-layer MoS2 /
- strain /
- photocatalytic water splitting /
- band structure
-
表 1 优化后晶格常数及组成比例
Table 1 Optimized structure lattice constant
Material and composition Lattice constant /nm Intrinsic MoS2 0.318 Mo8WS18 0.956 Mo9S17Te 0.961 Mo9S17Se 0.958 表 2 结构优化后的键长参数
Table 2 Bond length of optimized structure
Material MoS2 Mo8WS18 Mo9S17Te Mo9S17Se Bond length S-MO-S S-W-S S-Mo-Te S-Mo-Se /nm 0.243 0.241 0.272 0.254 0.241 0.241 0.240 0.240 Bond angle
/(°)81.98 82.37 78.15 80.49 -
[1] MARTHA S, CHANDRA SAHOO P, PARIDA K M. An overview on visible light responsive metal oxide based photocatalysts for hydrogen energy production[J]. RSC Adv, 2015, 5(76):61535-61553. doi: 10.1039/C5RA11682A [2] WANG F, DI VALENTIN C, PACCHIONI G. Doping of WO3 for photocatalytic water splitting:Hints from density functional theory[J]. J Phys Chem C, 2012, 116(16):8901-8909. doi: 10.1021/jp300867j [3] LI Y, LI Y L, ARAUJO C M, LUO W, AHUJIA R. Single-layer MoS2 as an efficient photocatalyst[J]. Catal Sci Technol, 2013, 3(9):2214. doi: 10.1039/c3cy00207a [4] KUDO A, MISEKI Y. Heterogeneous photocatalyst materials for water splitting[J]. Chem Soc Rev, 2009, 38(1):253-278. doi: 10.1039/B800489G [5] SPLENDIANI A, SUN L, ZHANG Y, LI T S. Emerging photoluminescence in monolayer MoS2[J]. Nano Lett, 2010(10):1271-1275. http://li.mit.edu/S/2d/Paper/Splendiani10Sun.pdf [6] JARAMILLO T F, JORGENSEN K P, BONDE J, NIELSEN J H, HORCH S, CHORKENDORFF Ⅰ. Identification of active edge sites for electrochemical H2 evolution from MoS2 nanocatalysts[J]. Science, 2007, 317(5834):100-102. doi: 10.1126/science.1141483 [7] HU W, LIN L, ZHANG R, YANG C, YANG J L. Highly Efficient photocatalytic water splitting over edge-modified phosphorene nanoribbons[J]. J Am Chem Soc, 2017, 139(43):15429-15436. doi: 10.1021/jacs.7b08474 [8] ZHUANG H L, HENNIG R G. Single-layer group-Ⅲ monochalcogenide photocatalysts for water splitting[J]. Chem Mater, 2013, 25(15):3232-3238. doi: 10.1021/cm401661x [9] DOLUI K, RUNGGER Ⅰ, DAS PEMMARAJU C, SANVITO S. Possible doping strategies for MoS2 monolayers:An ab initio study[J]. Phys Rev B, 2013, 88(7):075420. doi: 10.1103/PhysRevB.88.075420 [10] MAK K F, LEE C, HONE J, SHAN J, HEINZ T F. Atomically thin MoS2:A new direct-gap semiconductor[J]. Phys Rev Lett, 2010, 105(13):136805. doi: 10.1103/PhysRevLett.105.136805 [11] HINNEMANN B, MOSES P G, BONDE J, JØRGENSEN K P, NIELSEN J H, HORCH S, CHORKENDORFF Ⅰ, NØRSKOV J. Biomimetic hydrogen evolution:MoS2 nanoparticles as catalyst for hydrogen evolution[J]. J Am Chem Soc, 2005, 127(15):5308-5309. doi: 10.1021/ja0504690 [12] YANG L, CUI X, ZHANG J, WANG K, SHEN M, ZENG S S, DAYEH S A, FENG L, XIANG B. Lattice strain effects on the optical properties of MoS2 nanosheets[J]. Sci Rep, 2015, 4(1):5649. doi: 10.1038/srep05649 [13] MA Y, DAI Y, GUO M, NIU C W, ZHU Y T, HUANG B B. Evidence of the existence of magnetism in pristine VX2 monolayers (X=S, Se) and their strain-induced tunable magnetic properties[J]. ACS Nano, 2012, 6(2):1695-1701. doi: 10.1021/nn204667z [14] LEE J H, JANG W S, HAN S W, BAIK H K. Efficient hydrogen evolution by mechanically strained MoS2 nanosheets[J]. Langmuir, 2014, 30(32):9866-9873. doi: 10.1021/la501349k [15] KANG J, TONGAY S, ZHOU J, LI J B, WU J Q. Band offsets and heterostructures of two-dimensional semiconductors[J]. Appl Phys Lett, 2013, 102(1):012111. doi: 10.1063/1.4774090 [16] YUE Q, CHANG S, QIN S, LI J B. Functionalization of monolayer MoS2 by substitutional doping:A first-principles study[J]. Phys Lett A, 2013, 377(19/20):1362-1367. https://www.sciencedirect.com/science/article/pii/S0375960113003198 [17] KIRAN V, MUKHERJEE D, JENJETI R N, SAMPATH S. Active guests in the MoS2/MoSe2 host lattice:Efficient hydrogen evolution using few-layer alloys of MoS2(1-x) Se2x[J]. Nanoscale, 2014, 6(21):12856-12863. doi: 10.1039/C4NR03716B [18] LIN Z, THEE M T, ELÍAS A L, FENG S, ZHOU C J, FUJISAWA K, PEREA-LÓPEZ N, CAROZO V, TERRONES H, TERRONES M. Facile synthesis of MoS2 and Mox W1-xS2 triangular monolayers[J]. Apl Mater, 2014, 2(9):092514. doi: 10.1063/1.4895469 [19] YANG H, ZHANG T, ZHU H, ZHANG M M, WU W W, DU M L. Synthesis of a MoS2(1-x)Se2x ternary alloy on carbon nanofibers as the high efficient water splitting electrocatalyst[J]. Int J Hydrogen Energy, 2017, 42(4):1912-1918. doi: 10.1016/j.ijhydene.2016.10.075 [20] ZHUANG H L, HENNIG R G. Computational search for single-layer transition-metal dichalcogenide photocatalysts[J]. J Phys Chem C, 2013, 117(40):20440-20445. doi: 10.1021/jp405808a [21] LI C, FAN B, LI W, WEN W L, LIU Y, WANG T, SHENG K, YIN Y. Bandgap engineering of monolayer MoS2 under strain:A DFT study[J]. J Korean Phys Soc, 2015, 66(11):1789-1793. doi: 10.3938/jkps.66.1789 [22] LI T. Ideal strength and phonon instability in single-layer MoS2[J]. Phys Rev B, 2012, 85(23):235407. doi: 10.1103/PhysRevB.85.235407 [23] JOENSEN P, CROZIER E D, ALBERDING N, FRINDT R F. A study of single-layer and restacked MoS2 by X-ray diffraction and X-ray absorption spectroscopy[J]. J Phys C:Solid State Phys, 1987, 20(26):4043-4053. doi: 10.1088/0022-3719/20/26/009 [24] KONG L J, LIU G H, QIANG L. Electronic and optical properties of O-doped monolayer MoS2[J]. Comput Mater Sci, 2016, 111:416-423. doi: 10.1016/j.commatsci.2015.10.001 [25] 李刚, 陈敏强, 赵世雄, 李朋伟, 胡杰, 桑胜波, 侯静静. Se掺杂对单层MoS2电子能带结构和光吸收性质的影响[J].物理化学学报, 2016, 32(12):2905-2912. doi: 10.3866/PKU.WHXB201609201LI Gang, CHEN Min-qiang, ZHAO Shi-xiong, LI Peng-wei, HU Jie, SANG Sheng-bo, HOU Jing-jing. Effect of Se doping on the electronic band structure and optical absorption properties of single layer MoS2[J]. Acta Phys Chim Sin, 2016, 32(12):2905-2912. doi: 10.3866/PKU.WHXB201609201 [26] SINGH N, JABBOUR G, SCHWINGENSCHLÖGL U. Optical and photocatalytic properties of two-dimensional MoS2[J]. Eur Phys J B, 2012, 85(11):392. doi: 10.1140/epjb/e2012-30449-7 -





 下载:
下载: