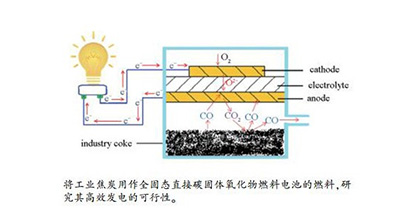
-
摘要: 直接碳固体氧化物燃料电池(DC-SOFC)是一种潜在的固体碳燃料高效率、低污染发电技术。本研究报道了将工业焦炭直接用作管式DC-SOFC燃料的研究。制备了电极材料为Ag-GDC(钆掺杂氧化铈)的YSZ(钇稳定化氧化锆)电解质支撑型管式固体氧化物燃料电池(SOFC)。采用拉曼光谱、扫描电镜和X射线能谱仪对焦炭燃料进行了性质表征。结果表明,焦炭燃料呈微米级的颗粒状,并含有大量对Boudouard反应有利的缺陷结构。电池以纯焦炭为燃料在850℃取得的最大功率密度为149mW/cm2,在碳燃料表面负载能提高Boudouard反应速率的Fe催化剂后,最大功率密度提高至217mW/cm2。通过电化学测试和尾气表征,分析了恒电流放电过程中电池的性能衰减机制。测试结果证明了将焦炭直接用作全固态DC-SOFC的燃料产生电能的可行性。Abstract: Direct carbon solid oxide fuel cell (DC-SOFC) is a potential technology for generating electricity from solid carbon fuel with high conversion efficiency and low pollution. In this study, the use of industrial coke as a fuel for a direct carbon solid oxide fuel cell (DC-SOFC) was investigated. Tubular yttrium-stabilized zirconia (YSZ) electrolyte-supported solid oxide fuel cells (SOFCs) with a cermet of silver and gadolinium-doped ceria (Ag-GDC) as electrode material were fabricated. Raman spectroscopy, scanning electron microscopy and energy dispersive X-ray spectroscopy were applied to characterize the investigated coke fuels. It was observed that the coke fuel wa micron-sized particles with many structural defects, which favored the Boudouard reaction occurring in a DC-SOFC. A peak power density of 149 mW/cm2 at 850 ℃ was observed for pure coke fuel, and it improved to 217 mW/cm2 when a Fe-based catalyst was added to enhance the Boudouard reaction. The degradation performance of the DC-SOFC during a discharging test was analyzed according to the electrochemical characterization and emitted gas measurements. The performed test supported the feasibility of using coke as fuel in an all-solid-state DC-SOFC to generate electricity.
-
Key words:
- solid oxide fuel cell /
- direct carbon /
- coke /
- iron catalyst
-
Table 1 Performance comparison of the presented DC-SOFCs with reported DC-SOFCs operated at 850 ℃
Cell configuration Anode electrode Carbon fuel Pmax /(mW·cm-2) at 850 ℃ Reference Anode-supported Ni-ScSZ carbon black 104 [10] Anode-supported Ni-YSZ coal char 100 [19] Electrolyte-supported Ag-GDC Fe-loaded activated carbon 297 [23] Electrolyte-supported Ag-GDC coke 149 this work Electrolyte-supported Ag-GDC Fe-loaded coke 217 this work -
[1] ZHAO Y, WANG S, DUAN L, LEI Y, CAO P, HAO J. Primary air pollutant emissions of coal-fired power plants in China:Current status and future prediction[J]. Atmos Environ, 2008, 42(36):8442-8452. doi: 10.1016/j.atmosenv.2008.08.021 [2] CAO D, SUN Y, WANG G. Direct carbon fuel cell:Fundamentals and recent developments[J]. J Power Sources, 2007, 167(2):250-257. doi: 10.1016/j.jpowsour.2007.02.034 [3] GIDDEY S, BADWAL S P S, KULKARNI A, MUNNINGS C. A comprehensive review of direct carbon fuel cell technology[J]. Prog Energy Combust, 2012, 38(3):360-399. doi: 10.1016/j.pecs.2012.01.003 [4] GVR T M. Critical review of carbon conversion in "carbon fuel cells"[J]. Chem Rev, 2013, 113(8):6179-6206. doi: 10.1021/cr400072b [5] LIU J, ZHOU M Y, ZHANG Y P, LIU Z J, XIE Y M, CAI W Z, YU F Y, ZHOU Q, WANG X Q, NI M, LIU M L. Electrochemical oxidation of carbon at high temperature:Principles and applications[J]. Energy Fuels, 2017, 32(4):4107-4117. http://d.old.wanfangdata.com.cn/OAPaper/oai_doaj-articles_91595f011edcc3ebcbbdfdebefb4b875 [6] NAKAGAWA N, ISHIDA M. Performance of an internal direct-oxidation carbon fuel cell and its evaluation by graphic exergy analysis[J]. Ind Eng Chem Res, 1988, 27(7):1181-1185. doi: 10.1021/ie00079a016 [7] XIE Y M, TANG Y B, LIU J. A verification of the reaction mechanism of direct carbon solid oxide fuel cells[J]. J Solid State Electr, 2013, 17(1):121-127. doi: 10.1007/s10008-012-1866-5 [8] TANG Y B, LIU J. Fueling solid oxide fuel cells with activated carbon[J]. Acta Phys Chim Sin, 2010, 26(5):1191-1194. http://en.cnki.com.cn/Article_en/CJFDTotal-WLHX201005003.htm [9] TANG Y B, LIU J. Effect of anode and boudouard reaction catalysts on the performance of direct carbon solid oxide fuel cells[J]. Int J Hydrogen Energy, 2010, 35(20):11188-11193. doi: 10.1016/j.ijhydene.2010.07.068 [10] LIU R Z, ZHAO C H, LI J L, ZENG F R, WANG S R, WEN T L, WEN Z Y. A novel direct carbon fuel cell by approach of tubular solid oxide fuel cells[J]. J Power Sources, 2010, 195(2):480-482. doi: 10.1016/j.jpowsour.2009.07.032 [11] WU Y Z, SU C, ZHANG C M, RAN R, SHAO Z P. A new carbon fuel cell with high power output by integrating with in situ catalytic reverse Boudouard reaction[J]. Electrochem Commun, 2009, 11(6):1265-1268. doi: 10.1016/j.elecom.2009.04.016 [12] BAI Y H, LIU Y, TANG Y B, XIE Y M, LIU J. Direct carbon solid oxide fuel cell-a potential high performance battery[J]. Int J Hydrogen Energy, 2011, 36(15):9189-9194. doi: 10.1016/j.ijhydene.2011.04.171 [13] YU F Y, ZHANG Y P, YU L, CAI W Z, YUAN L L, LIU J, LIU M L. All-solid-state direct carbon fuel cells with thin yttrium-stabilized-zirconia electrolyte supported on nickel and iron bimetal-based anodes[J]. Int J Hydrogen Energy, 2016, 41(21):9048-9058. doi: 10.1016/j.ijhydene.2016.04.063 [14] CAI W Z, ZHOU Q, XIE Y M, LIU J, LONG G Q, CHENG S, LIU M L. A direct carbon solid oxide fuel cell operated on a plant derived biofuel with natural catalyst[J]. Appl Energy, 2016, 179:1232-1241. doi: 10.1016/j.apenergy.2016.07.068 [15] ZHOU Q, CAI W Z, ZHANG Y P, LIU J, YUAN L L, YU F Y, WANG X Q, LIU M L. Electricity generation from corn cob char though a direct carbon solid oxide fuel cell[J]. Biomass Bioenergy, 2016, 91:250-258. doi: 10.1016/j.biombioe.2016.05.036 [16] RADY A C, GIDDEY S, KULKARNI A, BADWAL S P S, BHATTACHARYA S. Direct carbon fuel cell operation on brown coal with a Ni-GDC-YSZ anode[J]. Electrochim Acta, 2015, 178:721-731. doi: 10.1016/j.electacta.2015.08.064 [17] JIAO Y, ZHAO J H, AN W T, ZHANG L Q, SHA Y J, YANG G M, SHAO Z P, ZHU Z P, LI S D. Structurally modified coal char as a fuel for solid oxide-based carbon fuel cells with improved performance[J]. J Power Sources, 2015, 288:106-114. doi: 10.1016/j.jpowsour.2015.04.121 [18] XU K, CHEN C, LIU H, TIAN Y, LI X, YAO H. Effect of coal based pyrolysis gases on the performance of solid oxide direct carbon fuel cells[J]. Int J Hydrogen Energy, 2014, 39:17845-17851 doi: 10.1016/j.ijhydene.2014.08.133 [19] JIAO Y, TIAN W J, CHEN H L, SHI H G, YANG B B, LI C, SHAO Z P, ZHU Z P, LI S D. In situ catalyzed boudouard reaction of coal char for solid oxide-based carbon fuel cells with improved performance[J]. Appl Energy, 2015, 141:200-208. doi: 10.1016/j.apenergy.2014.12.048 [20] XIE Y M, CAI W Z, XIAO J, TANG Y B, LIU J, LIU M L. Electrochemical gas-electricity cogeneration through direct carbon solid oxide fuel cells[J]. J Power Sources, 2015, 277:1-8. doi: 10.1016/j.jpowsour.2014.12.016 [21] GUZMAN F, SINGH R, CHUANG S S C. Direct use of sulfur-containing coke on a Ni-yttria-stabilized zirconia anode solid oxide fuel cell[J]. Energy Fuels, 2011, 25(5):2179-2186. doi: 10.1021/ef1016363 [22] XIE Y M, WANG X Q, LIU J, YU C L. Fabrication and performance of tubular electrolyte-supporting direct carbon solid oxide fuel cell by dip coating technique[J]. Acta Phys Chim Sin, 2017, 33(2):386-392. doi: 10.3866/PKU.WHXB201610104 [23] LIU J, SU W H, LÜ Z, JI Y, PEI L, LIU W, HE T M. A rapid sealing method for solid oxide fuel cell using metal conductive adhesive: CN, 02133049.2[P]. 2002-09-25. [24] ZHANG L, XIAO J, XIE Y M, TANG Y B, LIU J, LIU M L. Behavior of strontium-and magnesium-doped gallate electrolyte in direct carbon solid oxide fuel cells[J]. J Alloy Compound, 2014, 608:272-277. doi: 10.1016/j.jallcom.2014.04.154 [25] CAI W Z, LIU J, XIE Y M, XIAO J, LIU M L. An investigation on the kinetics of direct carbon solid oxide fuel cells[J]. J Solid State Electr, 2016, 20(8):2207-2216. doi: 10.1007/s10008-016-3216-5 -





 下载:
下载: