Effect of pyrite removal by chromous chloride on organic matter structure in Huadian oil shale
-
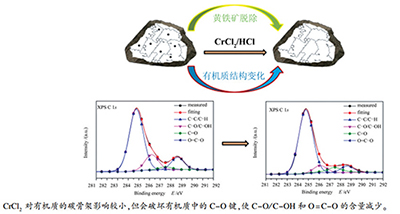
摘要: 采用元素分析、13C NMR、XPS和TG-MS技术考察了氯化亚铬(CrCl2)脱除黄铁矿对桦甸油页岩有机质结构的影响。结果表明,CrCl2可有效脱除有机质中的黄铁矿,脱除率为96.19%。CrCl2对有机质的碳骨架结构影响较小,脱除黄铁矿前后有机质中脂碳、芳碳和羧基/羰基碳的相对含量以及有机质的热解特征温度基本保持不变,但CrCl2可破坏有机质中的C-O键,使C-O/C-OH和O=C-O的含量减少,造成0.98%的有机碳损失和12.54%的有机质损失。CrCl2处理后,有机质的C含量显著增加,H含量稍有增加,O含量显著降低,使得H/C略微降低,O/C明显降低。另外,CrCl2处理后,单位质量有机质中脂碳的含量增加了5.28%,使其热解过程中产生了更多的挥发分,残留的氧化铬对有机质的热分解可能也具有促进作用。Abstract: Influence of pyrite removal by chromous chloride (CrCl2) on structure of organic matter in Huadian oil shale was examined using ultimate analysis, 13C NMR, XPS and TG-MS technology. The results show that CrCl2 treatment leads to 96.19% removal of pyrite from oil shale. The relative contents of aliphatic, aromatic, and carboxyl/carbonyl carbons of organic matter remain about the same level after pyrite removal by CrCl2, so does the pyrolysis characteristic temperature, indicating the carbon skeleton of organic matter is less affected by CrCl2. However, the CrCl2 treatment can break C-O bond in organic matter and reduce content of C-O/C-OH and O=C-O, leading to about 0.98% and 12.54% loss for organic carbon and organic matter, respectively. In addition, carbon content of organic matter significantly increases, hydrogen content slightly increases, and oxygen content is markedly reduced after pyrite removal by CrCl2, resulting in a slight decrease of H/C but a marked decrease of O/C in organic matter. The mass loss of organic matter treated by CrCl2 increases during pyrolysis because aliphatic carbon content of unit mass of organic matter increases 5.28%. Besides, the residual chromium oxide may also promote decomposition of organic matter during pyrolysis.
-
Key words:
- oil shale /
- pyrite /
- chromous chloride /
- organic matter /
- structure
-
表 1 桦甸油页岩的工业分析、元素分析及铝甑分析
Table 1 Proximate, ultimate and Fischer assay analyses of Huadian oil shale
Proximate analysis wad /% Ultimate analysis wad /% Fischer assay wad /% M A V FC C H N S char oil water gas 2.83 70.27 22.30 4.51 16.74 2.33 0.32 0.82 86.97 7.21 2.39 3.37 ad: air dry basis; M: moisture; A: ash; V: volatile matter; FC: fixed carbon 表 2 HD-CF和HD-CFCr的工业分析
Table 2 Proximate analysis of HD-CF and HD-CFCr
Sample Proximate analysis wad/% M A V FC HD-CF 0.93 13.58 64.08 21.41 HD-CFCr 0.25 1.69 75.95 22.11 表 3 HD-CF和HD-CFCr的XRF分析
Table 3 XRF analysis of HD-CF and HD-CFCr
Sample Component wad /% SO3 Fe2O3 F Cr2O3 Al2O3 MgO CaO TiO2 SiO2 Cl others HD-CF 20.30 15.48 20.77 0 18.17 7.73 4.02 1.68 0.87 4.49 6.48 HD-CFCr 44.45 3.91 0 26.04 1.73 0.61 2.58 7.25 2.05 10.58 0.81 表 4 HD-CF和HD-CFCr的灰分组成
Table 4 Ash component of HD-CF and HD-CFCr
Sample wad /% Component wad /% A Fe2O3 Cr2O3 Al2O3 MgO CaO TiO2 SiO2 others HD-CF 13.58 3.86 0 4.53 1.93 1.00 0.42 0.22 1.62 HD-CFCr 1.69 0.15 0.98 0.07 0.02 0.10 0.27 0.08 0.02 表 5 HD-CF和HD-CFCr的元素分析
Table 5 Ultimate analysis of HD-CF and HD-CFCr
Sample Ultimate analysis wdaf/% Molar ratio C H O* N St H/C O/C HD-CF 66.95 8.44 19.91 1.42 3.28 1.51 0.22 HD-CFCr 75.43 9.30 12.65 1.52 1.10 1.48 0.13 *: by difference; daf: dry and ash free basis 表 6 HD-CF和HD-CFCr的结构参数
Table 6 Structural parameters of HD-CF and HD-CFCr
Structural parameter Chemical shift δ Mole percentage /% HD-CF HD-CFCr fal 0-90 74.94 76.31 far 90-165 17.73 16.62 fac 165-220 7.33 7.07 fal: fraction of carbon atoms that are sp3 hybridized (aliphatic); far: fraction of carbon atoms that are sp2 hybridized (aromatic); fac: fraction of carbon atoms that are in carboxyl or carbonyl groups 表 7 HD-CF和HD-CFCr的XPS C 1s分析结果
Table 7 XPS C 1s data of HD-CF and HD-CFCr
Functionality Binding energy E/eV Mole content w/% HD-CF HD-CFCr C-C/C-H 284.75 68.48 76.60 C-O/C-OH 286.15 (±0.05) 16.09 8.01 C=O 287.45 (±0.05) 2.97 8.58 O=C-O 288.65 (±0.05) 12.47 6.81 -
[1] 王擎, 孙斌, 刘洪鹏, 柏静儒, 肖冠华.油页岩热解过程矿物质行为分析[J].燃料化学学报, 2013, 41(2):163-168. doi: 10.3969/j.issn.0253-2409.2013.02.007WANG Qing, SUN Bin, LIU Hong-peng, BAI Jing-ru, XIAO Guan-hua. Analysis of mineral behavior during pyrolysis of oil shale[J]. J Fuel Chem Technol, 2013, 41(2):163-168. doi: 10.3969/j.issn.0253-2409.2013.02.007 [2] 畅志兵, 初茉, 张超, 白书霞, 林浩.桦甸油页岩热解过程中热沥青的组成变化规律[J].燃料化学学报, 2016, 44(11):1310-1317. doi: 10.3969/j.issn.0253-2409.2016.11.005CHANG Zhi-bing, CHU Mo, ZHANG Chao, BAI Shu-xia, LIN Hao. Variation of chemical composition of thermal bitumen during Huadian oil shale pyrolysis[J]. J Fuel Chem Technol, 2016, 44(11):1310-1317. doi: 10.3969/j.issn.0253-2409.2016.11.005 [3] ORENDT A M, PIMIENTA I S, BADU S R, SOLUM M S, PUGMIRE R J, FACELLI J C, WINANS R E. Three-dimensional structure of the Siskin Green River Oil Shale Kerogen Model:A comparison between calculated and observed properties[J]. Energy Fuels, 2013, 27(2):702-710. doi: 10.1021/ef3017046 [4] GUAN X H, LIU Y, WANG D, WANG Q, CHI M S, LIU S, LIU C G. Three-dimensional structure of a huadian oil shale kerogen model:An experimental and theoretical study[J]. Energy Fuels, 2015, 29(7):4122-4136. doi: 10.1021/ef502759q [5] 王擎, 许祥成, 迟铭书, 张宏喜, 崔达, 柏静儒.干酪根组成结构及其热解生油特性的红外光谱研究[J].燃料化学学报, 2015, 43(10):1158-1166. doi: 10.3969/j.issn.0253-2409.2015.10.002WANG Qing, XU Xiang-cheng, CHI Ming-shu, ZHANG Hong-xi, CUI Da, BAI Jing-ru. FT-IR study on composition of oil shale kerogen and its pyrolysis oil generation characteristics[J]. J Fuel Chem Technol, 2015, 43(10):1158-1166. doi: 10.3969/j.issn.0253-2409.2015.10.002 [6] TONG J H, HAN X X, WANG S, JIANG X M. Evaluation of structural characteristics of Huadian oil shale kerogen using direct techniques (solid-state 13C NMR, XPS, FT-IR, and XRD)[J]. Energy Fuels, 2011, 25(9):4006-4013. doi: 10.1021/ef200738p [7] WANG Q, HOU Y C, WU W Z, YU Z, REN S H, LIU Q Y, LIU Z Y. A study on the structure of Yilan oil shale kerogen based on its alkali-oxygen oxidation yields of benzene carboxylic acids, 13C NMR and XPS[J]. Fuel Process Technol, 2017, 166:30-40. doi: 10.1016/j.fuproc.2017.05.024 [8] 畅志兵, 初茉, 张超, 白书霞, 林浩, 马良博.固有碳酸盐和硅酸盐对太姥油页岩热解产物的影响[J].化工学报, 2017, 68(4):1582-1589. http://d.old.wanfangdata.com.cn/Periodical/hgxb201704038CHANG Zhi-bing, CHU Mo, ZHANG Chao, BAI Shu-xia, LIN Hao, MA Liang-bo. Influence of inherent carbonates and silicates on pyrolytic products of Tailao oil shale[J]. J Chem Ind Eng (China), 2017, 68(4):1582-1589. http://d.old.wanfangdata.com.cn/Periodical/hgxb201704038 [9] BALLICE L. Effect of demineralization on yield and composition of the volatile products evolved from temperature-programmed pyrolysis of Beypazari (Turkey) oil shale[J]. Fuel Process Technol, 2005, 86(6):673-690. doi: 10.1016/j.fuproc.2004.07.003 [10] ZHAO X S, ZHANG X L, LIU Z Y, LU Z H, LIU Q Y. Organic matter in Yilan oil shale:Characterization and pyrolysis with or without inorganic minerals[J]. Energy Fuels, 2017, 31(4):3784-3792. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ025855287/ [11] YVRVM Y, DROR Y, LEVY M. Effect of acid dissolution on the mineral matrix and organic matter of Zefa Efe oil shale[J]. Fuel Process Technol, 1985, 11(1):71-86. http://www.sciencedirect.com/science/article/pii/0378382085900177 [12] 迟铭书, 王擎, 李松阳, 刘奇, 查伯宇.酸洗对桦甸油页岩矿物质以及有机结构的影响[J].燃料化学学报, 2017, 45(12):1424-1433. doi: 10.3969/j.issn.0253-2409.2017.12.003CHI Ming-shu, WANG Qing, LI Song-yang, LIU Qi, CHA Bo-yu. Influence of demineralization on minerals and organic structure in Huadian oil shale[J]. J Fuel Chem Technol, 2017, 45(12):1424-1433. doi: 10.3969/j.issn.0253-2409.2017.12.003 [13] LARSEN J W, PAN C S, SHAWVER S. Effect of demineralization on the macromolecular structure of coals[J]. Energy Fuels, 1989, 3(5):557-561. doi: 10.1021/ef00017a004 [14] GAI R H, JIN L J, ZHANG J B, WANG J Y, HU H Q. Effect of inherent and additional pyrite on the pyrolysis behavior of oil shale[J]. J Anal Appl Pyrolysis, 2014, 105:342-347. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=61f70b2f5941d1bf79c4ae5456c1fb22 [15] NEWTON R J, BOTTRELL S H, DEAN S P, HATFIELD D, RAISWELL R. An evaluation of the use of the chromous chloride reduction method for isotopic analyses of pyrite in rocks and sediment[J]. Chem Geol, 1995, 125(3/4):317-320. https://www.sciencedirect.com/science/article/abs/pii/0009254195000987 [16] CANFIELD D E, RAISWELL R, WESTRICH J T, REAVES C M, BERNER R A. The use of chromium reduction in the analysis of reduced inorganic sulfur in sediments and shales[J]. Chem Geol, 1986, 54(1/2):149-155. doi: 10.1016-0009-2541(86)90078-1/ [17] ACHOLLA F V, ORR W L. Pyrite removal from kerogen without altering organic matter:The chromous chloride method[J]. Energy Fuels, 1993, 7(3):406-410. doi: 10.1021/ef00039a012 [18] GALUKHIN A, GERASIMOV A, NIKOLAEV I, NOSOV R, OSIN Y. Pyrolysis of Kerogen of Bazhenov Shale:Kinetics and influence of inherent pyrite[J]. Energy Fuels, 2017, 31(7):6777-6781. [19] 周扬.依兰油页岩分级萃取物的结构研究[D].黑龙江: 黑龙江科技学院, 2011. http://cdmd.cnki.com.cn/Article/CDMD-10219-1011215358.htmZHOU Yang. Study on the structures of sequential extraction of Yi-lan oil-shale[D]. Heilongjiang: Heilongjiang University of Science and Technology, 2011. http://cdmd.cnki.com.cn/Article/CDMD-10219-1011215358.htm [20] LIU Q, HOU Y C, WU W Z, WANG Q, REN S H, LIU Q Y. New insight into the chemical structures of Huadian kerogen with supercritical ethanolysis:Cleavage of weak bonds to small molecular compounds[J]. Fuel Process Technol, 2018, 176:138-145. doi: 10.1016/j.fuproc.2018.03.029 [21] 柏静儒, 王擎, 魏艳珍, 关晓辉.桦甸油页岩的酸洗脱灰[J].中国石油大学学报(自然科学版), 2010, 34(2):150-153. doi: 10.3969/j.issn.1673-5005.2010.02.030BAI Jing-ru, WANG Qing, WEI Yan-zhen, GUAN Xiao-hui. Acid treatment de-ashing of Huadian oilshal[J]. J China Univ Pet (Nat Sci Ed), 2010, 34(2):150-153. doi: 10.3969/j.issn.1673-5005.2010.02.030 [22] CHANG Z B, CHU M, ZHANG C, BAI S X, LIN H, MA L B. Influence of inherent mineral matrix on the product yield and characterization from Huadian oil shale pyrolysis[J]. J Anal Appl Pyrolysis, 2018, 130:269-276. doi: 10.1016/j.jaap.2017.12.022 [23] VANDENBROUCKE M, LARGEAU C. Kerogen origin, evolution and structure[J]. Org Geochem, 2007, 38(5):719-833. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ028056181/ [24] 张晓亮.油页岩有机质结构组成及其热断裂行为研究[D].北京: 北京化工大学, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10010-1015723645.htmZHANG Xiao-liang. Study on sturcture composition and thermal fracture behavior of oil shale organic matter[D]. Beijing: Beijing University of Chemical Technology, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10010-1015723645.htm [25] ABOULKAS A, EL HARFI K. Effects of acid treatments on Moroccan Tarfaya oil shale and pyrolysis of oil shale and their kerogen[J]. J Fuel Chem Technol, 2009, 37(6):659-667. doi: 10.1016/S1872-5813(10)60013-8 [26] 刘建忠, 齐庆杰, 周俊虎, 曹欣玉, 岑可法.煤中氟化物在燃烧产物中的分布特征[J].环境科学, 2003, 24(4):127-130. doi: 10.3321/j.issn:0250-3301.2003.04.025LIU Jian-zhong, QI Qing-jie, ZHOU Jun-hu, CAO Xin-yu, CEN Ke-fa. Distribution of fluoride in the combustion products of coal[J]. Environ Sci, 2003, 24(4):127-130. doi: 10.3321/j.issn:0250-3301.2003.04.025 [27] 蒋旭光, 徐旭, 严建华, 何杰, 池涌, 岑可法.煤燃烧过程中氯析出特性的试验研究[J].煤炭学报, 2002, 27(4):398-401. doi: 10.3321/j.issn:0253-9993.2002.04.014JIANG Xu-guang, XU Xu, YAN Jian-hua, HE Jie, CHI Yong, CEN Ke-fa. Experimental study on the release characteristic of chlorine in coal combustion process[J]. J China Coal Soc, 2002, 27(4):398-401. doi: 10.3321/j.issn:0253-9993.2002.04.014 [28] GELINAS Y, BALDOCK J A, HEDGES J I. Demineralization of marine and freshwater sediments for CP/MAS 13C NMR analysis[J]. Org Geochem, 2001, 32(5):677-693. doi: 10.1016/S0146-6380(01)00018-3 [29] ZHAO X S, LIU Z Y, LU Z H, SHI L, LIU Q Y. A study on average molecular structure of eight oil shale organic matters and radical information during pyrolysis[J]. Fuel, 2018, 219:399-405. doi: 10.1016/j.fuel.2018.01.046 [30] HILLIER J L, FLETCHER T H, SOLUM M S, PUGMIRE R J. Characterization of macromolecular structure of pyrolysis products from a Colorado Green River oil shale[J]. Ind Eng Chem Res, 2013, 52(44):15522-15532. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=03237402c85d458cbb882a4130fdadb3 [31] KOSATEVA A, STEFANOVA M, MARINOV S, CZECH J, CARLEER R, YPERMAN J. Characterization of organic components in leachables from Bulgarian lignites by spectroscopy, chromatography and reductive pyrolysis[J]. Int J Coal Geol, 2017, 183:100-109. doi: 10.1016/j.coal.2017.10.005 [32] LI Q Y, HAN X X, LIU Q Q, JIANG X M. Thermal decomposition of Huadian oil shale. Part 1. Critical organic intermediates[J]. Fuel, 2014, 121:109-116. doi: 10.1016/j.fuel.2013.12.046 [33] 石剑, 李术元, 马跃.爱沙尼亚油页岩及其热解产物的电子顺磁共振研究[J].燃料化学学报, 2018, 46(1):1-7. doi: 10.3969/j.issn.0253-2409.2018.01.001SHI Jian, LI Shu-yuan, MA Yue. Electron paramagnetic resonance (EPR) properties of Estonia oil shale and its pyrolysates[J]. J Fuel Chem Technol, 2018, 46(1):1-7. doi: 10.3969/j.issn.0253-2409.2018.01.001 [34] PAN L W, DAI F Q, HUANG J N, LIU S, LI G Q. Study of the effect of mineral matters on the thermal decomposition of Jimsar oil shale using TG-MS[J]. Thermochim Acta, 2016, 627:31-38. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=49b42da1b671a61ba510f15fa6c1cbbd [35] TORRENTE M C, GALAN M A. Kinetics of the thermal decomposition of oil shale from Puertollano (Spain)[J]. Fuel, 2001, 80(3):327-334. doi: 10.1016/S0016-2361(00)00101-0 -





 下载:
下载: