Release of HCl and H2S during gasification of refuse derived-fuel chars
-
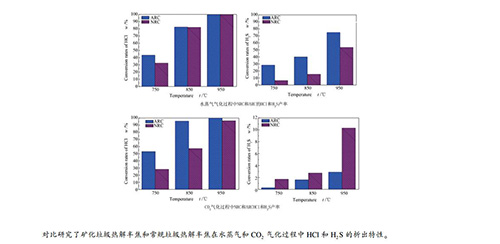
摘要: 通过水平管式气化炉和化学吸收法,对比研究了矿化垃圾热解半焦(ARC)和常规垃圾热解半焦(NRC)在水蒸气和CO2气化过程中腐蚀性气体(HCl和H2S)的析出特性,考察了气化温度、气化介质类型和流量对腐蚀性气体析出特性的影响。当气化温度升至950℃,ARC在水蒸气气化过程中的碳气化率、HCl和H2S产率分别为66.1%、100%和74.9%,而其在CO2气化过程中的碳气化率、HCl和H2S产率分别为77.8%、100%和2.9%;NRC在水蒸气气化过程中的碳气化率、HCl和H2S产率分别为98.8%、100%和53.7%,而其在CO2气化过程中的碳气化率、HCl和H2S产率分别为100%、96.2%和10.3%。以NRC为原料,考察了水蒸气和CO2流量对其HCl和CO2析出特性的影响。NRC的HCl和H2S产率均随水蒸气流量增加而增加,但当水碳比大于等于3.3时,其促进作用不再明显。NRC的HCl产率随CO2流量的增加而增加,而H2S产率随CO2流量的增加而减小。Abstract: Using a horizontal tubular reactor together with chemical adsorption, release characteristics of corrosive gases, viz., HCl and H2S, during steam and CO2 gasification process of aged and normal refuse derived-fuels char (ARC and NRC) were investigated. Effects of gasification temperature, type and flow rate of gasification medium on their release behaviors were examined. In H2O gasification at 950℃ the carbon gasification rates, HCl and H2S yields of ARC are 66.1%, 100% and 74.9%, respectively, and those are 77.8%, 100% and 2.9% in CO2 gasification, respectively. The carbon gasification rates, HCl and H2S yields of NRC in H2O gasification are 98.8%, 100% and 53.7%, and those are 100%, 96.2% and 10.3% in CO2 gasification, respectively. The release characteristics of HCl and H2S are investigated with different flow rates of H2O and CO2 in the NRC gasification. HCl and H2S yields of NRC increase with increasing flow rate of H2O, but the promoting effect can be ignored when H2O/C is ≥ 3.3. HCl yield of NRC increases but H2S yield decreases with increasing flow rate of CO2.
-
Key words:
- HCl /
- H2S /
- aged refuse derived-fules char /
- normal refuse derived-fuels char /
- gasification
-
表 1 ARDF和NRDF的元素分析与工业分析
Table 1 Ultimate and proximate analyses of ARDF and NRDF
Sample Ultimate analysis wdb*/% C H N total S inorganic sulfur total Cl inorganic Cl ARDF 35.95 2.56 0.85 0.65 0.103 0.95 0.46 NRDF 57.26 6.23 0.87 0.15 0.007 1.37 0.14 *: dried basis 表 2 气化半焦原料的元素分析
Table 2 Ultimate analysis of raw material in char gasification experiment
Sample Ultimate analysis wdb*/% C H N total S inorganic sulfur total Cl inorganic Cl ARC 37.78 0.62 0.44 0.99 0.784 1.77 1.23 NRC 60.09 0.73 0.99 0.14 0.086 2.34 1.49 *: dried basis -
[1] "十三五"全国城镇生活垃圾无害化处理设施建设规划[R].北京: 国家发展改革委住房城乡建设部, 2016."The 13th Five-Year Plan" construction planning for harmless treatment facilities of national municipal waste[R]. Beijing: Ministry of Housing and Urban-Rural Development of the People's Republic of China, 2016. [2] 赵由才, 柴晓利, 牛冬杰.矿化垃圾基本特性研究[J].同济大学学报(自然科学版), 2006, 34(10):1360-1364. doi: 10.3321/j.issn:0253-374X.2006.10.017ZHAO You-cai, CHAI Xiao-li, NIU Dong-jie. Characteristics of aged refuse in closed refuse landfill in Shanghai[J]. J Tongji Univ (Nat Sci)), 2006, 34(10):1360-1364. doi: 10.3321/j.issn:0253-374X.2006.10.017 [3] ROTHEUT M, QUICKER P. Energetic utilisation of refuse derived fuels from landfill mining[J]. Waste Manage, 2017, 62:101-117. doi: 10.1016/j.wasman.2017.02.002 [4] LI G K, HOU F, GUO Z, YAO G, SANG N. Analyzing nutrient distribution in different particle-size municipal aged refuse[J]. Waste Manage, 2011, 31(11):2203-2207. doi: 10.1016/j.wasman.2011.06.010 [5] COMMISSION E. Council Directive 99/31/EC of 26 April 1999 on the landfill of waste (Landfill Directive)[EB]. 1999. [6] BOSMANS A, VANDERREYDT I, GEYSEN D, HELSEN L. The crucial role of Waste-to-Energy technologies in enhanced landfill mining:A technology review[J]. J Clean Prod, 2013, 55(14):10-23. http://www.cabdirect.org/abstracts/20133320582.html [7] ROTHEUT M, QUICKER P. Energetic utilization of refuse derived fuels from landfill mining[J]. Waste Mange, 2017, 62:101-117. doi: 10.1016/j.wasman.2017.02.002 [8] CHALERMCHAROENRAT S, LAOHALIDANOND K, KERDSUWAN S. Optimization of combustion behavior and producer gas quality from reclaimed landfill through highly densify RDF-gasification[J]. Energy Procedia, 2015, 79:321-326. doi: 10.1016/j.egypro.2015.11.496 [9] 袁浩然, 鲁涛, 熊祖鸿, 黄宏宇, 小林敬幸, 陈勇, 黎志强.城市生活垃圾热解气化技术研究进展[J].化工进展, 2012, 31(2):421-427. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK201200023127YUAN Hao-ran, LU Tao, XIONG Zu-hong, HUANG Hong-yu, KOBAYASHI Noriyuki, CHEN Yong, LI Zhi-qiang. Advance in pyrolysis and gasification of municipal solid waste study[J]. Chem Ind Eng Prog, 2012, 31(2):421-427. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK201200023127 [10] HJELMAR O. Disposal strategies for municipal solid waste incineration residues[J]. J Hazard Mater, 1996, 47(1/3):345-368. http://www.sciencedirect.com/science/article/pii/0304389495001115 [11] MAKEN S, JANG S H, PARK J W, PARK J W, SONG H C, LEE S, CHANG E H. Vitrification of MSWI fly ash using Brown's gas and fate of heavy metals[J]. J Sci Ind Res, 2005, 64(3):198-204. [12] TUPPURAINEN K, HALONEN I, RUOKOJARVI P, TARHANEN J, RUUSKANEN J. Formation of PCDDs and PCDFs in municipal waste incineration and its inhibition mechanisms:A review[J]. Chemosphere, 1998, 36(7):1493-1511. doi: 10.1016/S0045-6535(97)10048-0 [13] SENNECA O. Kinetics of pyrolysis, combustion and gasification of three biomass fuels[J]. Fuel Process Technol, 2007, 88(1):87-97. doi: 10.1016/j.fuproc.2006.09.002 [14] SHUIT S H, TAN K T, LEE K T, KAMARUDDIN A H. Oil palm biomass as a sustainable energy source:A Malaysian case study[J]. Energy, 2009, 34(9):1225-1235. doi: 10.1016/j.energy.2009.05.008 [15] PAN T J, GESMUNDO F, NIU Y. Corrosion behavior of three iron-based model alloys in reducing atmospheres containing HCl and H2S at 600℃[J]. Corros Sci, 2007, 49(3):1362-1377. doi: 10.1016/j.corsci.2006.06.014 [16] HU H, FANG Y, LIU H, YU R, LUO G, LIU W, LI A, YAO H. The fate of sulfur during rapid pyrolysis of scrap tires[J]. Chemosphere, 2014, 97(1):102-107. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6bc4663d9c73f5d9a7e7e41b8c4bd721 [17] YUAN G, CHEN D, YIN L, WANG Z, ZHAO L, WANG J Y. High efficiency chlorine removal from polyvinyl chloride (PVC) pyrolysis with a gas-liquid fluidized bed reactor[J]. Waste Manage, 2014, 34(6):1045-1050. doi: 10.1016/j.wasman.2013.08.021 [18] ZHU H M, JIANG X G, YAN J H, CHI Y, CEN K F. TG-FTIR analysis of PVC thermal degradation and HCl removal[J]. J Anal Appl Pyrolysis, 2008, 82(1):1-9. doi: 10.1016/j.jaap.2007.11.011 [19] LANE D J, VAN EYK P J, ASHMAN P J, KWONG C W, DE NYS R, ROBERTS D A, COLE A J, LEWIS D M. Release of Cl, S, P, K, and Na during thermal conversion of algal biomass[J]. Energy Fuels, 2015, 29(4):2542-2554. doi: 10.1021/acs.energyfuels.5b00279 [20] 林均衡, 杨文申, 阴秀丽, 吴创之.矿化垃圾衍生燃料热解过程HCl与H2S析出规律[J].燃料化学学报, 2018, 46(2):152-159. doi: 10.3969/j.issn.0253-2409.2018.02.004LIN Jun-heng, YANG Wen-shen, YIN Xiu-li, WU Chuang-zhi. Release of HCl and H2S during pyrolysis of aged refuse derived-fuels[J]. J Fuel Chem Technol, 2018, 46(2):152-159. doi: 10.3969/j.issn.0253-2409.2018.02.004 [21] 阮松宾.广西县级城镇生活垃圾处理方式和工艺探讨[J].环境卫生工程, 2008, 16(1):27-30. doi: 10.3969/j.issn.1005-8206.2008.01.008RUAN Song-bin. Discussion about means and process of country towns domestic waste treatment in Guangxi[J]. Environ Sanit Eng, 2008, 16(1):27-30. doi: 10.3969/j.issn.1005-8206.2008.01.008 [22] HU H, FANG Y, LIU H, YU R, LUO G, LIU W, LI A, YAO H. The fate of sulfur during rapid pyrolysis of scrap tires[J]. Chemosphere, 2014, 97(1):102-107. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6bc4663d9c73f5d9a7e7e41b8c4bd721 [23] YU J, SUN L, MA C, QIAO Y, YAO H. Thermal degradation of PVC:A review[J]. Waste Manage, 2016, 48:300-314. doi: 10.1016/j.wasman.2015.11.041 [24] 杜耀, 方圆, 沈东升, 龙於洋.填埋场中硫化氢恶臭污染防治技术研究进展[J].农业工程学报, 2015, 31(1):269-275. http://d.old.wanfangdata.com.cn/Periodical/nygcxb2015z1032DU Yao, FANG Yuan, SHEN Dong-sheng, LONG Yu-yang. Review on pollution control technologies of hydrogen sulfide odor in landfill[J]. Trans Chin Soc Agric Eng, 2015, 31(1):269-275. http://d.old.wanfangdata.com.cn/Periodical/nygcxb2015z1032 [25] RECARI J, BERRUECO C, ABELLÍ S, MONTANÉ D, FARRIOL X. Gasification of two solid recovered fuels (SRFs) in a lab-scale fluidized bed reactor:Influence of experimental conditions on process performance and release of HCl, H2S, HCN and NH3[J]. Fuel Process Technol, 2016, 142:107-114. doi: 10.1016/j.fuproc.2015.10.006 [26] DUAN L, ZHAO C, ZHOU W, QU C, CHEN X. Investigation on coal pyrolysis in CO2 atmosphere[J]. Energy Fuels, 2009, 23(7):3826-3830. doi: 10.1021/ef9002473 [27] FRIGGE L, ELSERAFI G, STROHLE J, EPPLE B. Sulfur and chlorine gas species formation during coal pyrolysis in nitrogen and carbon dioxide atmosphere[J]. Energy Fuels, 2016, 30(9):7713-7720. doi: 10.1021/acs.energyfuels.6b01080 -





 下载:
下载: