Preparation of Ni2P/Zr-MCM-41 catalyst and its performance in the hydrodeoxygenation of Jatropha curcas oil
-
摘要: 采用水热法合成MCM-41和Zr-MCM-41, 由Ni (NO3)2和(NH4)2HPO4溶液共浸渍、高温焙烧、氢气还原和钝化制备了负载型Ni2P/Zr-MCM-41催化剂。采用XRD、TEM、氮气吸附、CO吸附、吡啶吸附红外和XPS等方法对催化剂进行了表征, 并在高压反应釜中研究了其对麻风树油加氢脱氧(HDO) 的催化性能。结果表明, 氢气还原温度为650 ℃、Ni2P负载量为20%(质量分数)、Ni2P物相呈晶型时, Ni2P/Zr-MCM-41催化剂的活性最佳; 较低的Ni2P负载量有利于其在Zr-MCM-41载体表面均匀分散, 而负载量高于25%(质量分数) 时, 活性组分少量团聚, 易导致孔道堵塞。催化剂表面存在部分因钝化而形成的NiO。对于麻风树油加氢脱氧, Ni2P负载量为20%(质量分数) Ni2P/Zr-MCM-41表现出优异的催化性能; 脱氧率高达93.90%, 直链烷烃含量高达85.36%, 其中柴油组分产率较高, C15~20组分占直连烷烃组分50%以上。
-
关键词:
- Ni2P/Zr-MCM-41 /
- 加氢脱氧 /
- 麻风树油 /
Abstract: With hydrothermally synthesized MCM-41 and Zr-MCM-41 as the supports, Ni2P/Zr-MCM-41 catalysts were prepared by co-impregnation with Ni (NO3)2 and (NH4)2HPO4 solution, calcination, reduction with H2 and subsequent passivation. The Ni2P/Zr-MCM-41 catalysts were characterized by XRD, Py-IR, TEM, XPS, N2 physisorption and CO chemisorption; their catalytic performance in the hydrodeoxygenation (HDO) of Jatropha curcas oil to produce the second-generation biodiesel was investigated in an autoclave. The results indicated that Ni2P can be well dispersed on Zr-MCM-41 with a loading of 20% by reduction at 650 ℃ from the phosphate precursors. The Ni2P/Zr-MCM-41 catalyst exhibit excellent performance in the HDO of Jatropha curcas oil. Over the Ni2P/Zr-MCM-41 catalyst with a Ni2P loading of 20%, the HDO conversion achieves 93.90% and the fraction of linear paraffins in the product oil reaches 85.36%, in which diesel fraction (C15~20) may exceed 61.90%; the components of oil generated through HDO are similar to those of fossil diesel.-
Key words:
- Ni2P /
- Zr-MCM-41 /
- hydrodeoxygenation /
- Jatropha curcas oil
-
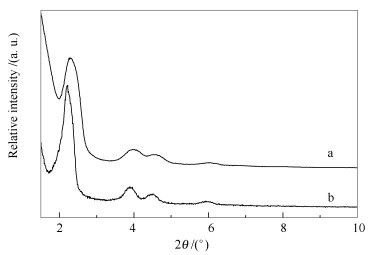
表 1 载体Zr-MCM-41与MCM-41在150和350 ℃下的L酸和B酸值
Table 1 Contents of Lewis and Brönsted acid sites of MCM-41 and Zr-MCM-41 measured by Py-IR at 150 and 350 ℃
Support Acid density at 150 ℃ /(μmol·g-1) Acid density at 350 ℃ /(μmol·g-1) Lewis Brönsted Lewis Brönsted MCM-41 0.012 0 0.004 0 Zr-MCM-41 0.022 0.066 0.005 0.002 表 2 载体与催化剂的孔结构参数
Table 2 Specific surface area, pore diameter, pore volume and CO uptake of various supports and catalysts
Sample ABET /(m2·g-1) vBJH /(cm3·g-1) Pore diameter d/nm CO uptakes v/(cm3·g-1) MCM-41 843.625 0.982 3.892 - Zr-MCM-41 828.091 0.922 3.387 - 10%Ni2P/Zr-MCM-41 647.810 0.607 3.349 0.086 15%Ni2P/Zr-MCM-41 576.164 0.555 3.393 0.116 20%Ni2P/Zr-MCM-41 500.438 0.798 2.843 0.170 25%Ni2P/Zr-MCM-41 379.940 0.573 2.902 0.165 表 3 麻风树油的脂肪酸组成
Table 3 Fatty acid composition of Jatropha curcas oil
Fatty acid component Content w/% C 14:0 myristic acid 0.1 C 16:0 palmitic acid 13.3 C 16:1 hypogaeic acid 1.1 C 18:0 octadecanoic acid 6.7 C 18:1 oleic acid 40.7 C 18:2 linoleic acid 37.6 C 18:3 linolenic acid 0.2 C 20:0 arachic acid 0.2 C 20:1 eicosenoic acid 0.1 表 4 不同负载量催化剂所得生成油组分含量
Table 4 Yields of various oil components obtained over the Ni2P/Zr-MCM-41 catalysts with different Ni2P loadings
Ni2P loading w/% Yield of components w/% linear paraffin oxy-compound isoparaffin cycloparaffin olefin aromatic 10 76.09 14.61 0 4.79 3.35 1.16 15 81.45 8.68 2.30 3.57 2.23 1.77 20 85.36 6.10 0.89 2.36 1.88 3.41 25 82.17 7.10 1.06 5.09 0.29 4.18 表 5 生物柴油中直链烷烃的组分含量
Table 5 Components of linear paraffins in the oil generated from HDO of Jatropha curcas oil over the Ni2P/Zr-MCM-41 catalysts with different Ni2P loadings
Sample Content w% 10% 15% 20% 25% Nonane 2.21 2.48 4.11 6.35 Decane 2.55 0 5.26 7.29 Undecane 3.29 4.00 6.14 8.38 Dodecane 3.60 4.41 7.03 8.62 Tridecane 3.96 4.74 6.89 8.78 Tetradecane 4.13 5.16 6.84 8.41 Pentadecane 11.84 13.67 15.41 16.32 Hexadecane 4.32 5.58 6.62 2.62 Heptadecane 29.45 31.14 32.67 6.43 Octadecane 4.76 2.08 2.44 0 Nonadecane 2.81 2.29 3.06 2.53 Eicosane 1.83 1.40 1.70 1.74 Heneicosane 0.49 3.27 1.02 1.06 Tetracosane 0 0.49 0 0.67 Pentacosane 0.86 0.76 0.81 2.97 -
[1] 王东军, 刘红岩, 刘玉香, 何昌洪, 刘春贵, 张宝军.生物燃料加氢脱氧催化剂的研究进展[J].石油化工, 2012, 41(10): 1214-1219. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHG201210021.htmWANG Dong-jun, LIU Hong-yan, LIU Yu-xiang, HE Chang-hong, LIU Chun-gui, ZHANG Bao-jun. Research progress in catalysts for biofuel hydrodeoxygenation[J]. Petrochem Technol, 2012, 41(10): 1214-1219. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHG201210021.htm [2] 齐泮仑, 张国静, 曹亦农, 董平, 赵光辉.中国生物柴油大规模发展应首先解决的问题[J].化工中间体, 2009, (7): 6-11. http://www.cnki.com.cn/Article/CJFDTOTAL-ZJTY200907001.htmQI Pan-lun, ZHANG Guo-jing, CAO Yin-nong, DONG Ping, ZHAO Guang-hui. Large-scale development of bio-diesel shouldfirst solve the probleminchina[J]. Chem Intermed, 2009, (7): 6-11. http://www.cnki.com.cn/Article/CJFDTOTAL-ZJTY200907001.htm [3] FURIMSKY E. Catalytic hydrodeoxygenation[J]. Appl Catal A: Gen, 2000, 199(2): 147-190. doi: 10.1016/S0926-860X(99)00555-4 [4] 张华涛, 殷福珊.第二代生物柴油的最新研究进展[J].日用化学品科学, 2009, 32(2): 17-20. http://www.cnki.com.cn/Article/CJFDTOTAL-RYHX200902006.htmZHANG Hua-tao, YIN Fu-shan. Advances in research for second generation of biodiesel[J]. Deteg Cosmet, 2009, 32(2): 17-20. http://www.cnki.com.cn/Article/CJFDTOTAL-RYHX200902006.htm [5] TOBA M, ABE Y, KURAMOCHI H, OSAKO M, MOCHIZUKI T, YOSHIMURA Y. Hydrodeoxygenation of waste vegetable oil over sulfidecatalysts[J]. Catal Today, 2011, 164(1): 533-537. doi: 10.1016/j.cattod.2010.11.049 [6] MONNIER J, SULIMMA H, DALAI A, CARAVAGGIO G. Hydrodeoxygenation of oleic acid and canola oil over alumina-supported metal nitrides[J]. Appl Catal A: Gen, 2010, 382(2): 176-180. doi: 10.1016/j.apcata.2010.04.035 [7] HAN J X, DUAN J Z, CHEN P, LOU H, ZHENG X M, HONG H P. Nanostructured molybdenum carbides supported on carbon nanotubes asefficient catalysts for one-step hydrodeoxygenation and isomerization of vegetable oils[J]. Green Chem., 2011, 13(9): 2561-2568. doi: 10.1039/c1gc15421d [8] GARCÍA-DÁVILA J, OCARANZA-SÓNCHEZ E, ROJAS-LÓPEZ M, MUÑOZ-ARROYO J A, RAMÍREZ J, MARTÍNEZ-AYALA A L. Jatropha curcas L.oil hydroconversion over hydrodesulfurization catalysts for biofuel production[J]. Fuel, 2014, 135(11): 380-386. https://www.researchgate.net/profile/Marlon_Rojas-Lopez/publication/264241857_Jatropha_curcas_L._oil_hydroconversion_over_hydrodesulfurization_catalysts_for_biofuel_production/links/5488d9ca0cf2ef344790a568.pdf?origin=publication_list [9] 蔡卫权, 余小锋.高比表面大中孔拟薄水铝石和γ-Al2O3的制备研究[J].化学进展, 2007, 19(9): 1322-1330.CAI Wei-quan, YU Xiao-feng. Preparation of macro-mesostructured pseudoboehmite and γ-Al2O3 with high surface area[J]. Prog Chem, 2007, 19(9): 1322-1330. [10] 潘惠芳, 唐爱军, 李丙兰, 邬晓风, 柯兴民.磷改性γ-Al2O3的酸性调变和积炭行为[J].石油学报(石油加工), 1996, 12(3): 1-8.PAN Hui-fang, TANG Ai-jun, LI Bing-lan, WU Xiao-feng, KE Xing-min. Acidity accommodation and coking behaviour of phosphorus modified γ-Al2O3[J]. Acta Petrol Sin (Pet Process Section), 1996, 12(3): 1-8. [11] TURAGA1 U T, SONG C S. MCM-41 supported Co-Mo catalysts for deep hydrodesulfurization of light cycle oil[J]. Catal Today, 2003, 86(1/4): 129-140. http://web.anl.gov/PCS/acsfuel/preprint%20archive/Files/47_2_Boston_10-02_0281.pdf [12] JIANG T S, ZHAO Q, YIN H B. Synthesis and characterization of Ni-mesoporous molecular sieves with high stability[J]. Inorg Mater, 2007, 43(1): 30-34. doi: 10.1134/S0020168507010086 [13] CHEN L F, WANG J A, NOREÑA L E, AGUILAR J, NAVARRETE J, SALAS P, MONTOYA J A, DEL ÁNGEL P. Synthesis and physicochemical properties of Zr-MCM-41 mesoporous molecular sieves and Pt/H3PW12O40/Zr-MCM-41 catalysts[J]. J Solid State Chem, 2007, 180(10): 2958-2972. doi: 10.1016/j.jssc.2007.08.023 [14] 宋华, 代敏, 宋华林. Ni2P加氢脱硫催化剂[J].化学进展, 2012, 24(5): 757-768.SONG Hua, DAI Min, SONG Hua-lin. Ni2P catalyst for hydrodesulfurization[J]. Prog Chem, 2012, 24(5): 757-768. [15] 余夕志, 王远强, 陈长林, 徐南平, 王延儒. Ni2P/TiO2的制备及其对苯加氢反应的催化性能[J].燃料化学学报, 2006, 34(1): 100-104. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract16959.shtmlYU Xi-zhi, WANG Yuan-qiang, CHEN Chang-lin, XU Nan-ping, WANG Yan-ru. Preparation of Ni2P/TiO2 catalyst and its reactivity for benzene hydrogenation[J]. J Fuel Chem Technol, 2006, 34(1): 100-104. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract16959.shtml [16] YANG Y X, OCHOA-HERNÁNDEZ C, PIZARRO P, O'SHEA V A D, CORONADO J M, SERRANO D P. Influence of the Ni/P ratio and metal loading on the performance of NixPy/SBA-15 catalysts for the hydrodeoxygenation of methyl oleate[J]. Fuel, 2015, 144: 60-70. doi: 10.1016/j.fuel.2014.12.008 [17] JI N, ZHANG T, ZHENG M Y, WANG A Q, WANG H, WANG X D, CHEN J G. Direct catalytic conversion of cellulose into ethylene glycol using nickel promoted tungsten carbide catalysts[J]. Angew Chem Int E, 2008, 47(44): 8510-8513. doi: 10.1002/anie.v47:44 [18] WANG X X, LEFEBVRE F, PATARIN J, BASSET J M. Synthesis and characterization of zirconium containing mesoporous silicas: Ⅰ. Hydrothermal synthesis of Zr-MCM-41-type materials[J]. Microporous Mesoporous Mater, 2001, 42(2/3): 269-276. https://www.researchgate.net/publication/257572036_Synthesis_and_characterization_of_zirconium_containing_mesoporous_silicas_I_Hydrothermal_synthesis_of_Zr-MCM-41-type_materials [19] 王威燕, 杨运泉, 罗和安, 杨彦松, 胡韬, 刘文英, 何兵, 钦柏豪.复合载体TiO2-Al2O3的制备及其对Ni-Mo-S负载型催化剂加氢脱氧性能的影响[J].燃料化学学报, 2011, 39(12): 924-929. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17846.shtmlWANG Wei-yan, YANG Yun-quan, LUO He-an, YANG Yan-song, HU Tao, LIU Wen-ying, HE Bing, QIN Bo-hao. Preparation of TiO2-Al2O3 composite support and its performance in catalystic hydrodeoxygenation[J]. J Fuel Chem Technol, 2011, 39(12): 924-929. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17846.shtml [20] 王健. Ni2P/Ti-MCM-41加氢脱硫催化剂的研制、表征及其加氢脱硫性能研究[D].黑龙江:东北石油大学, 2014.WANG Jian. Preparation, characterization of Ni2P/Ti-MCM-41 catalyst and the hydrodesulfurization performance of dibenzothiophene[D]. Heilongjiang: Northeast Petroleum University, 2014. [21] CECILIA J A, INFANTES-MOLINA A, RODRIGUEZ-CASTELLON E, JIMENEZ-LOPEZ A. Dibenzothiophene hydrodesulfurization over cobalt phosphide catalysts prepared through a new synthetic approach: Effect of the support[J]. Appl Catal B: Environ. 2009, 92(1/2): 100-113. http://www.doc88.com/p-3139737128813.html [22] SENOL O I, VILJAVA T R, KRAUSE A O I. Hydrodeoxygenation of methyl esters on sulphided NiMo/γ-Al2O3 and CoMo/γ-Al2O3Catalysts[J]. Catal Today, 2005, 100(3/4): 331-335. -





 下载:
下载: