NiO@SiO2 core-shell catalyst for low-temperature methanation of syngas in slurry reactor
-
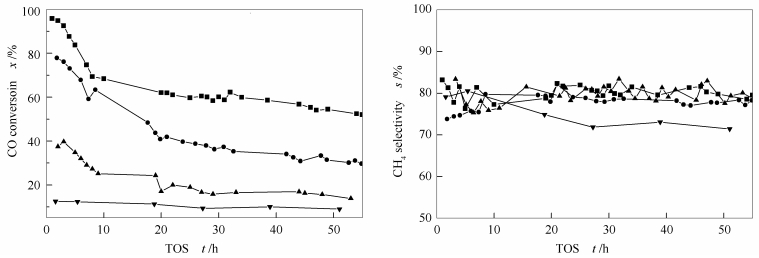
摘要: 采用改进的Stöber方法, 可控制备出具有不同形貌的NiO@SiO2核壳结构催化剂, 并在浆态床反应器(320 ℃) 上, 对其合成气低温甲烷化性能进行评价; 同时借助XRD、TEM、XPS和N2物理吸附等方法对反应前后催化剂的物化性质进行了表征。研究表明, 实验制备的催化剂形貌规整、粒径均匀, 且具有较好的热稳定性。在相同的制备条件下, 核颗粒粒径增大, 其SiO2壳层的厚度随之增加。在反应过程中, 部分催化剂的核壳结构遭到破坏并出现SiO2空壳, 是CO与壳层内的Ni作用生成易迁移的Ni羰基化物种(Ni (CO)x) 所致。催化剂的甲烷化活性随着核颗粒粒径的增加呈现下降趋势; 在不同的反应阶段, 催化剂的失活速率存在明显差异, 在反应的前20 h内, 催化剂出现快速失活, 20 h后失活缓慢, 但是催化剂的甲烷选择性都保持在80%左右。催化剂的失活, 一方面, 是因为反应过程中, Ni核颗粒发生了长大; 另一方面, 是由于壳层中3-5 nm的介孔的减少以及催化剂比表面积、孔容的下降。Abstract: A series of NiO@SiO2 core-shell catalysts were prepared using modified Stöber-method. Their catalytic performances in methanation of syngas were investigated in slurry reactor at 320 ℃. The catalysts before and after reaction were characterized by XRD, TEM, XPS, N2-physisorption, etc. It was found that the NiO@SiO2 core-shell samples had well-shape morphologies and relatively uniform scale. The catalyst test revealed that the methanation activity of these catalysts decreased dramatically with increase of core particle size. The three catalysts with distinct size of core and shell showed remarkably rapid deactivation in the initial period of 20 h and then deactivated slowly during the following reaction, while their CH4 selectivity maintained at about 80%. Void-shell was formed during the reaction probably because easy-migrated Ni (CO)x species were generated. Apparently, it was concluded that increase of core particle size, decrease of BET surface area and pore volume, and abatement of mesopores within 3-5 nm in the shell were responsible for the deactivation of these core-shell catalysts based on the characterization of catalysts.
-
Key words:
- core-shell /
- nickel-based catalyst /
- slurry reactor /
- low-temperature methanation
-
表 1 NiO颗粒粒径及反应前后NiO@SiO2的核壳尺寸
Table 1 Particle size of NiO, core size and shell thickness of NiO@SiO2
Sample Core d/nm Shell d/nm NiO-350 4.9 - NiO-350@SiO2 5.1 6.1 Used NiO-350@SiO2 8.1 5.0 NiO-400 8.1 - NiO-400@SiO2 8.6 11.2 Used NiO-400@SiO2 10.6 10.1 NiO-500 23.7 - NiO-500@SiO2 23.3 23.0 Used NiO-500@SiO2 27.8 20.6 表 2 反应前后催化剂的表面原子浓度及体相Ni含量
Table 2 Surface atom concentration and Ni content of catalyst before and after reaction
Sample Surface atomic concentrationsa/% Ni/Si
(atomic ratio)Nib/% Ni Si O NiO-350@SiO2 10.11 17.77 72.12 0.57 57.32 Used NiO-350@SiO2 9.85 17.12 73.03 0.58 55.92 NiO-400@SiO2 8.50 24.36 67.14 0.35 58.98 Used NiO-400@SiO2 5.09 27.73 67.18 0.18 61.21 NiO-500@SiO2 6.46 21.22 72.32 0.30 60.68 Used NiO-500@SiO2 4.61 28.38 67.01 0.16 63.14 Theoretical valuec 33.50 11.00 55.50 3.05 62.16 a: the surface atomic concentrations of catalysts were calculated by XPS; b: Ni bulk concentration of catalysts obtained from ICP c: based on the addition mass when catalyst is prepared 表 3 反应前后催化剂的N2吸附
Table 3 N2 sorption of NiO@SiO2 before and after reaction
Sample BET surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Pore diameter d/nm NiO-350@SiO2 117.2 0.218 7.4 Used NiO-350@SiO2 52.7 0.147 11.2 NiO-400@SiO2 82.9 0.197 8.2 Used NiO-400@SiO2 43.5 0.101 9.3 NiO-500@SiO2 34.4 0.074 8.6 Used NiO-500@SiO2 23.7 0.073 12.3 -
[1] 张俊峰, 白云星, 张清德, 解红娟, 谭猗生, 韩怡卓. Zr改性Ni/γ-Al2O3催化剂用于浆态相合成气的低温甲烷化[J].燃料化学学报, 2013, 41(8): 966-971. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18237.shtmlZHANG Jun-feng, BAI Yun-xing, ZHANG Qing-de, XIE Hong-juan, TAN Yi-sheng, HAN Yi-zhuo. Low temperature methanation of syngas in a slurry reactor over Zr-doped Ni/γ-Al2O3 catalyst[J]. J Fuel Chem Technol, 2013, 41(8): 966-971. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18237.shtml [2] 孟凡会, 常慧蓉, 李忠. Ni-Mn/Al2O3催化剂在浆态床中CO甲烷化催化性能[J].化工学报, 2014, 65(8): 2997-3003.MENG Fan-hui, CHANG Hui-rong, LI Zhong. Catalytic performance of Ni-Mn/Al2O3 catalyst for CO methanation in slurry-bed reactor[J]. CIESC J, 2014, 65(8): 2997-3003. [3] 孟凡会, 刘军, 李忠, 钟朋展, 郑华艳. Ce含量对Ni-Ce/Al2O3催化剂结构及浆态床CO甲烷化性能的影响[J].燃料化学学报, 2014, 42(2): 231-237. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18360.shtmlMENG Fan-hui, LIU Jun, LI Zhong, ZHONG Peng-zhan, ZHENG Hua-yan. Effect of Ce content of Ni-Ce/Al2O3 catalyst constructure and CO methanation in slurry-bed reactor[J]. J Fuel Chem Technol, 2014, 42(2): 231-237. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18360.shtml [4] ZHANG J F, BAI Y X, ZHANG Q D, WANG X X, ZHANG T, TAN Y S, HAN Y Z. Low-temperature methanation of syngas in slurry phase over Zr-doped Ni/γ-Al2O3 catalysts prepared using different methods[J]. Fuel, 2014, 132: 211-218. doi: 10.1016/j.fuel.2014.04.085 [5] 黄国宝, 王志青, 李庆峰, 黄戒介, 房倚天.液相中镍催化剂催化合成气甲烷化的初步研究[J].燃料化学学报, 2014, 42(8): 952-957. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18470.shtmlHUANG Guo-bao, WANG Zhi-qing, LI Qing-feng, HUANG Jie-jie, FANG Yi-tian. Syngas methanation over nickel catalyst in liquid-phase[J]. J Fuel Chem Technol, 2014, 42(8): 952-957. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18470.shtml [6] 贺龙.浆态床镍基甲烷化催化剂的研究[D].北京:中国矿业大学(北京), 2012.HE Long. Research of nickel based catalyst for methanation of synthetic gas in slurry bed reactor[D]. Beijing: China University of Mining and Technology (Beijing), 2012. [7] 马胜利.煤基合成气低温甲烷化镍基催化剂的研究[D].太原:中国科学院山西煤炭化学研究所, 2011.MA Sheng-li. Study on low temperature methanation for coal-derived syngas over Ni-based catalyst[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2011. [8] GRAF C, VOSSEN D L J, IMHOF A, VAN BLAADEREN A. A general method to coat colloidal particles with silica[J]. Langmuir, 2003, 19(17): 6693-6700. doi: 10.1021/la0347859 [9] YAO L H, LI Y X, ZHAO J, JI W J, AU C T. Core-shell structured nanoparticles (M@SiO2, Al2O3, MgO; M=Fe, Co, Ni, Ru) and their application in COx-free H2 production via NH3 decomposition[J]. Catal Today, 2010, 158(3/4): 401-408. http://www.sciencedirect.com/science/article/pii/S0920586110003251 [10] LI Z W, MO L Y, KATHIRASER Y, KAWI S. Yolk-Satellite-Shell Structured Ni-Yolk@Ni@SiO2 Nanocomposite: Superb Catalyst toward Methane CO2 Reforming Reaction[J]. ACS Catal, 2015, 4(5): 1526-1536. doi: 10.1021/cs401027p [11] LI Z W, KATHIRASER Y, KAWI S. Facile synthesis of high surface area yolk-shell Ni@Ni embedded SiO2 via Ni phyllosilicate with enhanced performance for CO2 reforming of CH4[J]. ChemCatChem, 2014, 7: 160-168. doi: 10.1002/cctc.201402673/full [12] LI L, YAO Y, SUN B, FEI Z Y, XIA H, ZHAO J, JI W J, AU C T. Highly active and stable lanthanum-doped core-shell-structured Ni@SiO2 catalysts for the partial oxidation of methane to syngas[J]. ChemCatChem, 2013, 5(12): 3781-3787. doi: 10.1002/cctc.201300537 [13] LI L, HE S C, SONG Y Y, ZHAO J, JI W J, AU C T. Fine-tunable Ni@porous silica core-shell nanocatalysts: Synthesis, characterization, and catalytic properties in partial oxidation of methane to syngas[J]. J Catal, 2012, 288: 54-64. doi: 10.1016/j.jcat.2012.01.004 [14] 李雷.核壳结构纳米催化剂的设计与制备及甲烷选择性氧化制合成气研究[D].南京:南京大学, 2012.LI Lei. Core-shell structured nanocatalysts: design, synthesize and their application in partial oxidation of methane to syngas[D]. Nanjing: Nanjing University, 2012. [15] TAKENAKA S, UMEBAYASHI H, TANABE E, MATSUNE H, KISHIDA M. Specific performance of silica-coated Ni catalysts for the partial oxidation of methane to synthesis gas[J]. J Catal, 2007, 245(2): 392-400. doi: 10.1016/j.jcat.2006.11.005 [16] ZHANG L F, LI M, REN T Z, LIU X Y, YUAN Z Y. Ce-modified Ni nanoparticles encapsulated in SiO2 for COx-free hydrogen production via ammonia decomposition[J]. Int J Hydrogen Energ, 2015, 40(6): 2648-2656. doi: 10.1016/j.ijhydene.2014.12.079 [17] 张明伟.核壳结构铁基催化剂的制备及在CO加氢中的应用[D].太原:中国科学院大学山西煤炭化学研究所, 2014.ZHANG Ming-wei. Preparation of core-shell iron-based catalysts and their application for CO hydrogenation[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2011. [18] ZHANG M W, FANG K G, LIN M G, HOU B, ZHONG L S, ZHU Y, WEI W, SUN Y H. Controlled Fabrication of Iron Oxide/Mesoporous Silica Core-Shell Nanostructures[J]. J Phys Chem C, 2013, 117: 21529-21538. doi: 10.1021/jp4049583 [19] XIE R Y, LI D B, HOU B, WANG J G, JIA L T, SUN Y H. Solvothermally derived Co3O4@m-SiO2 nanocomposites for Fischer-Tropschsynthesis[J]. Catal Commun, 2011, 12(5): 380-383. doi: 10.1016/j.catcom.2010.10.010 [20] XIE R Y, WANG H, GAO P, XIA L, ZHANG Z Z, ZHAO T J, SUN Y H. Core@shell Co3O4@C-m-SiO2 catalysts with inert C modified mesoporous channel for desired middle distillate[J]. Appl Catal A: Gen, 2015, 492: 93-99. doi: 10.1016/j.apcata.2014.12.023 [21] LI Y R, LU G X, MA J T. Highly active and stable nano NiO-MgO catalyst encapsulated by silica with a core-shell structurefor CO2 methanation[J]. RSC Adv, 2014, 4: 17420-17428. doi: 10.1039/c3ra46569a [22] MUNNIK P, VELTHOEN M E Z, DE JONGH P E, DE JONG K P, GOMMES C J. Nanoparticle growth in supported nickel catalysts during methanation reaction-larger is better[J]. Angew Chem, Int Ed, 2014, 53(36): 9493-9497. doi: 10.1002/anie.201404103 [23] ENGBAEK J, LYTKEN O, NIELSEN J H, CHORKENDORFF L. CO dissociation on Ni: The effect of steps and of nickel carbonyl[J]. Surf Sci, 2008, 602(3): 733-743. doi: 10.1016/j.susc.2007.12.008 [24] 曾波.核壳结构钴基催化剂及中间馏分油合成研究[D].太原:中国科学院大学山西煤炭化学研究所, 2014.ZENG Bo. Synthesis of middle distillates via cobalt-based catalysts with core-shell structures[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2014. [25] CHOU K, CHEN C. Fabrication and characterization of silver core and porous silica shell nanocomposite particles[J]. Microporous Mesoporous Mater, 2007, 98: 208-213. doi: 10.1016/j.micromeso.2006.09.006 -





 下载:
下载: