-
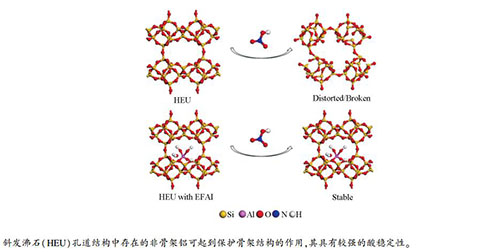
摘要: 采用不同浓度硝酸对斜发沸石(HEU)进行改性处理, 结合元素分析(ICP-AES)、N2物理吸附、X射线粉末衍射(XRD)、魔角旋转固体核磁(MAS NMR)等测试手段及DFT理论计算, 研究了酸处理对斜发沸石结构的影响。结果表明, 硝酸处理对不同平衡阳离子的HEU(Na-K-HEU、NH4-HEU、H-HEU)具有不同的脱铝效果。酸处理后Na-K-HEU和NH4-HEU的硅铝比、比表面积均显著升高。随硝酸浓度增大两个沸石样品的脱铝程度逐渐增加, 同时骨架结构也逐渐破坏, 硝酸浓度达到4 mol/L时其相对结晶度已低于50%。而H-HEU样品的骨架结构稳定, 随硝酸浓度的增大铝含量轻微降低, 硝酸浓度达到6 mol/L时相对结晶度仍高达94.8%。阳离子反交换实验结果证明, 平衡阳离子的类型不是影响HEU骨架稳定性的主要因素。Na-K-HEU和经硝酸铵交换后的NH4-HEU中铝都以骨架铝的形式存在, 而在后者焙烧成为H-HEU时出现部分非骨架铝, 伴随了骨架的稳定化过程。Abstract: The influence of nitric acid treatment on the composition and structure of clinoptilolite (HEU) was analyzed by various characterization techniques, such as element analysis (ICP-AES), N2 physical adsorption, X-ray powder diffraction (XRD), 27Al/29Si MAS NMR and DFT calculation.The Si/Al ratio and BET surface area of Na-K-HEU and NH4-HEU were significantly increased after nitric acid treatment, and meanwhile, their relative crystallinity decreased to below 50% when the concentration of nitric acid reached 4 mol/L, illustrating the severe distortion of zeolite framework.However, the structure of H-HEU was more stable and it could maintain a high relative crystallinity of 94.8% even the concentration of nitric acid reached 6 mol/L.The results of 27Al/29Si MAS NMR indicated that some extra-framework aluminum species appeared in H-HEU and they could protect the framework aluminum from further distortion, whereas this protective effect did not exist on Na-K-HEU and NH4-HEU, as they have little extra-framework aluminum species.It can be further proved that through back-exchanging the H-HEU to Na-HEU and K-HEU, their resistance to nitric acid treatment could be markedly improved.
-
Key words:
- clinoptilolite /
- nitric acid treatment /
- dealumination /
- framework structure
-
表 1 硝酸处理前后不同HEU样品的结构性质和化学组成
Table 1 The textural properties and chemical composition morphological of the samples
Sample Si/Ala ABET/(m2·g-1)b vmicropore /(cm3·g-1)b Relative crystallinity/%c Na-K-HEU 4.86 7.3 0.00050 100 Na-K-HEU-0.5 6.25 229.8 0.091 90.4 Na-K-HEU-1 7.83 205.9 0.083 88.5 Na-K-HEU-2 8.72 186.5 0.076 87.7 Na-K-HEU-4 11.33 126.7 0.046 44.7 Na-K-HEU-6 13.82 82.6 0.025 30.3 NH4-HEU 5.02 8.7 0.005 100 NH4-HEU-0.5 6.87 250.6 0.099 72.6 NH4-HEU-1 7.23 229.9 0.095 65.2 NH4-HEU-2 8.54 198.0 0.079 58.0 NH4-HEU-4 10.9 191.9 0.072 42.6 NH4-HEU-6 14.39 168.4 0.061 27.4 H-HEU 5.07 309.1 0.13 100 H-HEU-0.5 5.44 303.6 0.13 98.3 H-HEU-1 5.53 297.5 0.12 97.8 H-HEU-2 5.66 294.5 0.12 96.8 H-HEU-4 5.70 293.2 0.12 96.7 H-HEU-6 5.92 288.6 0.12 94.8 a:the Si/Al molar ratios were measured by ICP-AES; b:the surface area were calculated by BET method, and the micropore volume were determined by t-plot; c:the relative crystallinity determined by XRD was comparing the sum of diffraction peak area of a zeolite sample with 2θ of 9.9° and 22.5° to that of the reference samples (Na-K-HEU, NH4-HEU, H-HEU, assuming that it had a crystallinity of 100%, respectively) 表 2 4 mol/L硝酸处理前后不同HEU样品的29Si MAS NMR谱图拟合结果
Table 2 29Si chemical shifts and intensity of HEU samples before and after treatment with 4 mol/L nitric acid
Sample (Si/Al)Fa (molar ratio) W/ISi(nAl)/% Si(2Al) Si(1Al) Si(0Al) -95 -99 -101.5 -106 -108 -112 Na-K-HEU 4.86 6.5 8.4 16.7 36.6 16.3 15.5 Na-K-HEU-4 7.61 1.1 0.0 29.1 21.3 8.3 40.2 NH4-HEU 5.06 1.1 8.7 20.0 39.4 16.6 14.2 NH4-HEU-4 7.75 3.9 0.0 23.8 20.0 11.3 41.0 H-HEU 8.30 2.1 2.9 17.1 17.7 28.6 31.6 H-HEU-4 8.94 4.4 2.5 15.2 15.7 28.9 33.3 a:the (Si/Al)F molar ratios were obtained from 29Si MAS NMR spectra 表 3 不同HEU样品的27Al MAS NMR谱图拟合结果
Table 3 Al distribution of various HEU samples determined from 27Al MAS NMR
Sample 4-fold FAl/% 5-fold EFAl/% 6-fold EFAl/% FAl/%a EFAl/%b Na-K-HEU 100.0 0.0 0.0 100.0 0.0 Na-K-HEU-4 69.1 15.5 15.4 69.1 30.9 NH4-HEU 100.0 0.0 0.0 100.0 0.0 NH4-HEU-4 63.7 14.9 21.4 63.7 36.3 H-HEU 64.2 17.1 18.7 64.2 35.8 H-HEU-4 59.3 15.9 24.8 59.3 40.7 Na-HEU 60.1 36.9 3.0 60.1 39.9 K-HEU 68.1 29.9 2.0 68.1 31.9 a:framework Al content; b:etraframework Al content 表 4 不同HEU样品的T位骨架铝分布结果
Table 4 Al distribution at different T sites in various HEU samples
Sample 60/% a 56/% a 53/% a 47/% a Theoretical HEU 26.2 59.8 14.0 Na-K-HEU 24.6 62.2 13.2 0.0 NH4-HEU 23.1 66.6 10.3 0.0 H-HEU 4.1 50.3 8.1 37.5 Na-K-HEU-4 13.3 47.7 18.0 21.0 NH4-HEU-4 17.5 41.5 26.8 14.2 H-HEU-4 3.7 50.2 8.0 38.1 a:the 27Al chemical shifts of various HEU samples 表 5 4 mol/L硝酸处理前后Na-HEU和K-HEU的硅铝比和相对结晶度
Table 5 Si/Al molar rations and relative crystallinity of Na-HEU and K-HEU before and after treatment with 4 mol/L acid
Sample Si/Ala (molar ratio) Relative crystallinity/%b Na-HEU 4.95 100.0 Na-HEU-4 5.77 99.9 K-HEU 5.02 100.0 K-HEU-4 5.82 99.5 a:the Si/Al molar ratios were measured by ICP-AES;
b:the relative crystallinity determined by XRD was comparing the sum of diffraction peak area of a zeolite sample with 2θ of 9.9° and 22.5° to that of the reference samples (Na-K-HEU, NH4-HEU, H-HEU, assuming that it had a crystallinity of 100%, respectively) -
[1] 董殿权, 周志勇, 钟杰, 刘亦凡.合成钾型丝光沸石的酸改型及对K+的离子交换性[J].无机化学学报, 2000, 16(4):580-584.DONG Dian-quan, ZHOU Zhi-yong, ZHONG Jie, LIU Yi-fan.K-type mordenite synthesized by acid advancing and its remembering exchange to K+[J].Chin J Inorg Chem, 2000, 16(4):580-584. [2] 袁俊生. 离子交换法海水提钾技术的应用基础研究[D]. 天津: 天津大学, 2005.YUAN Jun-sheng. Research on fundamentals in the technology of extracting potash from seawater by ion exchange method[D]. Tianjing: Tianjing University, 2015. [3] ALVER B E.A comparative adsorption study of C2H4 and SO2 on clinoptilolite-rich tuff:Effect of acid treatment[J].J Hazard Mater, 2013, 262:627-633. doi: 10.1016/j.jhazmat.2013.09.014 [4] 王奖, 乌力吉日嘎拉, 赵希颖, 照日格图. 斜发沸石在废水处理中的应用研究进展[J]. 环境科学与技术, 2008, 31(8): 49-54.WANG Jiang, N. Ulziijargal, ZHAO Xi-ying, B. Zhaorigetu. Application of clinoptilolite in wastewater treatment[J]. Environ Sci Technol, 2008, 31(8): 49-54. [5] 李曼尼, 格日勒, 新民, 祁晓岚.不同处理方法对白庙子斜发沸石微孔结构的影响[J].内蒙古大学学报(自然科学版), 1995, 26(6):707-712. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=nmgx506.012&dbname=CJFD&dbcode=CJFQLI Man-ni, GE Rile, XIN Min, QI Xiao-lan.The effects of various treatments on the micropore structure of Baimiaozi natural clinoptilolite[J].Acta Sci Natl Univ Neimongol(Nat Sci), 1995, 26(6):707-712. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=nmgx506.012&dbname=CJFD&dbcode=CJFQ [6] GUO H, WANG Y.Batch and column experiments on fluoride removal from waters using modified zeolite[J].J China Univ Geosci, 2000, 11(3):271-274. http://www.cnki.com.cn/Article/CJFDTOTAL-ZDDY200003010.htm [7] 王奖, 特日格乐, 李明智, 照日格图, 贾美林.酸改性天然斜发沸石上丙烷氧化脱氢制丙烯的研究[J].内蒙古师范大学学报(自然科学汉文版), 2012, 41(1):74-80. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=nmgsdxb201201015WANG Jiang, LI Terigele, LI Ming-zhi, B.Zhaorigetu, JIA Mei-lin.Oxidative dehydrogenation of propane to propylene over acid-modified natural clinoptilolite[J].J Inner Mongolia Normal Univ(Nat Sci Ed), 2012, 41(1):74-80. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=nmgsdxb201201015 [8] DZIEDZICKA A, SULIKOWSKI B, RUGGIERO-MIKOLAJCZY M.Catalytic and physicochemical properties of modified natural clinoptilolite[J].Catal Today, 2016, 259:50-58. doi: 10.1016/j.cattod.2015.04.039 [9] ZHAO D Y, KEVAN L, SZOSTAK R.Hydrothermal synthesis of alkali cation heulandite aluminosilicate molecular sieves[J].Zeolites, 1997, 19(5/6):366-369. [10] ZHAO D Y, CLEARA K, OLIVER C, INGRAM C, COOK D, SZOSTAK R, KEVAN L.Characteristics of the synthetic heulandite-clinoptilolite family of zeolites[J].Microporous Mesoporous Mater, 1998, 21(4/6):371-379. [11] 张海燕, 杨承广, 孟祥举, 肖丰收.无有机模板条件下晶种导向合成微孔分子筛晶体材料[J].化学学报, 2012, 70(23):2387-2392. http://www.oalib.com/paper/4699996ZHANG Hai-yan, YANG Cheng-guang, MENG Xiang-ju, XIAO Feng-shou.Seed-directed and organotemplate-free synthesis of zeolites[J].Acta Chim Sin, 2012, 70(23):2387-2392. http://www.oalib.com/paper/4699996 [12] SATOKAWA S, ITABASHI K.Crystallization of single phase (K, Na)-clinoptilolite[J].Microporous Mater, 1997, 8(1/2):49-55. [13] 吴瑞凤, 李曼尼, 施和平.核磁共振法表征磷改性斜发沸石骨架硅铝的变化[J].内蒙古大学学报(自然科学版), 2003, 34(4):462-467. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=nmgdxxb200304022WU Rui-feng, LI Man-ni, SHI He-ping.The characterization of silicon and aluminum content in the frame work of phosphorus modified clinoptilolites by 27Al, 29Si, 31P MAS NMR[J].Acta Sci Nat Univ Neimongol(Nat Sci), 2003, 34(4):462-467. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=nmgdxxb200304022 [14] RIVERA A.Preliminary characterization of drug support systems based on natural clinoptilolite[J].Microporous Mesoporous Mater, 2003, 61(1/3):249-259. [15] RODRIGUEZ-FUENTES G, DE MENORVAL L C, REGUERA E, CHÁCEZ RIVAS F.Solid state multinuclear NMR study of iron species in natural and modified clinoptilolite from Tasajera deposit (Cuba)[J].Microporous Mesoporous Mater, 2008, 111(1/3):577-590. [16] LIN H, LIU Q, DONG Y B, HE Y, WANG L.Physicochemical properties and mechanism study of clinoptilolite modified by NaOH[J].Microporous Mesoporous Mater, 2015, 218:174-179. doi: 10.1016/j.micromeso.2015.07.017 [17] 潘晖华, 何鸣元, 宋家庆, 田辉平, 朱玉霞.USY沸石中非骨架铝形态分析及其对沸石酸性的影响[J].石油学报(石油加工), 2007, 23(2):1-7. http://cdmd.cnki.com.cn/Article/CDMD-10141-2004094295.htmPAN Hui-hua, HE Ming-yuan, SONG Jia-qing, TIAN Hui-ping, ZHU Yu-xia.Chemical state of non-framework aluminum in USY zeolite and its influence on zeolite acidity[J].Acta Pet Sin(Pet Process Sect), 2007, 23(2):1-7. http://cdmd.cnki.com.cn/Article/CDMD-10141-2004094295.htm -





 下载:
下载: