Effect of particle size on gasification of char with different coal ranks
-
摘要: 选取气流床气化炉所使用不同煤阶的八种煤焦,通过多级筛分制得单分散煤粉样本,利用热重分析仪考察了气化温度、煤焦粒径对不同煤阶煤焦CO2气化反应的影响。对比了不同碳转化率阶段下的反应差异,并讨论了高碳转化率阶段的情况。研究表明,随着煤阶的升高,煤焦碳微晶结构更为有序,其气化活性也随之降低。煤焦粒径对气化反应的影响与煤阶有关。对于无烟煤,平均粒径300 μm的无烟煤煤焦转化率达到95%所需时间可达40 μm煤焦的7倍;对于褐煤与烟煤,由于其孔隙结构较为发达,粒径变化对煤焦气化活性的影响并不明显。综合煤阶、气化温度、煤焦粒径对气化反应活性的影响发现,相较低阶煤,提高气化温度、减小煤焦粒径能够更有效地提升高阶煤气化反应活性。Abstract: Eight coal chars of different coal ranks were prepared to investigate the effects of coal rank, reaction temperature and particle size on char-CO2 gasification using a thermogravimetric analyzer (TGA). The variation of char-CO2 gasification rate with carbon conversion was studied, especially at high conversion stage. As a result, the crystalline structures of higher rank chars are more orderly, resulting in a lower gasification rate. For high rank coals like anthracite, the CO2 gasification rate of char with 40 μm size is about 7 times higher than that with 300 μm size to reach 95% conversion at 1 300℃. For low rank coals, particle size has little effect on the gasification rate. The results indicate that the effect of particle size on char-CO2 gasification is dependent on coal rank. The specific surface area of high rank coals is much smaller than that of low rank coals, which contributes to the significant effect of particle size on high rank coal gasification. The effect of gasification temperature and particle size on gasification rate for high rank coals is more significant than that for low rank coals.
-
Key words:
- coal rank /
- gasification temperature /
- particle size /
- CO2 gasification
-
表 1 煤样的工业分析、元素分析及灰熔点
Table 1 Proximate, ultimate analysis and ash fusion temperature of coal samples
Sample Proximate analysis wd/% Ultimate analysis wd/% Ash fusion temperature t/℃ V FC A C H N S O DT ST HT FT YN 55.23 39.86 4.91 59.80 4.86 0.76 0.26 29.41 1 081 1 140 1 151 1 163 NM 49.22 40.33 10.45 74.65 3.32 1.20 0.58 9.80 1 086 1 105 1 121 1 124 SF 35.42 58.29 6.29 79.14 2.32 1.12 0.77 10.36 1 152 1 167 1 175 1 179 SH 32.29 60.89 6.82 77.18 3.73 1.04 0.60 10.63 1 113 1 137 1 143 1 156 ZJX 30.19 51.58 18.23 70.23 4.28 1.19 0.34 5.73 1 458 >1 500 >1 500 >1 500 JC 5.35 78.32 16.33 78.76 1.08 0.91 1.97 0.95 1 440 >1 500 >1 500 >1 500 GZ 8.45 73.76 17.79 74.52 2.00 1.05 4.07 0.57 1 271 1 345 1 359 1 373 GP 7.96 76.81 15.23 74.59 3.03 0.91 3.12 3.12 1 342 >1 500 >1 500 >1 500 表 2 煤样的灰组分分析
Table 2 Ash compositions of samples
Sample Composition w/% SiO2 Al2O3 Fe2O3 CaO SO3 MgO Na2O TiO2 K2O SrO YN 29.17 9.40 15.29 16.17 12.89 14.29 0.47 0.49 0.06 0.09 NM 43.46 14.43 14.13 11.81 10.02 3.04 1.46 0.63 0.32 0.14 SF 44.20 18.19 11.28 15.58 5.63 1.12 1.89 0.60 0.95 0.41 SH 46.68 20.34 6.65 14.39 6.52 1.03 2.20 0.79 0.88 0.12 ZJX 58.06 34.19 2.33 0.85 0.50 0.43 0.58 1.99 0.83 0.19 GZ 50.73 31.36 8.93 1.79 1.46 1.05 0.35 2.29 1.47 0.05 JC 50.22 38.12 5.74 2.07 1.12 0.51 0.41 1.01 0.61 0.02 GP 52.85 35.43 3.95 2.32 2.39 0.72 0.16 1.18 0.75 0.06 表 3 煤样的粒径分布
Table 3 Particle size distribution of fine samples
Sample Volume average particle size d/μm < 10 μm 10-20 μm 20-50 μm 50-80 μm 80-120 μm 120-200 μm σg R2 YN 40 0 8.16 80.98 10.77 0.09 0 1.30 0.967 NM 40 0 5.56 75.22 17.76 1.47 0 1.29 0.973 SF 40 0 6.93 76.03 16.58 0.44 0 1.31 0.976 SH 40 0 2.69 77.37 18.66 1.28 0 1.29 0.968 ZJX 40 0 8.67 75.41 15.49 0.45 0 1.32 0.967 GZ 40 0 4.58 75.08 19.09 1.27 0 1.32 0.964 JC 40 0 10.65 73.51 15.48 0.33 0 1.30 0.969 GP 40 0 5.56 74.12 18.88 1.45 0 1.31 0.971 表 4 筛分煤样的σg和R2
Table 4 σg and R2 of sieved samples
Sample Volume average particle size d/μm σg R2 Sample Volume average particle size d/μm σg R2 NM 40 1.29 0.973 GZ 40 1.32 0.964 90 1.19 0.988 90 1.21 0.980 300 1.30 0.967 310 1.29 0.988 SF 40 1.31 0.976 JC 40 1.30 0.969 100 1.22 0.985 90 1.24 0.976 310 1.29 0.983 300 1.27 0.981 ZJX 40 1.32 0.967 GP 40 1.32 0.971 90 1.20 0.985 90 1.20 0.986 320 1.26 0.982 310 1.24 0.979 表 5 不同煤样的气化反应参数 (1 300 ℃)
Table 5 Reaction parameters of various chars gasification at 1 300 ℃
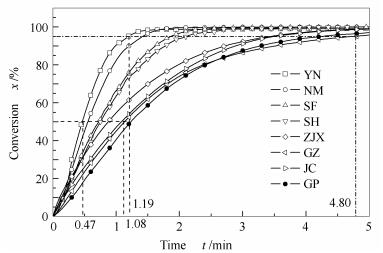
Sample t0.5/min t0.95/min R0.5/min-1 R0.95/min-1 r0.5, R* r0.95, R* YN 0.47 1.19 1.06 0.80 1.00 1.00 NM 0.57 1.42 0.88 0.67 1.21 1.19 SF 0.73 1.87 0.68 0.51 1.55 1.61 SH 0.76 2.12 0.66 0.45 1.62 1.82 ZJX 0.87 3.43 0.57 0.28 1.85 2.88 GZ 1.08 4.80 0.46 0.20 2.30 4.00 JC 1.16 3.60 0.43 0.26 2.47 3.03 GP 1.08 4.27 0.46 0.22 2.30 3.59 *:r0.5, R and r0.95, R is the ratio of the R0.5 and R0.95 of YN char sample to the R0.5 and R0.95 of other char, respectively 表 6 煤焦碳微晶结构参数
Table 6 Crystallite parameters of chars with different coal ranks
Sample d002 LC LC/ d002 NM char 0.373 8 0.850 9 2.276 4 SF char 0.368 1 0.878 5 2.386 8 JC char 0.363 7 1.025 3 2.819 5 表 7 不同煤样的气化反应参数
Table 7 Reaction parameters of chars at different temperatures
Sample t0.5/min t0.95/min R0.5/min R0.95/min r0.5, T* r0.95, T* NM-1 000 ℃ 1.69 4.25 0.30 0.22 3.7 3.2 NM-1 300 ℃ 0.45 1.32 1.11 0.72 SF-1 000 ℃ 2.89 8.22 0.19 0.12 4.0 5.2 SF-1 300 ℃ 0.73 1.87 0.68 0.50 ZJX-1 000 ℃ 11.59 43.49 0.04 0.02 13.3 12.7 ZJX-1 300 ℃ 0.87 3.43 0.57 0.28 JC-1 000 ℃ 21.56 111.42 0.02 0.01 18.4 31.0 JC-1 300 ℃ 1.16 3.60 0.43 0.26 *: r0.5, T and r0.95, T are the ratio of R0.5 and R0.95 at 1 300 ℃ to R0.5 and R0.95 at 1 000 ℃ 表 8 不同煤样的气化反应参数 (1 300 ℃)
Table 8 Reaction parameters of chars with different particle sizes at 1 300 ℃
Sample t0.5/min t0.95/min R0.5/min R0.95/min r0.5, P* r0.95, P* NM-40 μm 0.57 1.42 0.88 0.67 1.18 1.20 NM-300 μm 0.67 1.71 0.75 0.56 SF-40 μm 0.65 1.57 0.77 0.61 1.11 1.15 SF-300 μm 0.72 1.80 0.69 0.53 ZJX-40 μm 0.87 3.43 0.57 0.28 2.13 1.88 ZJX-300 μm 1.85 6.44 0.27 0.15 GZ-40 μm 1.08 4.80 0.46 0.20 5.94 6.55 GZ-300 μm 6.42 31.43 0.08 0.03 JC-40 μm 1.17 3.60 0.43 0.26 5.01 7.38 JC-300 μm 5.86 26.56 0.09 0.04 GP-40 μm 1.08 4.27 0.40 0.22 5.06 6.78 GP-300 μm 5.47 28.95 0.09 0.03 *: r0.5, p and r0.95, p were the ratio of R0.5 and R0.95 of 40 μm chars to R0.5 and R0.95 of 300 μm chars 表 9 不同粒径NM焦和JC焦的比表面积
Table 9 Specific surface area of NM chars and JC chars
Sample 40 μm 100 μm 300 μm NM A/(m2·g-1) 108.5 153.9 149.2 IGR /min-1 1.13 1.19 0.94 JC A/(m2·g-1) 1.96 1.78 1.50 IGR /min-1 0.39 0.29 0.13 表 10 气化反应参数对比
Table 10 Comparison of different kinetic parameters
Sample r0.5, R r0.95, R r0.5, P r0.95, P r0.5, T r0.95, T NM 1.18 1.20 1.27 1.16 3.76 3.22 SF 1.55 1.61 1.11 1.15 3.96 5.24 ZJX 1.85 2.88 2.13 1.88 13.32 12.68 JC 2.47 3.03 5.01 7.38 18.43 30.95 -
[1] FUNG D P C, KIM S D. Laboratory gasification study of Canadian coals:2. Chemical reactivity and coal rank[J]. Fuel, 1983, 62(11):1337-1340. doi: 10.1016/S0016-2361(83)80020-9 [2] MIURA K, HASHIMOTO K, SILVESTON P L. Factors affecting the reactivity of coal chars during gasification, and indices representing reactivity[J]. Fuel, 1989, 68(11):1461-1475. doi: 10.1016/0016-2361(89)90046-X [3] ZHANG L X, HUANG J J, FANG Y T, WANG Y. Gasification reactivity and kinetics of typical Chinese anthracite chars with steam and CO2[J]. Energy Fuels, 2006, 20(3):1201-1210. doi: 10.1021/ef050343o [4] 向银花, 王洋, 张建民, 张守玉, 房倚天, 董众兵.部分气化焦的水蒸气气化动力学[J].化工学报, 2003, 54(3):368-373. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ200303017.htmXIANG Yin-hua, WANG Yang, ZHANG Jian-min, ZHANG Shou-yu, FANG Yi-tian, DONG Zhong-bing. Kinetic on steam gasification of partially gasified char[J]. J Chem Ind Eng (China), 2003, 54(3):368-373. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ200303017.htm [5] LIU H, LUO C, KATO S, UEMIYA S, KANEKO M, KOJIMA T. Kinetics of CO2/Char gasification at elevated temperatures:Part I:Experimental results[J]. Fuel Process Technol, 2006, 87:775-781. doi: 10.1016/j.fuproc.2006.02.006 [6] KOVACIK G, CHAMBERS A, ÖZÜM B. CO2 gasification kinetics of two Alberta coal chars[J]. Can J Chem Eng, 1991, 69(3):811-815. doi: 10.1002/cjce.v69:3 [7] 林善俊, 周志杰, 霍威, 丁路, 于广锁.内扩散对煤和石油焦水蒸气气化反应性能的影响[J].燃料化学学报, 2014, 42(8):905-912. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18464.shtmlLIN Shan-jun, ZHOU Zhi-jie, HUO Wei, DING Lu, YU Guang-suo. Effect of internal diffusion on steam gasification reactivity of coal and petroleum coke[J]. J Fuel Chem Technol, 2014, 42(8):905-912. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18464.shtml [8] HUO W, ZHOU Z J, WANG F C, WANG Y F, YU G S. Experimental study of pore diffusion effect on char gasification with CO2 and steam[J]. Fuel, 2014, 131:59-65. doi: 10.1016/j.fuel.2014.04.058 [9] MATSUI I, KUNII D, FURUSAWA T. Study of char gasification by carbon dioxide 1. Kinetic study by thermogravimetric analysis[J]. Ind Eng Chem Res, 1987, 26(1):91-95. doi: 10.1021/ie00061a017 [10] YONG T K, DONG K S, JUNGHO H. Study of the effect of coal type and particle size on Char-CO2 gasification via gas analysis[J]. Energy Fuels, 2011, 25:5044-5054. doi: 10.1021/ef200745x [11] TAKARADA T, TAMAI Y, TOMITA A. Reactivities of 34 coals under steam gasification[J]. Fuel, 1985, 64(10):1438-1442. doi: 10.1016/0016-2361(85)90347-3 [12] 陆厚根.粉体工程导论[M].上海:同济大学出版社, 1993.LU Hou-gen. Introduction to Powder Technology[M]. Shanghai:Tongji University Press, 1993. [13] 王芳, 曾玺, 王永刚, 余剑, 岳君容, 张建岭, 许光文.微型流化床与热重测定煤焦非等温气化反应动力学对比[J].化工学报, 2015, 66(5):1716-1722. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ201505013.htmWANG Fang, ZENG Xi, WANG Yong-gang, YU Jian, YUE Jun-rong, ZHANG Jian-ling, XU Guang-wen. Comparation of non-isothermal coal char gasification in micro fluidized bed and hermogravimetric analyzer[J]. J Chem Ind Eng (China), 2015, 66(5):1716-1722. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ201505013.htm [14] FENG B, BHATIA S K, BARRY J C. Variation of the crystalline structure of coal char during gasification[J]. Energy Fuels, 2003, 17(3):744-754. doi: 10.1021/ef0202541 [15] DING L U, ZHOU Z J, GUO Q H, WANG Y F, YU G S. In situ analysis and mechanism study of char-ash/slag transition in pulverized coal gasification[J]. Energy Fuels, 2015, 29(6):3532-3544. doi: 10.1021/acs.energyfuels.5b00322 [16] 潘宗林, 李寒旭, 张颂, 赵瑞, 刘铭, 钱宁波.淮南煤Fe2O3催化气化及硅铝对催化的影响[J].煤炭技术, 2015, 34(3):301-303. http://www.cnki.com.cn/Article/CJFDTotal-MTJS201503114.htmPAN Zong-lin, LI Han-xu, ZHANG Song, ZHANG Rui, LIU Ming, QIAN Ning-bo. Catalytic gasification of Huainan coal by using Fe2O3 as catalysts and effect of SiO2 and Al2O3 on catalytic gasification[J]. Coal Technol, 2015, 34(3):301-303. http://www.cnki.com.cn/Article/CJFDTotal-MTJS201503114.htm [17] ÖZTAS N A, YÜRÜM Y. Pyrolysis of turkish zonguldak bituminous coal. Part 1. Effect of mineral matter[J]. Fuel, 2000, 79(10):1221-1227. doi: 10.1016/S0016-2361(99)00255-0 [18] 陈路, 周志杰, 刘鑫, 袁帅, 王辅臣.煤快速热解焦的微观结构对其气化活性的影响[J].燃料化学学报, 2012, 40(6):648-654. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17958.shtmlCHEN Lu, ZHOU Zhi-jie, LIU Xin, YUAN Shuai, WANG Fu-chen. Effect of microstructure of rapid pyrolysis char on its gasification reactivity[J]. J Fuel Chem Technol, 2012, 40(6):648-654. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17958.shtml [19] HUO W, ZHOU Z J, CHEN X L, DAI Z H, YU G S. Study on CO2 gasification reactivity and physical characteristics of biomass, petroleum coke and coal chars[J]. Bioresour Technol, 2014, 159:143-149. doi: 10.1016/j.biortech.2014.02.117 [20] YE D P, AGNEW J B, ZHANG D K. Gasification of a South Australian low-rank coal with carbon dioxide and steam:Kinetics and reactivity studies[J]. Fuel, 1998, 77(11):1209-1219. doi: 10.1016/S0016-2361(98)00014-3 [21] 申峻, 王志忠.不同煤阶煤炭化过程中挥发分组成及微孔变化的研究[J].煤炭学报, 2007, 32(6):626-629. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB200706016.htmSHEN Jun, WANG Zhi-zhong. Study on variation of micro-pores ( < 100 nm) and volatile components of different rank coals during carbonization[J]. J China Coal Soc, 2007, 32(6):626-629. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB200706016.htm -





 下载:
下载: