Effect of Ni2P loading on the structure and naphthalene hydrogenation performance of Ni2P/Ce-Al2O3 catalyst
-
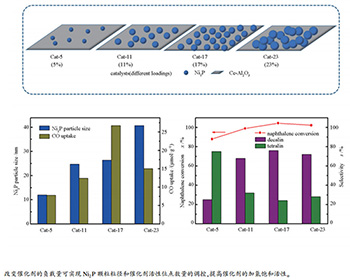
摘要: 采用程序升温还原法制备了一系列Ni2P/Ce-Al2O3催化剂,考察了制备过程中Ni2P负载量对催化剂结构及萘加氢饱和性能的影响。结果表明,Ni2P负载量可调控活性组分Ni2P与载体Ce-Al2O3之间的相互作用,进而调变催化剂的比表面积、Ni2P粒径及催化剂活性位点数量。当Ni2P负载量(质量分数)为17%时,催化剂具有较大的比表面积(40 m2/g)、较小的Ni2P粒径(26.3 nm)和最多的活性位点数量(26.7 μmol/g);同时,该催化剂萘转化率为95%,十氢萘选择性为76%,且活性稳定性良好,这主要归因于催化剂大的比表面积和高的活性位点数量为反应提供了更多的场所。
-
关键词:
- 萘加氢 /
- Ni2P/Ce-Al2O3 /
- Ni2P负载量 /
- 程序升温还原法
Abstract: A series of Ni2P/Ce-Al2O3 catalysts were prepared by temperature-programmed reduction method, and the influence of Ni2P loading on the catalyst structure and naphthalene hydrogenation saturation performance was investigated. The results show that the specific surface area, the Ni2P particle size and the active site number of the as-synthesized catalysts are greatly affected by Ni2P loading, which is derived from the variable interaction between active component Ni2P and the support Ce-Al2O3. When the Ni2P loading is 17% (mass ratio), the catalyst possesses a large specific surface area (40 m2/g), a small Ni2P particle size (26.3 nm), and the maximum number of active sites (26.7 μmol/g). Meanwhile, the conversion rate of naphthalene and the selectivity of decalin reach to 95% and 76%, respectively, and the activity stability of the catalyst is good, which is mainly attributed to the large specific surface area and high number of active sites of the catalyst providing more sites for the reaction. -
表 1 不同Ni2P负载量的Ni2P/Ce-Al2O3催化剂的织构性质和CO吸附量
Table 1 Textural and structural properties of the Ni2P/Ce-Al2O3 catalysts with different Ni2P loadings
Sample BET surface area
A/(m2·g-1)Maximum aperture
/nmCrystallite size
d/nm aParticle
size d/nm bCO uptake
/(μmol·g-1)Al2O3 107 6.4 - - - Ce-Al2O3 93 6.7 - - - Cat-5 87 4.3 - 12.0 7.8 Cat-11 60 5.2 13.1 24.7 12.4 Cat-17 40 5.7 13.7 26.3 26.7 Cat-23 15 6.1 33.1 40.6 15.0 Cat-100 7 10.7 44.9 - 0.5 a: calculated from XRD results; b: calculated from TEM results -
[1] COOPER B H, DONNIS B. Aromatic saturation of distillates:An overview[J]. Appl Catal A:Gen, 1996, 137(2):203-223. doi: 10.1016/0926-860X(95)00258-8 [2] 焦燕, 冯利利, 朱岳麟, 熊常健.美国军用喷气燃料发展综述[J].火箭推进, 2008, 34(1):30-35. doi: 10.3969/j.issn.1672-9374.2008.01.007JIAO Yan, FENG Li-li, ZHU Yue-lin, XIONG Chang-jian. Review of American military jet fuels development[J]. J Rocket Propul, 2008, 34(1):30-35). doi: 10.3969/j.issn.1672-9374.2008.01.007 [3] 佟瑞利, 王永刚, 张旭, 张海永, 戴谨泽, 林雄超, 许德平. P改性NiW/Al2O3的低温焦油芳烃组分加氢性能研究[J].燃料化学学报, 2015, 43(12):1461-1469. doi: 10.3969/j.issn.0253-2409.2015.12.009TONG Rui-li, WANG Yong-gang, ZHANG Xu, ZHANG Hai-yong, DAI Jin-ze, LIN Xiong-chao, XU De-ping. Effect of phosphorus modification on the catalytic properties of NiW/Al2O3 in the hydrogenation of aromatics from coal tar[J].J Fuel Chem Technol, 2015, 43(12):1461-1469. doi: 10.3969/j.issn.0253-2409.2015.12.009 [4] HE T, WANG Y, MIAO P, LI J, WU J, FANG Y. Hydrogenation of naphthalene over noble metal supported onmesoporous zeolite in the absence and presence of sulfur[J]. Fuel, 2013, 106:365-371. doi: 10.1016/j.fuel.2012.12.025 [5] 李贺, 殷长龙, 赵雪萍, 李秀峥, 柳云骐, 刘晨光.萘、四氢萘和十氢萘的加氢或脱氢反应与催化剂的研究进展[J].石油化工, 2014, 43(8):971-979. doi: 10.3969/j.issn.1000-8144.2014.08.020LI He, YIN Chang-long, ZHAO Xue-ping, LI Xiu-zheng, LIU Yun-qi, LIU Chen-guang. Progresses in hydrogenation orhehydrogenation of naphthalene, tetralin and decalin, and the catalysts[J]. Petrochem Technol, 2014, 43(8):971-979. doi: 10.3969/j.issn.1000-8144.2014.08.020 [6] LI R, GUAN Q, WEI R, YANG S, SHU Z, DONG Y, CHEN J, LI W. A potential regularity for enhancing the hydrogenation properties of Ni2P[J]. J Phy Chem C, 2015, 119(5):2557-2565. doi: 10.1021/jp511191e [7] LIU D, WANG A, LIU C, PRINS R. Bulk and Al2O3-supported Ni2P HDS catalysts prepared by separating the nickel and hypophosphite sources[J]. Catal Commun, 2016, 77:13-17. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=412aa624e55137137db3b75efb902e64 [8] YUN G, GUAN Q, LI W. The synthesis and mechanistic studies of a highly active nickel phosphide catalyst for naphthalene hydrodearomatization[J]. RSC Adv, 2017, 7(14):8677-8687. doi: 10.1039/C7RA00250E [9] YANG Y, LI J, LV G, ZHANG L. Novel method to synthesize Ni2P/SBA-15 adsorbents for the adsorptive desulfurization of model diesel fuel[J]. J Alloy Compd, 2018, 745:467-476. doi: 10.1016/j.jallcom.2018.02.156 [10] SONG H, WANG J, WANG Z, SONG H, LI F, JIN Z. Effect of titanium content on dibenzothiophene HDS performance over Ni2P/Ti-MCM-41 catalyst[J]. J Catal, 2014, 311:257-265. doi: 10.1016/j.jcat.2013.11.021 [11] D'AQUINO A I, DANFORTH S J, CLINKINGBEARD T R, ILIC B, PULLAN L, REYNOLDS M A, MURRAY B D, BUSSELL M E. Highly-active nickel phosphide hydrotreating catalysts prepared in situ using nickel hypophosphite precursors[J]. J Catal, 2016, 335:204-214. doi: 10.1016/j.jcat.2015.12.006 [12] 宋华, 姜楠, 宋华林, 李锋, 代敏, 万霞, 徐晓伟.镍磷物质的量比对负载型Ni2P/MCM-41催化剂结构及加氢脱硫性能的影响[J].燃料化学学报, 2015, 43(3):338-343. doi: 10.3969/j.issn.0253-2409.2015.03.012SONG Hua, JIANG Nan, SONG Hua-lin, LI Feng, DAI Min, WAN Xia, XU Xiao-wei. Effect of Ni/P mol ratio on structure and performance of hydrodesulfurization of Ni2P/MCM-41 catalyst[J].J Fuel Chem Technol, 2015, 43(3):338-343. doi: 10.3969/j.issn.0253-2409.2015.03.012 [13] 米星, 何广湘, 郭晓燕, 杨索和, 罗国华, 徐新, 靳海波. Ni/γ-Al2O3催化剂上萘加氢生成十氢萘的催化反应研究[J].燃料化学学报, 2018, 46(7):879-885. doi: 10.3969/j.issn.0253-2409.2018.07.015MI Xing, HE Guang-xiang, GUO Xiao-yan, YANG Suo-he, LUO Guo-hua, XU Xin, JIN Hai-bo. Effect of reaction conditions on the hydrogenation of naphthalene to decalin over Ni/Al2O3 catalyst[J]. J Fuel Chem Technol, 2018, 46(7):879-885. doi: 10.3969/j.issn.0253-2409.2018.07.015 [14] LI X, FENG J, GUO J, WANG A, PRINS R, DUAN X, CHEN Y. Preparation of Ni 2P/Al2O3 by temperature-programmed reduction of a phosphate precursor with a low P/Ni ratio[J]. J Catal, 2016, 334:116-119. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=739a7343455ae38b51c8703efca97ae9 [15] ZHANG X, ZHANG Q, ZHAO A, GUAN J, HE D, HU H, LIANG C. Naphthalene hydrogenation over silica supported nickel phosphide in the absence and presence of N-containing compounds[J]. Energy Fuels, 2010, 24(7):3796-3803. doi: 10.1021/ef100342z [16] OYAMA S T, GOTT T, ZHAO H, LEE Y K. Transition metal phosphide hydroprocessing catalysts:A review[J]. Catal Today, 2009, 143(1/2):94-107. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0226572816/ [17] 姚程程.负载量对Ni2P/SiO2催化剂结构及氯苯加氢脱氯性能的影响[D].天津: 天津大学, 2010. http://cdmd.cnki.com.cn/Article/CDMD-10056-1011262518.htmYAO Cheng-cheng. Effect of loading on Ni2P/SiO2 catalyst structure and hydrodechlorination performance of chlorobenzene[D]. Tianjin: Tianjin University, 2010. http://cdmd.cnki.com.cn/Article/CDMD-10056-1011262518.htm [18] SONG H, DAI M, SONG H, WAN X, XU X. A novel synthesis of Ni2P/MCM-41 catalysts by reducing a precursor of ammonium hypophosphite and nickel chloride at low temperature[J]. Appl Catal A:Gen, 2013, 462:247-255. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=52ccf707bd8757a5597297c77cd9c953 -





 下载:
下载: