Preparation and characterization of Ni/TPC catalyst and applied in straw pyrolysis gas reforming
-
摘要: 以废弃汽车外轮胎热解后的副产物轮胎热解焦(Tyre pyrolysis char,TPC)为原料,利用均匀沉淀法制备以轮胎焦为载体的负载型Ni/TPC催化剂,采用EDX、SEM、XRD、TG、BET手段对催化剂进行了表征与分析,同时使用管式炉测试了Ni/TPC催化剂在秸秆热解燃气重整中的催化性能,并考察了热解温度、保温时间、镍负载量及催化时间对秸秆热解燃气重整效果的影响。研究结果表明,TPC富含焦和金属,Ni/TPC催化剂分散均匀,热稳定性好,比表面积为62 m2/g。催化剂活性测试显示,Ni/TPC催化剂用于作物秸秆热解燃气重整具有很强的催化活性,可显著提高燃气中可燃气体含量;热解温度在750℃、保温时间10 min、30%的Ni负载量时Ni/TPC催化剂的催化效率最高,连续使用850 min后,燃气中的H2含量仍相对提高到50%以上,长时间使用后活性结构由Ni3ZnC0.7转变成FeNi3,催化活性依然较强且趋于稳定,TPC可以作为良好的新型镍基催化剂载体。Abstract: Tire pyrolysis char (TPC) was used as a carrier to prepare Ni/TPC catalyst by homogeneous precipitation method. The characteristic of synthetic catalyst was determined by EDX, SEM, XRD, TG and BET. Meanwhile, the performance of Ni/TPC catalyst including reforming temperature, holding time, nickel loading and usage time on the straw pyrolysis gas reforming was investigated in a tube furnace. The results showed that TPC was rich in char and metal. Ni was well loaded on TPC which had a good thermal stability with a specific surface area of 62 m2/g. The Ni/TPC catalyst could obviously improve the burning gas content. The highest catalytic efficiency was obtained at reforming temperature of 750 ℃ and 10 min holding time with 30% Ni loading. The content of H2 in the gas was high and relatively increased by 50% after using the catalyst for 850 min. The Ni3ZnC0.7 active component structure converted to FeNi3 after long-term used with high and stable catalytic activity. TPC had the ability to be a new type of carrier for nickel catalyst.
-
Key words:
- biomass pyrolysis /
- gas reforming /
- carrier /
- Ni catalyst /
- catalyst life
-
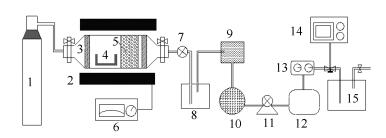
Figure 1 Flow scheme of bench scale reactor for catalytic pyrolysis of straws biomass
1: nitrogen; 2: pyrolysis gasifier and thermocouples; 3: insulation brick; 4: porcelain boat; 5: catalytic bed and catalyst; 6: temperature controller; 7: waterproof valve; 8: water channel; 9: particle filter; 10: gas dryer (silica gel); 11: pump; 12: gas buffer package; 13: flowmeter; 14: gas analyzer; 15: fire prevention
Table 1 Ultimate and proximate analyses of wheat straw sample
Ultimate analysis w/% Proximate analysis w/% C H O N S M A V FC 33.65 4.91 51.88 0.89 0.15 11.15 8.43 78.1 2.69 Table 2 EDX element analysis of three samples
Sample Main composition and content w/% Ni C S Zn Si Ca Fe K TPC - 61.49 15.47 13.94 5.85 1.16 0.93 0.73 30% Ni/TPC 28.44 42.81 11.25 10.46 4.66 0.86 0.71 0.54 Waste Ni/TPC 27.91 43.75 11.44 9.96 4.61 0.82 0.65 0.55 Table 3 BET surface area of TPC and Ni/TPC waste Ni/TPC
Sample TPC Ni/TPC Waste Ni/TPC BET surface area A/(m2·g-1) 84 62 49 -
[1] STIEGEL G J, MAXWELL R C. Gasification technologies:The path to clean, affordable energy in the 21st century[J]. Fuel Process Technol, 2001, 71(1/3):79-97. https://www.deepdyve.com/lp/elsevier/gasification-technologies-the-path-to-clean-affordable-energy-in-the-VjhaZhSPsF [2] WALTERTORRES, PANSARE S, GOODWINJR J. Hot gas removal of tars, ammonia, and hydrogen sulfide from biomass gasification gas[J]. Catal Rev, 2007, 49(4):407-456. doi: 10.1080/01614940701375134 [3] NAZEMI M, PADGETT J, HATZELL M C. Acid/base multi-ion exchange membrane-based electrolysis system for water splitting[J]. Energy Technol, 2017. [4] PALO D R, AND R A D, HOLLADAY J D. Methanol steam reforming for hydrogen production[J]. Chem Rev, 2007, 107(10):3992. doi: 10.1021/cr050198b [5] BULUSHEV D A, ROSS J R H. Catalysis for conversion of biomass to fuels via pyrolysis and gasification:A review[J]. Cataly Today, 2011, 171(1):1-13. doi: 10.1016/j.cattod.2011.02.005 [6] GARC A-D EZ E, GARC A-LABIANO F, DIEGO L F D, ABAD A, GAYÁN P, ADÁNEZ J. Autothermal chemical looping reforming process of different fossil liquid fuels[J]. Int J Hydrogen Energy, 2017, 42(19):13633-13640. doi: 10.1016/j.ijhydene.2016.12.109 [7] BRIDGWATER A V. The technical and economic feasibility of biomass gasification for power generation[J]. Fuel, 1995, 74(5):631-653. doi: 10.1016/0016-2361(95)00001-L [8] HAN J, KIM H. The reduction and control technology of tar during biomass gasification/pyrolysis:An overview[J]. Renewable Sustainable Energy Rev, 2008, 12(2):397-416. doi: 10.1016/j.rser.2006.07.015 [9] HUBER G W, IBORRA S, CORMA A. Synthesis of transportation fuels from biomass:Chemistry, catalysts, and engineering[J]. Chem Rev, 2006, 106(9):4044-4098. doi: 10.1021/cr068360d [10] HE M, XIAO B, HU Z, LIU S M, GUO X J, LUO S Y. Syngas production from catalytic gasification of waste polyethylene:Influence of temperature on gas yield and composition[J]. Int J Hydrogen Energy, 2009, 34(3):1342-1348. doi: 10.1016/j.ijhydene.2008.12.023 [11] YU Q Z, BRAGE C, NORDGREEN T, SJÖSTRÖM K. Effects of Chinese dolomites on tar cracking in gasification of birch[J]. Fuel, 2009, 88(10):1922-1926. doi: 10.1016/j.fuel.2009.04.020 [12] FURUSAWA T, TSUTSUMI A. Comparison of Co/MgO and Ni/MgO catalysts for the steam reforming of naphthalene as a model compound of tar derived from biomass gasification[J]. Appl Catal A:Gen, 2005, 278(2):207-212. doi: 10.1016/j.apcata.2004.09.035 [13] WANG D, YUAN W, JI W. Use of biomass hydrothermal conversion char as the Ni catalyst support in benzene and gasification tar removal[J]. Trans ASABE, 2010, 53(3):795-800. doi: 10.13031/2013.30053 [14] CHOI Y K, CHO M H, KIM J S, LUND H, KAISER M J. Steam/oxygen gasification of dried sewage sludge in a two-stage gasifier:Effects of the steam to fuel ratio and ash of the activated carbon on the production of hydrogen and tar removal[J]. Energy, 2015, 91(suppl 2):160-167. [15] DUC L D, XIAO X, MORISHITA K, TAKARADA T. Biomass gasification using nickel loaded brown coal char in fluidized bed gasifier at relatively low temperature[J]. J Chem Eng Jpn, 2009, 42(1):51-57. doi: 10.1252/jcej.08we218 [16] WANG T J, CHEN Y WU C Z, FU Y, CHANG J. The steam reforming of naphthalene over a nickel-dolomite cracking catalyst[J]. Biomass Bioenergy, 2005, 28(5):508-514. doi: 10.1016/j.biombioe.2004.11.006 [17] WANG D, YUAN W, JI W. Char and char-supported nickel catalysts for secondary syngas cleanup and conditioning[J]. Appl Energy, 2011, 88(5):1656-1663. doi: 10.1016/j.apenergy.2010.11.041 [18] SHEN Y, ZHAO P, SHAO Q, MA D C, TAKAHASHI F, YOSHIKAWA K. In-situ catalytic conversion of tar using rice husk char-supported nickel-iron catalysts for biomass pyrolysis/gasification[J]. Appl Catal B:Environ, 2014, s152/153(1):140-151. [19] MIN Z, ASADULLAH M, YIMSIRI P, SHU Z, WU H W, CHUN Z L. Catalytic reforming of tar during gasification. Part Ⅰ. Steam reforming of biomass tar using ilmenite as a catalyst[J]. Fuel, 2011, 90(5):1847-1854. doi: 10.1016/j.fuel.2010.12.039 [20] ALRAHBI A S, WILLIAMS P T, YAN J. Hydrogen-rich syngas production and tar removal from biomass gasification using sacrificial tire pyrolysis char[J]. Appl Energy, 2017, 190:501-509. doi: 10.1016/j.apenergy.2016.12.099 [21] BAE K W. The role of carbon deposition in the gas phase transesterification of dimethylcarbonate and phenol over TiO2/SiO2 catalyst[J]. Appl Catal A:Gen, 2015, 194(1):403-414. https://www.researchgate.net/publication/225183096_Transesterification_of_Dimethylcarbonate_and_Phenol_Over_Silica_Supported_TiO2_and_Ti-MCM_41_Catalysts_Structure_Insensitivity [22] QIAN K, KUMAR A. Catalytic reforming of toluene and naphthalene (model tar) by char supported nickel catalyst[J]. Fuel, 2017, 187:128-136. doi: 10.1016/j.fuel.2016.09.043 [23] ZHAO Y, LI X, LIU J, WANG C, ZHAO Y. MOF-Derived ZnO/Ni3ZnC0.7/C Hybrids Yolk-Shell microspheres with excellent electrochemical performances for lithium ion batteries[J]. Acs Appl Mater Inter, 2016, 8(10):6472-6480. doi: 10.1021/acsami.5b12562 [24] WANG Y, JIANG L, HU S, SU S, ZHOU Y B, XIANG J, ZHANG S, CHUN Z L. Evolution of structure and activity of char-supported iron catalysts prepared for steam reforming of bio-oil[J]. Fuel Process Technol, 2017, 158:180-190. doi: 10.1016/j.fuproc.2017.01.002 [25] LIU X, XIONG B, HUANG X, DING H R, ZHENGY, LIU Z H, ZHENG C G. Effect of catalysts on char structural evolution during hydrogasification under high pressure[J]. Fuel, 2017, 188(2):474-482. https://www.researchgate.net/publication/309176313_Effect_of_catalysts_on_char_structural_evolution_during_hydrogasification_under_high_pressure [26] BRIDGWATER A V. Renewable fuels and chemicals by thermal processing of biomass[J]. Chem Eng J, 2003, 91(2):87-102. https://www.deepdyve.com/lp/elsevier/renewable-fuels-and-chemicals-by-thermal-processing-of-biomass-O0l7vwwNq3 [27] BRIDGWATER A, GERHAUSER H, EFFENDI A. Biomass pyrolysis process: Australia, 2074192[P]. 2014-05-07. [28] NEGRO M J, MANZANARES P, OLIVA J M, BALLESTEROS I, BALLESTEROS M. Changes in various physical/chemical parameters of Pinus pinaster wood after steam explosion pretreatment[J]. Biomass Bioenergy, 2003, 25(3):301-308. doi: 10.1016/S0961-9534(03)00017-5 [29] TABA L E, IRFAN M F, WAN A M W D, CHAKRABARTI M H. The effect of temperature on various parameters in coal, biomass and CO-gasification:A review[J]. Renewable Sustainable Energy Rev, 2012, 16(8):5584-5596. doi: 10.1016/j.rser.2012.06.015 [30] ANTONAKOU E, DIMITROPOULOS V, LAPPAS A. Production and characterization of bio-oil from catalytic biomass pyrolysis[J]. Therm Sci, 2014, 10(3):151-160. [31] DONALD J, XU C, HASHIMOTO H, BYAMBAJAV E, OHTSUKA Y. Novel carbon-based Ni/Fe catalysts derived from peat for hot gas ammonia decomposition in an inert helium atmosphere[J]. Appl Catal A:Gen, 2010, 375(1):124-133. doi: 10.1016/j.apcata.2009.12.030 [32] ARKATOVA L A. The deposition of coke during carbon dioxide reforming of methane over intermetallides[J]. Catal Today, 2010, 157(1/4):170-176. https://www.deepdyve.com/lp/elsevier/the-deposition-of-coke-from-methane-on-a-ni-mgal-2-o-4-catalyst-qCTK8Ibjae [33] XU L, SONG H, CHOU L. Carbon dioxide reforming of methane over ordered mesoporous NiO-MgO-Al2O3 composite oxides[J]. Appl Catal B:Environ, 2011, s108/109(6):177-190. [34] ZHAO B F, ZHANG X D, LEI C, QU R B, MENG G F, YI X L, LI S. Steam reforming of toluene as model compound of biomass pyrolysis tar for hydrogen.[J]. Biomass Bioenergy, 2010, 34(1):140-144. doi: 10.1016/j.biombioe.2009.10.011 -





 下载:
下载: