Properties of palm oil biodiesels derived from different alcohols
-
摘要: 以棕榈油为研究对象,分别与甲醇、乙醇、异丙醇和异丁醇进行酯交换反应制备了不同酯基结构的生物柴油。利用差示扫描量热仪和应力控制流变仪分析了不同酯基结构棕榈油生物柴油的结晶行为和低温流变性能,同时分析了酯基结构对生物柴油的氧化稳定性,40℃时的动力黏度、20℃时的密度等重要性质的影响。结果表明,随着醇的碳链长度的增加,棕榈油生物柴油的析蜡点和胶凝点均降低,特别是棕榈油异丁酯的析蜡点和胶凝点分别降低到了-2.57和-8.09℃,低温流动性得到了明显改善,且氧化诱导期略有延长,氧化稳定性有所改善。生物柴油的密度和黏度随着酯基结构的不同而有所变化,但其数值都符合中国生物柴油标准。Abstract: The palm oil biodiesels with different ester alkyls were prepared through transesterification of palm oil with methanol, ethanol, isopropanol and isobutanol. The crystallization behavior and cold flow properties of the palm oil biodiesels were characterized by differential scanning calorimeter (DSC) and stress controlled rheometer; the effect of ester alkyl on certain important properties such as the oxidation stability, kinematic viscosity (40℃) and density (20℃) was then investigated. The results suggested that the palm oil biodiesels with larger ester alkyls exhibit lower crystal precipitation temperature and gelation point than those biodiesels with ester methyl; especially, the crystal precipitation temperature and gelation point for the palm oil biodiesel derived from isobutanol reach -2.57 and -8.09℃, respectively. An increase in the length of ester alkyl chain in alcohol moiety can significantly improve the cold flow properties of the palm oil biodiesels, by slightly prolonging the oxidation induction period and enhancing the oxidative stability. The palm oil biodiesels with different ester alkyls are also different in their kinematic viscosity and density; however, all these values can meet the requirement from the biodiesel standard of China.
-
Key words:
- palm oil biodiesel /
- ester alkyl /
- crystallization behavior /
- cold flow properties /
- oxidation stability
-
表 1 不同醇制备生物柴油的实验条件
Table 1 Parameters for the preparation of palm oil biodiesels with different alcohols
Alcohol Molar ratio Catalyst Catalyst quantity*
w/%Reaction temperature
t/℃Reaction time
t/hStirring speed /
(r·min-1)Methanol 6 KOH 1.0 60 1 600 Ethanol 6 KOH 1.0 60 3 600 Isopropanol 9 H2SO4 1.5 84 4 600 Isobutanol 9 H2SO4 1.5 100 4 600 *: weight percentage of the catalyst used on the basis of oil 表 2 不同酯基结构棕榈油生物柴油的组成
Table 2 Composition of palm oil biodiesels derived from different alcohols
Fatty acid Composition w/% methyl ester ethyl ester isopropyl ester isobutyl ester C14:0 1.23 1.17 1.15 1.06 C16:0 40.41 39.70 39.25 39.14 C18:0 4.40 4.55 4.26 4.71 C16:1 0.22 0.53 0.55 0.28 C18:1 43.52 43.61 43.66 43.60 C18:2 8.41 8.42 8.25 8.28 C18:3 0.51 0.52 0.45 0.28 Others 1.30 1.50 2.43 2.65 表 3 不同酯基结构棕榈油生物柴油的特性温度
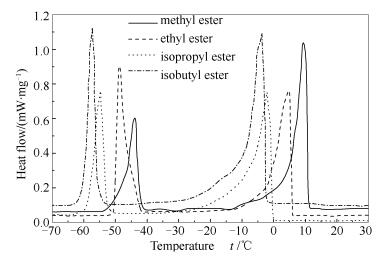
Table 3 Characteristic temperatures of palm oil biodiesel derived from different alcohols
Ester tco/℃ tp1/℃ t2/℃ tp2/℃ Methyl 11.37 9.48 -41.36 -43.80 Ethyl 5.93 4.90 -42.13 -49.03 Isopropyl -0.28 -2.08 -52.57 -54.81 Isobutyl -2.57 -3.74 -53.56 -57.15 表 4 不同酯基结构棕榈油生物柴油的tgel,tδ及Δ(tco-tgel)
Table 4 The tgel, tδ and Δ(tco-tgel) of palm oil biodiesels derived from different alcohols
Ester tδ/℃ tgel/℃ Δ(tco-tgel)/℃ Methyl ester 11.30 10.30 1.07 Ethyl ester 4.39 3.39 2.54 Isopropyl ester -2.60 -3.59 3.31 Isobutyl ester -7.60 -8.09 5.52 -
[1] KNOTHE G, STEIDLEY K R. Kinematic viscosity of biodiesel fuel components and related compounds. Influence of compound structure and comparison to petrodiesel fuel components[J]. Fuel, 2005, 84(9):1059-1065. https://www.researchgate.net/publication/222514930_Kinematic_viscosity_of_biodiesel_fuel_components_and_related_compounds_Influence_of_compound_structure_and_comparison_to_petrodiesel_fuel_components [2] ATABANI A E, SILITONGA A S, BADRUDDIN I A, MAHLIA T M I, MASJUKI H H, MEKHILEF S. A comprehensive review on biodiesel as an alternative energy resource and its characteristics[J]. Renew Sustainable Energy Rev, 2012, 16(4):2070-2093. doi: 10.1016/j.rser.2012.01.003 [3] MOSER B R. Biodiesel production, properties, and feedstocks[J]. In Vitro Cell Dev Biol Plant, 2009, 45(3):229-266. doi: 10.1007/s11627-009-9204-z [4] ROBERT O D.Cold flow properties of soybean oil fatty acid monoalkyl ester admixtures[J]. Energy Fuels, 2009, 23(8):4082-4091. doi: 10.1021/ef9002582 [5] 陈秀, 袁银南, 来永斌, 王利平.生物柴油组成与组分结构对其低温流动性的影响[J].石油学报(石油加工), 2009, 25(5):673-677. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG200905011.htmCHEN Xiu, YUAN Yin-nan, LAI Yong-bin, WANG Li-ping. Impact of composition and molecular structure upon the cold flow proprtties for biodiesel[J]. Acta Pet Sin (Pet Process Sect), 2009, 25(5):673-677. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG200905011.htm [6] 陈五花, 王业飞, 陈建.低温下生物柴油的胶凝特性研究[J].燃料化学学报, 2015, 43(6):669-676. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18639.shtmlCHEN Wu-hua, WANG Ye-fei, CHEN Jian. Gelling properties of biodiesel at low temperatures[J]. J Fuel Chem Technol, 2015, 43(6):669-676. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18639.shtml [7] 白禹, 李雪, 章留留, 王渊涛, 顾全荣.生物柴油的催化改性对其冷滤点的影响[J].燃料化学学报, 2009, 37(1):53-57. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17401.shtmlBAI Yu, LI Xue, ZHANG Liu-liu, WANG Yuan-tao, GU Quan-rong. Effect of modification of biodiesel by HZSM-5 zeolite on cold filter plug point[J]. J Fuel Chem Technol, 2009, 37(1):53-57. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17401.shtml [8] CHEN B S, SUN Y Q, FANG J H, WANG J, JIANG W. Effect of cold flow improvers on flow properties of soybean biodiesel[J]. Biomass Bioenergy, 2010, 34(9):1309-1313. doi: 10.1016/j.biombioe.2010.04.001 [9] WANG J N, CAO L C, HAN S. Effect of polymeric cold flow improvers on cold flow properties of biodiesel from waste cooking oil[J]. Fuel, 2014, 117(1):876-881. https://www.researchgate.net/publication/270872885_Effect_of_polymeric_cold_flow_improvers_on_flow_properties_of_biodiesel_from_waste_cooking_oil?_sg=WCDoZoOYYjmHfXie9-6Tk6Q1H9N4WYTv4YEKgHrvL2iW-psW1nB-mWF_k1oUXPMZ09_zX2wM0okoX7fsBhQlyg [10] 耿再新, 张苗娟, 付丽丽, 蒋登高.低凝点支链醇生物柴油合成新工艺研究[J].高校化学工程学报, 2012, 26(6):1073-1076. http://www.cnki.com.cn/Article/CJFDTOTAL-GXHX201206029.htmGENG Zai-xin, ZHANG Miao-juan, FU Li-li, JIANG Deng-gao. Study on the new synthetic technology of the branched-chain alcohol biodiesel with low freezing point[J]. J Chem Eng Chin Univ, 2012, 26(6):1073-1076. http://www.cnki.com.cn/Article/CJFDTOTAL-GXHX201206029.htm [11] MALINS K, KAMPARS V, KAMPARE R, PRILUCKA J, BRINKS J, MURNIEKS R, APSENIECE L. Properties of rapeseed oil fatty acid alkyl esters derived from different alcohols[J]. Fuel, 2014, 137(4):28-35. https://www.researchgate.net/publication/264827261_Properties_of_rapeseed_oil_fatty_acid_alkyl_esters_derived_from_different_alcohols [12] NAINWAL S, SHARMA N, SHARMA A S, JAIN S, JAIN S. Cold flow properties improvement of Jatropha curcas biodiesel and waste cooking oil biodiesel using winterization and blending[J]. Energy, 2015, 89:702-707. doi: 10.1016/j.energy.2015.05.147 [13] 吕涯, 李骏, 欧阳福生.生物柴油调和对其低温流动性能的改善[J].燃料化学学报, 2011, 39(3):189-193. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17707.shtmlLÜ Ya, LI Jun, OUYANG Fu-sheng. Effect of biodiesels blending on their low-temperature fluidity[J]. J Fuel Chem Technol, 2011, 39(3):189-193. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17707.shtml [14] SERRANO M, OLIVEROS R, SÁNCHEZ M, MORASCHINI A, MARTÍNEZ M, ARACIL J. Influence of blending vegetable oil mwthyl esters on biodiesel fule properties:Oxidative stability and cold flow properties[J]. Energy, 2014, 65(2):109-115. https://www.researchgate.net/profile/Marcos_Sanchez7/publication/259514751_Influence_of_blending_vegetable_oil_methyl_esters_on_biodiesel_fuel_properties_Oxidative_stability_and_cold_flow_properties/links/54e9d4110cf2f7aa4d5430b2.pdf [15] ROMANO S D, SORICHETTI P A. Dielectric Spectroscopy in Biodiesel Production and Characterization, Green Energy and Technology[M]. NewYork:Springer-Verlag London Limited, 2011:71-82. [16] 徐辉辉.生物柴油氧化安定性研究[D].郑州:郑州大学, 2010.XU Hui-hui. Study of oxidative atability of biodiesel[D]. Zhengzhou:Zhengzhou University, 2010. [17] PULLEN J, SAEED K. An overview of biodiesel oxidation stability[J]. Renewable Sustainable Energy Rev, 2012, 16(8):5924-5950. doi: 10.1016/j.rser.2012.06.024 [18] ZULETA E C, RIOS L A, BENJUMEA P N. Oxidative stability and cold flow behavior of palm, sacha-inchi, jatropha and castor oil biodiesel blends[J]. Fuel Process Technol, 2012, 102(1):96-101. [19] NIMCEVIC D, PUNTIGAM R, WÖRGETTER M, GAPES R. Preparation of rapeseed oil esters of lower aliphatic alcohols[J]. J Am Oil Chem Soc, 2000, 77(3):275-280. doi: 10.1007/s11746-000-0045-1 [20] 陈五花, 陈建, 蒋金兴, 陈本军.生物柴油在低温下析出晶体的热力学规律研究[J].石油炼制与化工, 2014, 45(3):14-17. http://www.cnki.com.cn/Article/CJFDTOTAL-SYLH201403006.htmCHEN Wu-hua, CHEN Jian, JIANG Jin-xing, CHEN Ben-jun. Crystal precipitation law of biodiesel at low temperatures[J]. Pet Process Petrochem, 2014, 45(3):14-17. http://www.cnki.com.cn/Article/CJFDTOTAL-SYLH201403006.htm [21] REFAAT A A. Correlation between the chemical structure of biodiesel and its physical properties[J]. Int J Environ Sci Technol, 2009, 6(4):677-694. doi: 10.1007/BF03326109 [22] HOLMAN R A, ELMER O C. The rates of oxidation of unsaturated fatty acids and esters[J]. J Am Oil Chem Soc, 1947, 24(4):127-129. doi: 10.1007/BF02643258 [23] MOSER B R. Comparative oxidative stability of fatty acid alkyl esters by accelerated methods[J]. J Am Oil Chem Soc, 2009, 86(7):699-706. doi: 10.1007/s11746-009-1376-5 -





 下载:
下载: