Catalytic performance of the Mn-Ce catalysts in lean methane combustion prepared by a redox co-precipitation method
-
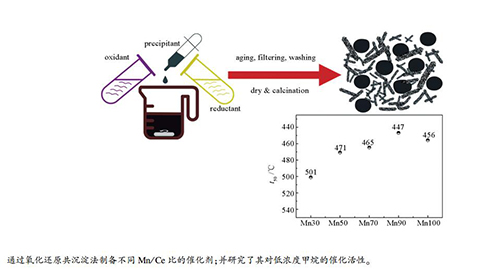
摘要: 采用氧化还原共沉淀法制备了一系列不同Mn/Ce比的Mn-Ce催化剂,用N2吸附、XRD、XRF、XPS等手段进行了表征,对其低浓度甲烷催化燃烧活性进行了研究。结果表明,Mn/Ce比对Mn-Ce催化剂的活性有较大的影响;当Mn/Ce比从3:7增加到9:1时,其催化活性逐渐增加,甲烷转化率为50%的温度(t50)从501 ℃降低到446 ℃;而进一步增加Mn含量则会导致其催化活性降低。表征结果显示,Mn-Ce催化剂活性与其比表面积、表面Mn4+浓度、Ce3+含量和晶格氧浓度等密切相关;物相KMn8O16有利于Mn-Ce催化剂活性的提升。Abstract: A series of Mn-Ce catalysts with different Mn/Ce ratios were prepared by a redox co-precipitation method and characterized by N2 sorption, XRD, XRF and XPS; their catalytic performance in lean methane combustion was investigated. The results indicate that the Mn/Ce ratio has a great influence on the activity of Mn-Ce catalyst in lean methane combustion. With an increase of the Mn/Ce ratio from 3:7 to 9:1, the activity of the Mn-Ce catalyst increases gradually and the reaction temperature needed for a methane conversion of 50% (t50) decreases from 501 ℃ to 446 ℃; however, a further increase in the Mn/Ce ratio may lead to a decrease in the catalytic activity. The performance of Mn-Ce catalyst is related to many factors such as the surface area and the concentration of higher valence manganese (Mn4+) species, lower valence cerium (Ce3+) species and lattice oxygen; in particular, KMn8O16 is of benefit to enhancing the activity of Mn-Ce catalyst.
-
Key words:
- lean methane /
- Mn-Ce catalyst /
- catalytic combustion /
- redox-precipitation method
-
表 1 不同催化剂的氮吸附表征结果
Table 1 Nitroge sorption results for different catalysts
Sample BET surface area A/ (m2·g-1) Pore volume v/(cm3·g-1) Average pore diameter d/nm Mn30 100 0.392 10.7 Mn50 118 0.786 21.6 Mn70 117 0.763 23.6 Mn90 111 0.624 21.8 Mn100 39 0.253 25.8 表 2 MnCe催化剂的XRF表征
Table 2 XRF results of different catalysts
Sample Metal content wmol/% K Mn Ce Mn/Cecal Mn/Ceexp Mn30 13.61 24.91 61.48 0.43 0.41 Mn50 5.61 43.83 50.56 1.00 0.86 Mn70 5.66 65.32 29.02 2.33 2.25 Mn90 12.23 78.55 9.22 9.00 8.52 Mn100 13.83 86.17 0 - - 表 3 Mn-Ce催化剂表面的的元素组成
Table 3 Surface atomic composition of binary MnOx-CeO2 catalysts determined from XPS spectra
Sample O 1s position E/eV Oα/(Oα+ Oβ) Mn 2p3/2 position E/eV Mn4+/Mn3+ Ce3+/(Ce4++ Ce3+) Oα Oβ Oω Mn4+ Mn3+ Mn30 529.5 531.4 533.0 0.64 642.9 641.9 0.72 0.11 Mn50 529.4 531.4 533.0 0.70 642.5 641.6 0.94 0.17 Mn70 529.5 531.4 533.0 0.83 642.4 641.6 1.95 0.19 Mn90 529.7 531.6 533.0 0.85 642.6 641.6 3.56 0.28 Mn100 529.7 531.6 533.0 0.79 642.6 641.6 7.67 - -
[1] REAY D, SMITH P, AMSTEL A. Methane and Climate Change[M]. 2010. [2] CARGNELLO M, JAÉN J J D, GARRIDO J C H, BAKHMUTSKY K, MONTINI T, GÁMEZ J J C, GORTE R J, FORNASIERO P. Exceptional activity for methane combustion over modular Pd@CeO2 subunits on functionalized Al2O3[J]. Science, 2012, 337(6095):713-717. doi: 10.1126/science.1222887 [3] KERR R A. A quick (partial) fix for an ailing atmosphere[J]. Science, 2012, 335(6065):156-156. doi: 10.1126/science.335.6065.156 [4] STOLAROFF J K, BHATTACHARYYA S, SMITH C A, BOURCIER W L, CAMERON-SMITH P J, AINES R D. Review of methane mitigation technologies with application to rapid release of methane from the arctic[J]. Environ Sci Technol, 2012, 46(12):6455-6469. doi: 10.1021/es204686w [5] 霍春秀, 李强.乏风瓦斯催化燃烧实验研究[J].矿业安全与环保, 2014, 41(4):1-3 doi: 10.3969/j.issn.1008-4495.2014.04.001GUO Chun-xiu, LI Qiang. Experimental study on catalytic combustion of ventilation air methane[J]. Min Saf Environ Prot, 2014, 41(4):1-3. doi: 10.3969/j.issn.1008-4495.2014.04.001 [6] 李旭.世界煤层气开发利用现状[J].煤炭加工与综合利用, 2006, (6):41-45. doi: 10.3969/j.issn.1005-8397.2006.06.016LI Xu. Progress of development and utilization of coalbed methane in the world[J]. Coal Process Compr Util, 2006, (6):41-45. doi: 10.3969/j.issn.1005-8397.2006.06.016 [7] OBER J A. Mineral commodity summaries 2018[R]. Reston, VA: U.S. Geological Survey, 2018. [8] MONAI M, MONTINI T, MELCHIONNA M, DUCHOŇT, KÚŠP, CHEN C, TSUD N, NASI L, PRINCE KC, VELTRUSKÁ K, MATOLÍN V, KHADER M M, GORTE R J, FORNASIERO P. The effect of sulfur dioxide on the activity of hierarchical Pd-based catalysts in methane combustion[J]. Appl Catal B:Environ, 2017, 202:72-83. doi: 10.1016/j.apcatb.2016.09.016 [9] CHEN J, ARANDIYAN H, GAO X, LI J. Recent advances in catalysts for methane combustion[J]. Catal Surv Asia, 2015, 19(3):140-171. doi: 10.1007/s10563-015-9191-5 [10] WANG B, ALBARRACÍN-SUAZO S, PAGÁN-TORRES Y, NIKOLLA E. Advances in methane conversion processes[J]. Catal Today, 2017, 285:147-158. doi: 10.1016/j.cattod.2017.01.023 [11] ZHANG Y, QIN Z, WANG G, ZHU H, DONG M, LI S, WU Z, LI Z, WU Z, ZHANG J. Catalytic performance of MnOx-NiO composite oxide in lean methane combustion at low temperature[J]. Appl Catal B:Environ, 2013, 129(2):172-181. http://www.sciencedirect.com/science/article/pii/S0926337312004225 [12] HU Z, QIU S, YOU Y, GUO Y, GUO Y L, WANG L, ZHAN W C, LU G Z. Hydrothermal synthesis of NiCeOx nanosheets and its application to the total oxidation of propane[J]. Appl Catal B:Environ, 2018, 225:110-120. doi: 10.1016/j.apcatb.2017.08.068 [13] SHENG Z Y, HU Y F, XUE J M, WANG X M, LIAO W P. A novel co-precipitation method for preparation of Mn-Ce/TiO2 composites for NOx reduction with NH3 at low temperature[J]. Environ Technol, 2012, 33(21):2421-2428. doi: 10.1080/09593330.2012.671370 [14] CYBULSKI A. Catalytic wet air oxidation:Are monolithic catalysts and reactors feasible?[J]. Ind Eng Chem Res, 2007, 46(12):4007-4033. doi: 10.1021/ie060906z [15] DING Z Y, LI L X, WADE D, GLOYNA E F. Supercritical water oxidation of NH3 over a MnO2/CeO2 catalyst[J]. Ind Eng Chem Res, 1998, 37(5):1707-1716. doi: 10.1021/ie9709345 [16] FIUK M M, ADAMSKI A. Activity of MnOx-CeO2 catalysts in combustion of low concentrated methane[J]. Catal Today, 2015, 257:131-135. doi: 10.1016/j.cattod.2015.01.029 [17] SHI L M, CHU W, QU F F, LUO S H. Low-temperature catalytic combustion of methane over MnOx-CeO2 mixed oxide catalysts:Effect of preparation method[J]. Catal Lett, 2007, 113(1/2):59-64. doi: 10.1007/s10562-006-9012-6 [18] ZHANG H, YANG W L, LI D, WANG X Y. Influence of preparation method on the performance of Mn-Ce-O catalysts[J]. React Kinet Catal Lett, 2009, 97(2):263-268. doi: 10.1007/s11144-009-0024-2 [19] 李树娜, 宋佩, 张金丽, 贺小霞, 解一昕, 张亚刚, 王瑞义, 李志凯, 朱华青. CeO2-MnOx催化剂形貌对低浓度甲烷催化燃烧反应性能的影响[J].燃料化学学报, 2018, 46(5):615-624. doi: 10.3969/j.issn.0253-2409.2018.05.015LI Shu-na, SONG Pei, ZHANG Jin-li, HE Xiao-xia, XIE Yi-xin, ZHANG Ya-gang, WANG Rui-yi, LI Zhi-kai, ZHU Hua-qing. morphology effect of CeO2-MnOx catalyst on their catalytic performance in lean methane combustion[J]. J Fuel Chem Technol, 2018, 46(5):615-624. doi: 10.3969/j.issn.0253-2409.2018.05.015 [20] 刘长春, 於俊杰, 蒋政, 陶炎鑫, 郝郑平, 何绪文. Ce(1-x)MnxO(2-a)复合氧化物催化剂甲烷催化燃烧性能的研究[J].无机化学学报, 2007, (2):217-224 doi: 10.3321/j.issn:1001-4861.2007.02.005LIU Chang-chun, YU Jun-jie, JIANG Zheng, TAO Yan-xin, HAO Zheng-ping, HE Xu-wen. Methane catalytic combustion on Ce1-xMnxO2-a mixed oxide catalysts[J]. Chin J Inorg Chem, 2007, (2):217-224. doi: 10.3321/j.issn:1001-4861.2007.02.005 [21] ZHANG P, LU H, ZHOU Y, ZHANG L, WU Z, YANG S, SHI H, ZHU Q, CHEN Y, DAI S. Mesoporous MnCeOx solid solutions for low temperature and selective oxidation of hydrocarbons[J]. Nat Commun, 2015, 6:8446. doi: 10.1038/ncomms9446 [22] WU Y, LU Y, SONG C, MA Z, XING S, GAO Y. A novel redox-precipitation method for the preparation of α-MnO2 with a high surface Mn4+ concentration and its activity toward complete catalytic oxidation of o-xylene[J]. Catal Today, 2013, 201:32-39. doi: 10.1016/j.cattod.2012.04.032 [23] WANG X, LIU Y, ZHANG Y, ZHANG T, CHANG H, ZHANG Y, JIANG L. Structural requirements of manganese oxides for methane oxidation:XAS spectroscopy and transition-state studies[J]. Appl Catal B:Environ, 2018, 229:52-62. doi: 10.1016/j.apcatb.2018.02.007 [24] ARENA F, GUMINA B, LOMBARDO AF, ESPRO C, PATTI A, SPADARO L, SPICCIA L. Nanostructured MnOx catalysts in the liquid phase selective oxidation of benzyl alcohol with oxygen:Part I. Effects of Ce and Fe addition on structure and reactivity[J]. Appl Catal B:Environ, 2015, 162:260-267. doi: 10.1016/j.apcatb.2014.06.054 [25] ARENA F, TRUNFIO G, NEGRO J, FAZIO B, SPADARO L. Basic evidence of the molecular dispersion of MnCeOx catalysts synthesized via a novel "redox-precipitation" route[J]. Chem Mater, 2007, 19(9):2269-2276. doi: 10.1021/cm070198n [26] XU J, LI P, SONG X F, HE C H, YU J G, HAN Y F. Operando raman spectroscopy for determining the active phase in one- dimensional Mn1-xCexO2±y nanorod catalysts during methane combustion[J]. J Phys Chem Lett, 2015, 1(10):1648-1654. doi: 10.1021/jz1004522 [27] PAPAVASILIOU J, AVGOUROPOULOS G, IOANNIDES T. In situ combustion synthesis of structured Cu-Ce-O and Cu-Mn-O catalysts for the production and purification of hydrogen[J]. Appl Catal B:Environ, 2006, 66(3):168-174. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=fa379eb7d9e49bed1e27dd4bed63664e [28] 李云霞.低温等离子体-锰氧化物联合处理气体中二硫化碳和甲苯[D].合肥: 合肥工业大学, 2016. http://cdmd.cnki.com.cn/Article/CDMD-10359-1017037340.htmLI Yun-xia. Reasearch on treatment of caron disulfide and toluene gas by low temperature plasma-manganese oxide[D]. Heifei: Heifei University of Technology, 2016. http://cdmd.cnki.com.cn/Article/CDMD-10359-1017037340.htm [29] JIRÁTOVÁ K, MIKULOVÁ J, KLEMPA J, GRYGAR T, BASTL Z, KOVANDA F. Modification of Co-Mn-Al mixed oxide with potassium and its effect on deep oxidation of VOC[J]. Appl Catal A:Gen, 2009, 361(1):106-116. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=426c58b948e9112a6112c482963e9d7a [30] KUMAR M, YUN J H, BHATT V, SINGH B, KIM J, KIM J S, KIM B S, LEE CY. Role of Ce3+ valence state and surface oxygen vacancies on enhanced electrochemical performance of single step solvothermally synthesized CeO2 nanoparticles[J]. Electrochim Acta, 2018, 284:709-720. doi: 10.1016/j.electacta.2018.07.184 [31] CHEN X, SHEN Y F, SUIB S L, O'Young C L. Characterization of manganese oxide octahedral molecular sieve (M-OMS-2) materials with different metal cation dopants[J]. Chem Mater, 2002, 14(2):940-948. doi: 10.1021/cm000868o [32] HAN Y F, CHEN L, RAMESH K, WIDJAJA E, CHILUKOTI S, KESUMAWINATA SURJAMI I, CHEN J. Kinetic and spectroscopic study of methane combustion over α-Mn2O3 nanocrystal catalysts[J]. J Catal, 2008, 253(2):261-268. doi: 10.1016/j.jcat.2007.11.010 -





 下载:
下载: