Transformation of sulfur forms during pyrolysis of mild liquefaction solid product of Hami coal
-
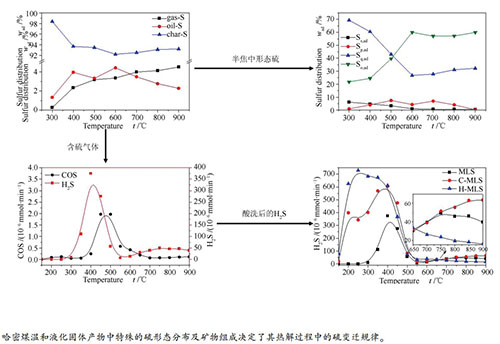
摘要: 利用固定床反应器研究了哈密煤温和液化固体产物(MLS)在热解过程中含硫气体的释放规律以及不同形态硫的变迁规律,并分析了矿物质对硫变迁规律的影响。结果表明,在实验考察的条件范围内,MLS热解过程中大部分的硫残留在半焦中,仅有不到10%的硫迁移到焦油中或转化为含硫气体逸出。热解生成的含硫气体以H2S为主,当热解温度为400℃时H2S的逸出速率达到最大。通过改进方法测定了MLS及其热解半焦中各种形态硫的含量,发现MLS热解过程中以硫化物硫和有机硫的分解和转化为主。随着热解温度的升高,MLS中有机硫逐渐分解并以含硫气体的形式逸出;当热解温度低于600℃时,MLS中硫化物硫逐渐转化为含硫气体、有机硫和少量的黄铁矿硫;当热解温度高于600℃时,MLS中碱性矿物质吸收气相中的H2S转化为硫化物硫,硫化物硫缓慢增加。醋酸酸洗可以保留MLS中大部分的硫化物硫,且酸洗后MLS热解生成的H2S逸出速率增大,峰温向低温方向移动;当热解温度高于600℃时,有机硫和硫化物硫的脱硫反应速率降低,并且MLS中的碱性矿物质与H2S反应生成金属硫化物,导致H2S逸出速率明显降低。Abstract: The sulfur-containing gases evolution and the transformation of sulfur during pyrolysis of mild liquefaction solid product (MLS) were studied in a fixed-bed reactor. Meanwhile, the effects of mineral matters on the sulfur transformation were explored. The results show that most of the sulfur remains in the char under the experimental conditions in this work. Less than 10% of the sulfur migrates into tar and sulfur-containing gases. The sulfur-containing gases generated in pyrolysis is mainly H2S. In addition, the generation rate of H2S reaches the maximum when MLS is pyrolyzed at 400 ℃. The contents of various forms of sulfur in both MLS and its pyrolysis char were determined by the modified method. The analysis shows that the decomposition and conversion of sulfide sulfur and organic sulfur are the main reactions of sulfur-containing matters during the pyrolysis process of MLS. As the pyrolysis temperature increases, the organic sulfur in MLS gradually decomposes and is converted into sulfur-containing gases. When the temperature is lower than 600 ℃, the sulfide sulfur in MLS is gradually transformed into sulfur-containing gases, organic sulfur and a small amount of pyrite sulfur. When the pyrolysis temperature is higher than 600 ℃, the alkaline minerals in MLS would absorb H2S and convert into sulfide sulfur. Consequently, the content of sulfide sulfur slowly increases. Acetic acid pickling treatment can retain most of the sulfide sulfur in MLS. After pickling, the generation rate of H2S during MLS pyrolysis increases, and the peak temperature shifts to the lower temperature. When the pyrolysis temperature is over 600 ℃ the desulfurization reaction rate of the organic sulfur and sulfide sulfur decreases. Meanwhile, the alkaline minerals in MLS can react with H2S to form metal sulfides, leading to a significant decrease in H2S generation rate.
-
表 1 MLS的工业分析和元素分析
Table 1 Proximate and ultimate analyses of MLS
Proximate analysis wad/% Ultimate analysis wdaf/% St, ad/% H/C(atomic ratio) M A V C H O* N 0.26 15.28 50.87 87.73 6.38 2.68 1.12 1.76 0.87 ad: air-dried basis; daf: dry and ash-free basis; M: moisture; St: total sulfur; *: by difference 表 2 MLS的灰成分分析
Table 2 Ash composition of MLS
Content w/% SiO2 Al2O3 Fe2O3 CaO MgO SO3 TiO2 K2O Na2O P2O5 11.07 8.30 25.87 23.57 1.26 26.83 0.19 0.27 2.37 0.21 表 3 国标和改进方法测定的MLS中不同形态硫含量
Table 3 Different forms of sulfur content in MLS determined by standard method and improved method
Method Content wad/% St Ss Sp Sq So Improved-method 1.76 0.09 0.08 1.10 0.49 Standard method 1.76 0.09 0.08 - 1.59 St: total sulfur; Ss: sulfate sulfur; Sp: pyritic sulfur; Sq: sulfide sulfur; So: organic sulfur; -: not detected -
[1] 黄传峰, 韩磊, 王孟艳, 李慧慧, 杨帆, 王永娟, 李大鹏, 王明峰, 霍鹏举, 王坚强.煤加氢液化残渣的性质及应用研究进展[J].现代化工, 2016, 36(6):19-23. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=xdhg201606005HUANG Chuan-feng, HAN Lei, WANG Meng-yan, LI Hui-hui, YANG Fan, WANG Yong-juan, WANG Ming-feng, LI Da-peng, HUO Peng-ju, WANG Jian-qiang. Research development of properties and application of coal hydrogenation liquefaction residue[J]. Mod Chem Ind, 2016, 36(6):19-23. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=xdhg201606005 [2] 国家能源局.国家能源局关于印发《煤炭深加工产业示范"十三五"规划》的通知[Z].中国: 国家能源局, 2017. [3] LUO W J, LAN X Z, SONG Y H, FU J P. Research progress on utilization of coal liquefaction residue[J]. Mater Rev, 2013, 27(6A):153-157. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cldb201311029 [4] 许邦, 初茉, 张慧慧, 王芳, 刘立新.煤直接液化残渣热解研究现状[J].洁净煤技术, 2013, 19(4):81-84. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jjmjs201304020XU Bang, CHU Mo, ZHANG Hui-hui, WANG Fang, LIU Li-xin. Research status of direct coal liquefaction residue pyrolysis[J]. Clean Coal Technol, 2013, 19(4):81-84. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jjmjs201304020 [5] 王忠臣, 戴鑫, 魏江涛.煤加氢液化残渣利用研究进展[J].煤炭加工与综合利用, 2019, (10):44-48. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=mtjgyzhly201910014WANG Zhong-chen, DAI Xin, WEI Jiang-tao. Research progress on utilization of coal hydroliquefaction residue[J]. Coal Process Compr Util, 2019, (10):44-48. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=mtjgyzhly201910014 [6] 常卫科, 徐洁, 孙伟, 陈胜利, 张胜振.煤液化残渣中硫的迁移和转化研究现状及展望[J].洁净煤技术, 2017, 23(3):1-6, 15. http://www.cqvip.com/QK/98393X/201703/672269956.htmlCHANG Wei-ke, XU Jie, SUN Wei, CHEN Sheng-li, ZHANG Sheng-zhen.Research progress and prospect of sulfur migration and transformation in coal liquefaction residue[J]. Clean Coal Technol, 2017, 23(3):1-6, 15. http://www.cqvip.com/QK/98393X/201703/672269956.html [7] 史士东.煤加氢液化工程学基础[M].北京:化学工业出版社, 2012.SHI Shi-dong. Fundamentals of Coal Hydrogenation and Liquefaction Engineering[M]. Beijing:Chemical Industry Press, 2012. [8] 张德祥, RILEY J T.煤中形态硫分析及脱除能力研究[J].燃料化学学报, 1996, 24(2):150-154. http://www.cnki.com.cn/Article/CJFDTotal-RLHX602.009.htmZHANG De-xiang, RILEY J T. Investigations into analysis and desulfurization reactivity of sulfur forms in coals[J]. J Fuel Chem Technol, 1996, 24(2):150-154. http://www.cnki.com.cn/Article/CJFDTotal-RLHX602.009.htm [9] 李文, 郭树才.煤的超临界醇萃取脱硫:Ⅲ.形态硫的变化[J].燃料化学学报, 1995, 23(1):94-98. http://www.cqvip.com/Main/Detail.aspx?id=1853602LI Wen, GUO Shu-cai. Supercritical desulfurization of coal with alcohols:Ⅲ. Variation of sulfur forms[J]. J Fuel Chem Technol, 1995, 23(1):94-98. http://www.cqvip.com/Main/Detail.aspx?id=1853602 [10] 许俊丽.煤直接液化残渣与低阶煤共热解特性及交互作用机制[D].太原: 中国科学院山西煤炭化学研究所, 2018.XU Jun-li. Co-pyrolysis characteristics of direct coal liquefaction residue with low-rank coals and their interactive mechanism[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2018. [11] 赵丽红, 楚希杰.催化剂对煤直接液化残渣气化反应性的影响[J].煤炭科学技术, 2011, 39(9):125-128. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=mtkxjs201109034ZHAO Li-hong, CHU Xi-jie. Catalyst affected to residue gasification reactivity of direct coal liquefaction[J]. Coal Sci Technol, 2011, 39(9):125-128. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=mtkxjs201109034 [12] 崔洪.煤液化残渣的物化性质及其反应性研究[D].太原: 中国科学院山西煤炭化学研究所, 2001. http://d.wanfangdata.com.cn/thesis/Y390418CUI Hong. Property and reactivity of residue from coal hydroliquefaction[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2001. http://d.wanfangdata.com.cn/thesis/Y390418 [13] YAN J C, BAI Z Q, ZHAO H L, BAI J, LI W. Inappropriateness of the standard method in sulfur form analysis of char from coal pyrolysis[J]. Energy Fuels, 2012, 26(9):5837-5842. doi: 10.1021/ef301041p [14] 张文凯.液化残渣及四组分中硫热迁移特征研究[D].西安: 西北大学, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10697-1015326686.htmZHANG Wen-kai. Study on the characteristics of sulfur thermal migration in liquefaction residue and four components[D]. Xi'an: Northwest University, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10697-1015326686.htm [15] 刘亚楠.煤液化残渣及含硫模型物中硫热转化迁移特征研究[D].西安: 西北大学, 2014. http://cdmd.cnki.com.cn/article/cdmd-10697-1014254126.htmLIU Ya-nan. Study on the characteristics of sulfur thermal conversion and migration in coal liquefaction residue and sulfur-containing model[D]. Xi'an: Northwest University, 2014. http://cdmd.cnki.com.cn/article/cdmd-10697-1014254126.htm [16] 杨蕾.煤液化残渣热转化行为及硫的热迁移特征研究[D].西安: 西北大学, 2013. http://cdmd.cnki.com.cn/Article/CDMD-10697-1013254611.htmYANG Lei. Study on thermal conversion behavior of coal liquefaction residue and thermal migration characteristics of sulfur[D]. Xi'an: Northwest University, 2013. http://cdmd.cnki.com.cn/Article/CDMD-10697-1013254611.htm [17] CHU X J, LI W, LI B Q, CHEN H K. Sulfur transfers from pyrolysis and gasification of direct liquefaction residue of Shenhua coal[J]. Fuel, 2008, 87(2):211-215. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=21b40b449984f6d6fb04f0373f38edca [18] GRYGLEWICZ G, JASIENKO S. The behavior of sulfur forms during pyrolysis of low-rank coal[J]. Fuel, 1992, 71(11):1225-1229. doi: 10.1016/0016-2361(92)90047-R [19] FURFARI S, CYPRES R. Hydropyrolysis of a high-sulfur, high-calcite Italian Sulcis coal 2. Importance of the mineral matter on the sulfur behavior[J]. Fuel, 1982, 61(5):453-459. doi: 10.1016/0016-2361(82)90071-0 [20] 楚希杰.神华煤直接液化残渣热解和气化反应性基础研究[D].太原: 中国科学院山西煤炭化学研究所, 2008. http://d.wanfangdata.com.cn/thesis/Y1613565CHU Xi-jie. Pyrolysis and gasification characteristic of Shenhua coal direct liquefaction residue[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2008. http://d.wanfangdata.com.cn/thesis/Y1613565 [21] 冯智皓, 许俊丽, 郝盼, 侯冉冉, 郭振兴, 白进, 白宗庆, 李文.哈密煤温和液化固体产物的理化性质及热解特性[J].燃料化学学报, 2018, 46(10):12-19. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb201810001FENG Zhi-hao, XU Jun-li, HAO Pan, HOU Ran-ran, GUO Zhen-xing, BAI Jin, BAI Zong-qing, LI Wen. Physicochemical properties and pyrolysis characteristics of mild liquefaction solid product of Hami coal[J]. J Fuel Chem Technol, 2018, 46(10):12-19. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb201810001 [22] FENG Z H, BAI Z Q, ZHENG H Y, GUO Z X, KONG L X, BAI J, LI W. Towards understanding the interactions between mild liquefaction solid product and Hami sub-bituminous coal during their co-pyrolysis[J]. J Anal Appl Pyrolysis, 2020, 145:104742. doi: 10.1016/j.jaap.2019.104742 [23] FENG Z H, BAI Z Q, ZHENG H Y, ZHENG K W, HOU R R, GUO Z X, KONG L X, BAI J, LI W. Study on the pyrolysis characteristic of mild liquefaction solid product of Hami coal and CO2 gasification of its char[J]. Fuel, 2019, 253:1034-1041. doi: 10.1016/j.fuel.2019.05.084 [24] SONEDA Y, MAKINO M, YASUDA H, YAMADA O, KOBAYASHI M, KAIHO M. The effect of acid treatment of coal on H2S evolution during pyrolysis in hydrogen[J]. Fuel, 1998, 77(9/10):907-911. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=fd646b2ef075a9f0dbc857c6fe67caf0 [25] CALKINS, WILLIAM H. Investigation of organic sulfur-containing structures in coal by flash pyrolysis experiments[J]. Energy Fuels, 1987, 1(1):59-64. doi: 10.1021/ef00001a011 [26] KHAN M R. Prediction of sulfur distribution in products during low-temperature coal pyrolysis and gasification[J]. Fuel, 1989, 68(11):1439-1449. doi: 10.1016/0016-2361(89)90043-4 [27] 闫金定.炭载含硫化合物热解行为的研究[D].太原: 中国科学院山西煤炭化学研究所, 2005. http://d.wanfangdata.com.cn/thesis/Y764961YAN Jin-ding. Sulfur transformation during pyrolysis of sulfur model compounds under coal-like environment[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2005. http://d.wanfangdata.com.cn/thesis/Y764961 [28] 丁康乐, 李术元, 岳长涛, 钟宁宁.原油中有机硫化物成因的硫酸盐热化学还原反应模拟研究[J].燃料化学学报, 2008, 36(1):48-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb200801009DING Kang-le, LI Shu-yuan, YUE Chang-tao, ZHONG Ning-ning. A simulation on the formation of organic sulfur compounds in petroleum from thermochemical sulfate reduction[J]. J Fuel Chem Technol, 2008, 36(1):48-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb200801009 [29] ATTAR A. Chemistry, thermodynamics and kinetics of reactions of sulphur in coal-gas reactions:A review[J]. Fuel, 1978, 57(4):201-212. http://www.sciencedirect.com/science/article/pii/0016236178901175 [30] TOUCHY A S, SIDDIKI S M A H, ONODERA W, KONA K, SHIMIZU K. Hydrodeoxygenation of sulfoxides to sulfides by a Pt and MoOx co-loaded TiO2 catalyst[J]. Green Chem, 2016, 18(8):2554-2560. doi: 10.1039/C5GC02806J [31] MITSUDOME T, TAKAHASHI Y, MIZUGAKI T, JITSUKAWA K, KANEDA K. Hydrogenation of sulfoxides to sulfides under mild conditions using ruthenium nanoparticle catalysts[J]. Angew Chem Int Ed, 2014, 53(32):8348-8351. doi: 10.1002/anie.201403425 [32] DU B, ZHANG W. Computational study on the mechanism and kinetics for reaction of CH3SH+H with water vapor[J]. Comput Theor Chem, 2017:S2210271X17303730. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=e1bb9d5b314bb1a20fd32ba94ffe2be8 [33] 熊南安, 董滨, 戴晓虎.反向高效液相色谱法检测污泥中含硫氨基酸[J].中国环境科学, 2017, 37(12):4614-4619. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zghjkx201712026XIONG Nan-an, DONG Bin, DAI Xiao-Hu. Determination of sulfur amino acids in sludge by RP-HPLC[J]. China Environ Sci, 2017, 37(12):4614-4619. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zghjkx201712026 [34] IULIA V, ARABELA E U, TEODOR M, MARGARETA O, TATIANA D P, MARIA S, ILIE V. Development and validation of an RP-HPLC method for methionine, cystine and lysine separation and determination in corn samples[J]. Revista de Chimie, 2013, 64(7):673-679. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a2bb1bf6b360f28745a54f7b8800e2eb [35] WANG Y, KANG X J, GE W H, SUN X Z, PENG J. Simple, rapid, and accurate RP-HPLC method for determination of cystine in human urine after derivatization with dansyl chloride[J]. Chromatographia, 2007, 65(9/10):527-532. doi: 10.1365/s10337-007-0210-1 [36] BARKER J, ANDO D J. Mass Spectrometry:Analytical Chemistry by Open Learning, 2nd Edition[M]. Hoboken:John Wiley, 1996. -





 下载:
下载: