Influence of coal blending on ash fusion characteristics for coal with high ash fusion temperature
-
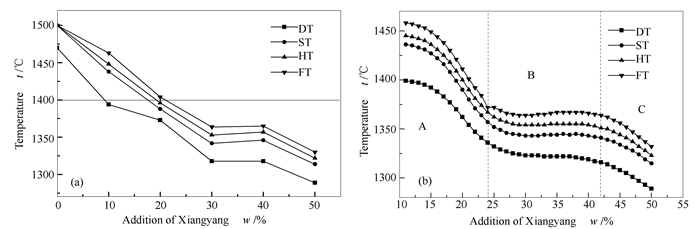
摘要: 采用灰熔点较低的襄阳煤和灰熔点较高的晋城无烟煤组成的混合煤样,利用XRF、SEM、DSC、XRD、三元相图等分析方法,探究了襄阳煤对晋城无烟煤煤灰熔融温度的影响。结果表明,配煤能有效降低高熔点煤灰的熔融温度,当襄阳煤的加入量小于24%时,混合煤灰熔融温度显著降低;襄阳煤的加入量在24%-40%时,混合煤灰熔融温度变化平缓且流动温度低于1 400℃。混合煤灰中的成分在1 000-1 200℃发生一系列的化学反应,主要包括SiO2与Al2O3结合产生高熔点物质莫来石以及Fe2O3、CaO与莫来石反应转化形成铁尖晶石、钙长石等新物质,由此造成了煤灰熔融温度的变化。基于BP神经网络对实验数据建立预测模型,其预测效果优于前人总结的经验公式,平均准确度高于99%。利用热力学软件HSC 5.0分析了CaO、Fe2O3对降低煤灰熔融温度的影响,分析表明,CaO对莫来石的转化作用优于Fe2O3。Abstract: Xiangyang coal with low ash fusion temperature (AFT) and Jincheng coal with high AFT were used to prepare the blending samples. The influence of Xiangyang coal addition on AFT of Jincheng coal was explored by XRF, SEM, DSC, XRD, and ternary phase diagram analysis. The results show that blending coal can reduce the AFT effectively. The AFT of blending coal is lowered significantly when the adding amount of Xiangyang coal is lower than 24%. Whereas, when the adding amount is between 24% and 40%, AFT of the mixed coal has a slight change and the ash flow temperature is below 1 400℃. A series of chemical reactions among ash composition of mixed coal occur at 1 000-1 200℃, mainly including formation of high melting point compound (mullite) from SiO2 with A12O3, and that of low melting point compounds (anorthite and hercynite) from the reactions between mullite and CaO or Fe2O3. The above reactions mainly cause the changes of ash fusion temperature in blending coal. Based on BP neural network, a prediction model of ash fusion temperature was built. It is proved that the prediction average accuracy by BP neural network is higher than 99%, which is better than that of a previous empirical formula. Furthermore, analysis by thermodynamics software (HSC 5.0) shows that mullite prefers to react with CaO rather than Fe2O3.
-
Key words:
- coal blending /
- ash fusion temperature /
- BP neural network
-
图 6 不同含量的襄阳煤在1100℃(a)、1200℃(b)、1350℃(c) 的XRD谱图
Figure 6 XRD pattern of different quantity of Xiangyang coal ash at 1100℃(a), 1200℃ (b) and 1350℃(c)
1: quartz, SiO2; 2: rankinite, Ca3Si2O7; 3: mullite, Al6Si2O13; 4: anorthite, CaAl2Si2O8; 5: sillimanite, Al2SiO5; 6: anhydrite, CaSO4; 7: mayenite, Ca12Al14O33; 8: calcium iron oxide, CaO·Fe3O4; 9: alumina, Al2O3; 10: fayalite, Fe2SiO4; 11: calcium iron oxide, CaO·Fe2O3; 12: hercynite, FeAl2O4; 13: yeelimite, Ca4Al6O12SO4; 14: clinotobermorite, Ca5Si6O17 JC: Jincheng coal; XY: Xiangyang coal
图 7 不同温度下30%襄阳煤 (a) 和20%襄阳煤 (b) 的XRD谱图
Figure 7 XRD patterns of 30% Xiangyang coal ash (a) and 20% Xiangyang coal ash (b) at different temperatures
1: quartz, SiO2; 2: rankinite, Ca3Si2O7; 3: mullite, Al6Si2O13; 4: anorthite, CaAl2Si2O8; 5: sillimanite, Al2SiO5; 6: anhydrite, CaSO4; 7: mayenite, Ca12Al14O33; 8: calcium iron oxide, CaO·Fe3O4; 9: alumina, Al2O3; 10: fayalite, Fe2SiO4; 11: calcium iron oxide, CaO·Fe2O3; 12: hercynite, FeAl2O4; 13: yeelimite, Ca4Al6O12SO4; 14:clinotobermorite, Ca5Si6O17
表 1 晋城无烟煤和襄阳煤的工业分析与元素分析
Table 1 Proximate and ultimate analysis of coal samples
Coal sample Proximate analysis wad/% Ultimate analysis w/% M A V FC C H N S O* Jincheng 1.17 17.40 9.08 72.35 73.31 2.87 1.07 0.79 3.39 Xiangyang 10.06 13.36 34.04 42.54 49.63 3.76 0.70 0.28 22.21 *: by difference 表 2 晋城无烟煤和襄阳煤的煤灰成分分析
Table 2 Ash composition of coal samples
Coal sample Ash composition w/% SiO2 A12O3 Fe2O3 CaO MgO SO3 K2O Na2O TiO2 P2O3 Jincheng 47.00 33.55 7.99 5.16 1.60 2.92 0.38 0.46 0.85 0.01 Xiangyang 31.06 14.32 18.95 28.62 2.98 0.99 2.08 0.20 0.74 0.16 表 3 不同质量的襄阳煤与晋城无烟煤组成的混合煤灰的酸碱比
Table 3 Ratio of acid to alkali in blending ash with addition of Xiangyang coal into Jincheng coal
Xiangyang coal w/% 0 10 20 24 26 28 30 32 34 36 40 50 100 Ratio of acid to alkali (A/B) 5.22 4.32 3.51 3.25 3.15 3.04 2.94 2.82 2.74 2.63 2.47 2.08 0.87 Ratio of acid to alkali (x) 5.52 4.42 3.69 3.41 3.31 3.20 3.08 2.97 2.88 2.76 2.59 2.18 0.91 note: $\begin{align} & \text{A/B=}\frac{\text{Si}{{\text{O}}_{2}}\text{+A}{{\text{l}}_{2}}{{\text{O}}_{3}}\text{+Ti}{{\text{O}}_{2}}}{\text{F}{{\text{e}}_{2}}{{\text{O}}_{3}}\text{+CaO+}{{\text{K}}_{2}}\text{O+MgO+N}{{\text{a}}_{2}}\text{O}};x=\frac{\text{Si}{{\text{O}}_{2}}\text{+A}{{\text{l}}_{2}}{{\text{O}}_{3}}\text{+Ti}{{\text{O}}_{2}}}{\text{F}{{\text{e}}_{2}}{{\text{O}}_{3}}\text{+CaO}+\text{MgO}} \\ & \\ \end{align}$ 表 4 经验公式和BP神经网络的预测准确度
Table 4 Forecast precision of empirical formulas and BP neural network
Prediction models (1) (2) (3) BP K /% 92.68 97.40 95.06 99.12 Kmin/% 91.15 96.26 93.46 98.17 -
[1] 谢克昌.绿色低碳持续发展[J].太原理工大学学报, 2010, 41(6):445-448. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201005000.htmXIE Ke-chang.Sustainable development on green and low-carbon[J].J Taiyuan Univ Sci Technol, 2010, 41(6):445-448. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201005000.htm [2] 李庆峰, 赵霄鹏, 黄戒介, 房倚天, 王洋.灰融聚流化床粉煤气化技术0.6MPa工业炉运行概况[J].化学工程, 2010, 38(10):123-126. http://www.cnki.com.cn/Article/CJFDTOTAL-IMIY201010028.htmLI Qing-feng, ZhAO Xiao-peng, HUANG Jie-jie, FANG Yi-tian, WANG Yang.Introduction to 0.6MPa industrial gasifier of ash agglomerating fluidized-bed coal gasification technology[J].Chem Eng, 2010, 38(10):123-126. http://www.cnki.com.cn/Article/CJFDTOTAL-IMIY201010028.htm [3] 许洁, 刘霞, 李德侠, 周志杰, 王辅臣, 于广锁.煤灰流动温度预测模型的研究[J].燃料化学学报, 2012, 40(12):1415-1420. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18077.shtmlXU Jie, LIU Xia, LI De-xia, ZHOU Zhi-jie, WANG Fu-chen, YU Guang-suo.Predication model for flow temperature of coal ash[J].J Fuel Chem Technol, 2012, 40(12):1415-1420. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18077.shtml [4] 陈玉爽, 张忠孝, 乌晓江, 李洁, 管嵘清, 闫博.配煤对煤灰熔融特性影响的实验与量化研究[J].燃料化学学报, 2009, 37(5):521-526. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17485.shtmlCHEN Yu-shuang, ZHANG Zhong-xiao, WU Xiao-jiang, LI Jie, GUAN Rong-qing, YAN Bo.Experimental study on coal ash fusion characteristics of blend coal[J].J Fuel Chem Technol, 2009, 37(5):521-526. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17485.shtml [5] 张德祥, 龙永华, 高晋生, 郑斌.煤灰中矿物的化学组成与灰熔融性的关系[J].华东理工大学学报, 2003, 29(6):590-594. http://www.cnki.com.cn/Article/CJFDTOTAL-HLDX200306012.htmZHANG De-xiang, LONG Yong-hua, GAO Jin-sheng, ZHENG Bin.Relationship between the coal ash fusibility and its chemical composition[J].J East China Univ Sci Technol, 2003, 29(6):590-594. http://www.cnki.com.cn/Article/CJFDTOTAL-HLDX200306012.htm [6] 刘胜华, 高娜, 郭延红, 杨倩.配煤对煤灰熔融特性的影响[J].煤炭转化, 2014, 37(2):46-49. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH201403011.htmLIU Sheng-hua, GAO Na, GUO Yan-hong, YANG Qian.Influence of coal blending on ash fusion temperature[J].Coal Convers, 2014, 37(2):46-49. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH201403011.htm [7] GENTILE A L, FOSTER W R.Calcium hexaluminate and its stability relations in the system CaO-Al2O3-SiO2[J].J Am Ceram Soc, 1963, 46(2):74-76. doi: 10.1111/jace.1963.46.issue-2 [8] 刘文胜, 赵虹, 杨建国, 翁善勇, 周永刚, 冯国华, 凌柏林, 郑航.三元相图在配煤结渣特性研究中的应用[J].热力发电, 2009, 5(10):5-10.LIU Wen-sheng, ZHAO Hong, YANG Jian-guo, WENG Shan-yong, ZHOU Yong-gang, FENG Guo-hua, LING Bo-lin, ZHENG Hang.Application of three-element phase diagram in study on slagging behavior of blended coal[J].Therm Power Gener, 2009, 5(10):5-10. [9] 戴爱军.煤灰成分对灰熔融性影响研究[J].洁净煤技术, 2007, 13(5):23-29. http://www.cnki.com.cn/Article/CJFDTOTAL-JJMS200705006.htmDAI Ai-jun.Investigation on the influence of ash composition for ash fusion temperature[J].Clean Coal Technol, 2007, 13(5):23-29. http://www.cnki.com.cn/Article/CJFDTOTAL-JJMS200705006.htm [10] KAHRAMAN H, BOS F, REIFENSTEIN A, COIN C D A.Application of a new ash fusion test to Theodore coals[J].Fuel, 1998, 77(10):1005-1011. https://www.researchgate.net/publication/222983828_Application_of_a_new_ash_fusion_test_to_Theodore_coals [11] 邓芙蓉.利用TG-DSC、XRD、SEM等多种手段研究煤灰的熔融特性[D].杭州:浙江大学, 2005.DENG Fu-rong.Investigation on ash fusion characteristics using TG-DSC, XRD and SEM[D].Hangzhou:Zhejiang University, 2005. [12] 徐荣声, 王永刚, 林雄超, 杨萨莎, 艾沙江, 杨远平.配煤和助溶剂降低煤灰熔融温度的矿物学特性研究[J].燃料化学学报, 2015, 43(11):1303-1310. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18723.shtmlXU Rong-sheng, WANG Yong-gang, LIN Xiong-chao, YANG Sa-sha, AI Sha-jiang, YANG Yuan-ping.Mineralogical properties of lowering coal ash melting temperature using blending coal and fluxing agent[J].J Fuel Chem Technol, 2015, 43(11):1303-1310. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18723.shtml [13] 禹立坚, 黄镇宇, 程军, 潘华引, 周俊虎, 岑可发.配煤燃烧过程中煤灰熔融性研究[J].燃料化学学报, 2009, 37(2):139-144. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17417.shtmlYU Li-jian, HUANG Zhen-yu, CHENG Jun, PAN Hua-yin, ZHOU Jun-hu, CEN Ke-fa.Study on the coal ash fusibility during blending coal combustion[J].J Fuel Chem Technol, 2009, 37(2):139-144. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17417.shtml [14] WU X J, ZhANG Z X, PIAO G L, HE X, CHEN Y S, KOBAYASHI N, MORI S, ITAYA Y.Behavior of mineral matters in Chinese coal ash melting during char-CO2/H2O gasification reaction[J].Energy Fuels, 2009, 23(5):2420-2428. doi: 10.1021/ef801002n [15] 丰芸, 李寒旭, 丁立明.利用XRD分析高温下淮南煤灰矿物质变化[J].安徽建筑工业学院学报, 2008, 16(10):53-57. http://www.cnki.com.cn/Article/CJFDTOTAL-AHJG200805015.htmFENG Yun, LI Han-xu, DING Li-ming.Study of huainan coal ash mineral variation under high temperature with XRD[J].J Anhui Inst Archit, 2008, 16(10):53-57. http://www.cnki.com.cn/Article/CJFDTOTAL-AHJG200805015.htm [16] 李风海, 黄戒介, 房倚天, 王洋.晋城无烟煤流化床气化结渣机理的探索[J].太原理工大学学报, 2010, 41(5):666-669. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201005050.htmLI Feng-hai, HUANG Jie-jie, FANG Yi-tian, WANG Yang.Study on fluidized bed gasification slagging mechanism of Jincheng coal[J].J Taiyuan Univ Sci Technol, 2010, 41(5):666-669. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201005050.htm [17] MATJIE R H, LI Z S, WARD C R, FRENCH D.Chemical composition of glass and crystalline phase in coal gasification[J].Fuel, 2008, 87(6):857-869. doi: 10.1016/j.fuel.2007.05.050 [18] REITER F M.How sulfur content of coal Relates to ash fusion charaeteristies[J].Power Eng, 1995, 59(5):98-100. -





 下载:
下载: