Effect of the additives PVP and MTS on structural properties and synthesis mechanism of Beta zeolite
-
摘要: 分别以铝酸钠和硅溶胶为铝源和硅源,四乙基氢氧化铵为模板剂,在水热条件下,考察了添加剂(聚乙烯吡咯烷酮(PVP)和甲基三乙氧基硅烷(MTS))对合成Beta分子筛结构与性能的影响,并通过XRD、TEM、BET、ICP、29Si-NMR和NH3-TPD等方法对合成样品进行了结构表征和作用机理讨论,同时以催化裂化异丙苯为模型反应评价其催化性能。结果表明,与传统Beta分子筛相比,加入PVP后所得样品具有更高结晶度和较高的硅铝物质的量比(25.68)以及较大的比表面积(772 m2/g);而加入MTS后尽管具有较大比表面积(657 m2/g)和较高硅铝物质的量比(25.76),但是结晶度却相对降低,且粒径减小(160-320 nm)。两种添加剂作用下所得样品均具有更多酸量,在催化裂化异丙苯的反应中表现出较高的催化活性。Abstract: Beta zeolites were successfully synthesized via hydrothermal method using sodium aluminate as aluminium source, silica sol (Ludox) as silicon source and TEAOH as template, respectively. The effects of different additives polyvinyl pyrrolidone (PVP) and Methyltriethoxysilane (MTS) on their structural properties and synthesis mechanism were investigated in detail by various characterizations, such as XRD, TEM, BET, ICP, 29Si-NMR and NH3-TPD method. Also, their catalytic performances were evaluated through catalytic cracking of cumene. The results showed that the beta zeolite obtained in the PVP-added system presented a better crystallinity, higher Si/Al molar ratio (25.68), and larger surface area (772 m2/g). Comparably, the MTS-effected samples revealed a larger surface area (657 m2/g), higher Si/Al molar ratio (25.76), but lower crystallinity and smaller particle sizes (around 160-320 nm). Moreover, both of them exhibited a good catalytic activity for cumene cracking due to the existences of abundant and strong acid content.
-
Key words:
- PVP /
- MTS /
- Beta zeolite /
- catalytic cracking /
- structural characterization /
- hydrothermal synthesis
-
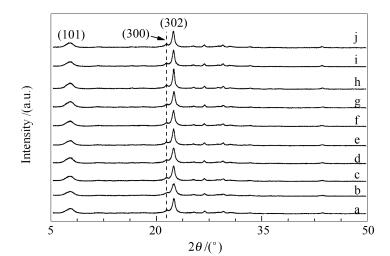
图 1 不同添加剂合成Beta分子筛的XRD谱图
Figure 1 XRD patterns of Beta zeolite with different additives
a: traditional; b: (MTS/Al2O3) 2.3; c: (PVP (K15)/Al2O3) 4.54×10-2; d: (PVP (K15)/Al2O3) 1.36×10-1; e: (PVP (K15)/Al2O3) 2.72×10-1; f: (PVP (K30)/Al2O3) 7.81×10-3; g: (PVP (K30)/Al2O3) 2.34×10-2; h: (PVP (K30)/Al2O3) 4.68×10-2; i: (PVP (K90)/Al2O3) 3.50×10-4; j: (PVP (K90)/Al2O3) 1.05×10-3
表 1 样品的织构参数
Table 1 Textural parameters of the samples
Sample ABET /(m2·g-1) Amicro/(m2·g-1) vmicro/(cm3·g-1) vpore/(cm3·g-1) Traditional 577 390 0.22 0.59 MTS 657 394 0.26 0.67 PVP (K30)/Al2O3(7.81×10-3) 772 578 0.30 0.57 PVP (K30)/Al2O3(2.34×10-2) 755 589 0.29 0.52 表 2 NMR及ICP测定的样品元素含量
Table 2 Elemental content measured by ICP and NMR
Sample NMR ICP/(mg·L-1) Si/Al
(molar ratio)Q4:Q3:Q2 Si/Al Na Al Si Beta (traditional) 0.22:1:5.89 19.75 0.27 1.73 38.52 21.47 Beta (traditional) (after ion exchanged) 0.15 1.46 39.82 26.37 Beta (MTS) 0.08:1:5.67 23.28 0.27 2.05 54.80 25.76 Beta (K30 2.34×10-2) 0.36:1:8.57 23.09 0.24 1.66 44.21 25.68 表 3 不同PVP添加量的样品酸量
Table 3 Acid amount of Samples with different additives
Catalyst Acid amount /(μmol·g-1) weak-
acidmedium-
acidstrong-
acidTraditional 6.9 143.3 230.9 MTS 6.0 280.4 648.6 PVP (K30)/Al2O3(7.81×10-3) 3.9 162.0 577.0 PVP (K30)/Al2O3 (2.34×10-2) 4.0 220.0 395.0 PVP (K30)/Al2O3(4.68×10-2) 5.7 205.8 342.8 -
[1] WADLINGER R L, KERR G T, ROSINSKI E J. Catalytic composition of a crystalline zeolite:US, 3308069[P]. 1967-03-07. [2] BELLUSSI G, PAZZUCONI G, PEREGO C, GIROTTI G, TERZONI G. Liquid-phase alkylation of benzene with light olefinscatalyzed by β-Zeolites[J]. J Catal, 1995, 157(1):227-234. doi: 10.1006/jcat.1995.1283 [3] ARRIBAS M A, MARTINEZ A. Simultaneous isomerization of n-heptane and saturation of benzene over Pt/Beta catalysts:The influence of zeolite crystal size on product selectivity and sulfur resistance[J]. Catal Today, 2001, 65(2/4):117-122. [4] DING L, ZHENG Y, HONG Y, RING Z. Effect of particle size on the hydrothermal stability of zeolite Beta[J]. Microporous Mesoporous Mater, 2007, 101(3):432-439. doi: 10.1016/j.micromeso.2006.12.008 [5] SERRANO D P, AGUADO J, ESCOLA J M, RODRIGUEZ J M, PERAL A. Hierarchical zeolites with enhanced textural and catalytic properties synthesized from organofunctionalized seeds[J]. Chem Mater, 2006, 18(10):2462-2464. doi: 10.1021/cm060080r [6] AGUADO J, SERRANO D P, RODRIGUEZ J M. Zeolite Beta with hierarchical porosity prepared from arganofuctionalized seed[J]. Microporous Mesoporous Mater, 2008, 115(3):504-513. doi: 10.1016/j.micromeso.2008.02.026 [7] 王润伟, 林森, 孟祥举, 李滨松, 蒋大振, 肖丰收, 裘式纶. Hβ沸石粒度对异丁烷与丁烯烷基化反应活性和选择性的影响[J].高等学校化学学报, 2003, 24(2):205-207. http://www.cjcu.jlu.edu.cn/CN/abstract/abstract15348.shtmlWANG Run-wei, LIN Sen, MENG Xiang-ju, LI Bin-song, JIANG Da-zhen, XIAO Feng-shou, QIU Shi-lun. Influence of catalysts particle size on alkylation of isobutane with butene over Hβ zeolites[J]. Chem J Chin Univ, 2003, 24(2):205-207. http://www.cjcu.jlu.edu.cn/CN/abstract/abstract15348.shtml [8] MOLLER K, YILMAZ B, JACUBINAS R M, MULLER U, BEIN T. One-step synthesis of hierarchical zeolite beta via network formation of uniform nanocrystals[J]. J Am Chem Soc, 2011, 133(14):5284-5295. doi: 10.1021/ja108698s [9] DING L, ZHENG Y, ZHANG Z, RING Z, CHEN J. Effect of agitation on the synthesis of zeolite beta and its synthesis mechanism in absence of alkali cations[J]. Microporous Mesoporous Mater, 2006, 94(1/3):1-8. [10] 单志超, 刘思宇, 李彩今, 朱龙凤, 孟祥举, 肖丰收. Y沸石的高温合成[J].物理化学学报, 2011, 27(4):959-964. http://www.cnki.com.cn/Article/CJFDTOTAL-WLHX201104035.htmSHAN Zhi-chao, LIU Si-yu, LI Cai-jin, ZHU Long-feng, MENG Xiang-ju, XIAO Feng-shou. High-temperature synthesis of zeolite Y[J]. Acta Phys Chim Sin, 2011, 27(4):959-964. http://www.cnki.com.cn/Article/CJFDTOTAL-WLHX201104035.htm [11] VUONG G T, DO T O. A new route for the synthesis of uniform nanozeolites with hydrophobic external surface in organic solvent medium[J]. J Am Chem Soc, 2007, 129(13):3810-3811. doi: 10.1021/ja069058p [12] VAN BOKHOVEN J A, AM V D E, KONINGSBERGER D C. Three-coordinate aluminum in zeolites observed with in situ X-Ray absorption near-edge spectroscopy at the Al K-edge:Flexibility of aluminum coordinations in zeolites[J]. J Am Chem Soc, 2003, 125(24):7435-7442. doi: 10.1021/ja0292905 [13] 徐如人, 庞文琴, 于吉红, 霍启升, 陈接胜.分子筛与多孔材料化学[M].北京:科学出版社, 2004:74-76.XU Ru-ren, PANG Wen-qin, YU Ji-hong, HUO Qi-sheng, CHEN Jie-sheng. Chemistry-Zeolites and Porous Materials[M]. Beijing:Science Press, 2004:74-76. [14] YARIPOUR F, SHARIATINIA Z, SAHEBDELFAR S, LRANDOUKHT A. Effect of boron incorporation on the structure, products selectivities and lifetime of H-ZSM-5 nanocatalyst designed for application in methanol-to-olefins (MTO) reaction[J]. Microporous Mesoporous Mater, 2015, 203:41-53. doi: 10.1016/j.micromeso.2014.10.024 [15] 朱金红, 刘靖, 赵文江, 王祥生, 刘秀梅, 包信和. Al-MUS-S介孔及介孔-微孔分子筛的合成、表征及其催化性能[J].催化学报, 2004, 25(9):741-747. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA200409016.htmZHU Jin-hong, LIU Jing, ZHAO Wen-jiang, WANG Xiang-sheng, LIU Xiu-mei, BAO Xin-he. Synthesis and characterization of Al-MSU-S mesoporous and meso-microporous molecular sieves and their catalytic performance[J]. Chin J catal, 2004, 25(9):741-747. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA200409016.htm [16] KUMARAN G M, GARG S, SONI K, KUMAR M, GUPTA J K, SHARMA L D, RAMA RAO K S, MURALI DHAR G. Synthesis and characterization of acidic properties of Al-SBA-15 materials with varing Si/Al ratios[J]. Microporous Mesoporous Mater, 2008, 114(1/3):103-109. https://www.researchgate.net/publication/229316495_Synthesis_and_characterization_of_acidic_properties_of_Al-SBA-15_materials_with_varying_SiAl_ratios -





 下载:
下载: