Catalytic fructose dehydration to 5-hydroxymethylfurfural over sulfonated carbons with hierarchically ordered pores
-
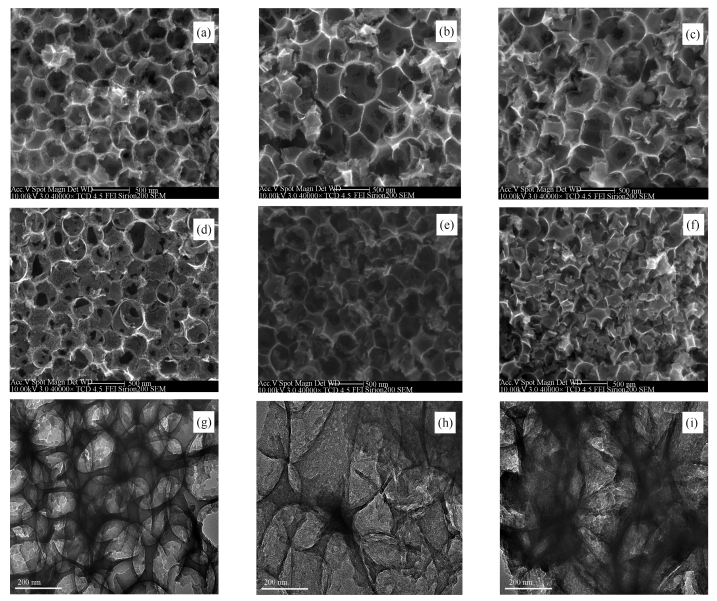
摘要: 采用双模板自组装、炭化、氢氟酸蚀刻和磺化等手段制备了具有分级有序多孔结构的磺化碳(SCHOP),并分别在500、600和700℃考察了炭化温度对分级有序多孔碳微观结构的影响;以催化果糖脱水制备5-羟甲基糠醛(5-HMF)为探针反应,评价了SCHOP的催化效果。结果表明,500℃焙烧所制备的SCHOP具有最高的催化活性。SEM、TEM和N2吸附-脱附表明,所制备的催化剂具有规整的分级有序孔结构,但过高的炭化温度会降低炭材料微观结构的有序性;FT-IR、EDS和-SO3H含量测定表明,通过磺化可在碳基体上有效引入磺酸基,炭化温度过高会降低炭材料的芳香性,不利于磺酸基的引入。130℃下反应20 min,果糖的转化率和5-HMF的收率分别高达96.1%和93.4%,表明SCHOP是一种高效固体酸催化剂。Abstract: Sulfonated carbons with hierarchically ordered pores (SCHOP) were prepared by means of dual template self-assembly, carbonization, HF etching, and sulfonation. The effect of carbonization temperature on microstructure of the prepared carbons as well as the SCHOPs were surveyed at 500-700℃. Using fructose dehydration to 5-hydroxymethylfurfural as a probe reaction, catalytic performance of SCHOPs was examined. The results show that SCHOP-500 carbonized at 500℃ exhibits the highest catalytic activity. SEM and TEM characterization as well as N2 adsorption desorption show that all of the SCHOPs possess hierarchically ordered macropores and mesopores, but high carbonization temperature damages the regularity of carbon materials slightly. FT-IR, EDS and -SO3H determination confirm that sulfonic acid group could be successfully introduced to carbon materials by sulfonation. However, high carbonization temperature lowers aromaticity of the carbon materials, and as a result leading to low sulfonation degree. Under the catalysis of SCHOP-500, as high as 93.4% of 5-HMF yield with 96.1% fructose conversion rate is achieved after reacting at 130℃ for 20 min, indicating that SCHOP-500 is a highly effective and efficient solid acid catalyst.
-
Key words:
- hierarchically ordered /
- sulfonated carbon /
- fructose /
- 5-HMF /
- porous
-
表 1 不同样品的结构和磺酸基含量
Table 1 Specific area and sulfonic acid group content of different samples
Catalyst ABET/(m2·g-1) -SO3H concentration /(mmol·g-1) SCHOP-500 181 1.30 SCHOP-600 382 1.14 SCHOP-700 590 0.64 表 2 基体焙烧温度对催化剂催化果糖脱水性能的影响
Table 2 Effect of the carbonizations temperature of catalyst on the dehydration of fructose
Catalyst Fructose conversion x/% 5-HMF yield w/% 5-HMF selectivity s/% SCHOP-500 96.1 93.4 97.2 SCHOP-600 82.1 78.4 95.5 SCHOP-700 72.5 67.5 93.1 reaction conditions: 1.0 g fructose dissolved in 30 mL DMSO, 0.5 g catalyst, reacted at 130 ℃ for 20 min -
[1] CAI H, LI C, WANG A, ZHANG T. Biomass into chemicals:One-pot production of furan-based diols from carbohydrates via tandem reactions[J]. Catal Today, 2014, 234(10):59-65. https://www.researchgate.net/publication/263092828_Biomass_into_chemicals_One-pot_production_of_furan-based_diols_from_carbohydrates_via_tandem_reactions [2] KAZI F K, PATEL A D, SERRANO-RUIZ J C, DUMESIC J A, ANEX R P. Techno-economic analysis of dimethylfuran (DMF) and hydroxymethylfurfural (HMF) production from pure fructose in catalytic processes[J]. Chem Eng J, 2011, 169(1/3):329-338. https://www.researchgate.net/profile/Akshay_Patel4/publication/251672938_Techno-economic_analysis_of_Dimethylfuran_DMF_and_Hydroxymethylfurfural_HMF_production_from_pure_fructose_in_catalytic_processes/links/55b8f9ff08ae092e965afe40.pdf [3] RAS E J, MAISULS S, HAESAKKERS P, GRUTER G J, ROTHENBERG G. Selective hydrogenation of 5-ethoxymethylfurfural over alumina-supported heterogeneous catalysts[J]. Adv Synth Catal, 2009, 351(18):3175-3185. doi: 10.1002/adsc.v351:18 [4] TONG X, MA Y, LI Y. Biomass into chemicals:Conversion of sugars to furan derivatives by catalytic processes[J]. Appl Catal A:Gen, 2010, 385(1/2):1-13. https://www.researchgate.net/profile/Xinli_Tong2/publication/244109263_Biomass_into_chemicals_Conversion_of_sugars_to_furan_derivatives_by_catalytic_processes/links/5406ee170cf2bba34c1e784a.pdf [5] SCHUETTE H A, THOMAS R W. Normal valerolaction. Ⅲ. Its preparation by the catalytic reduction of levulinic acid with hydrogen in the presence of platinum oxide[J]. Chem Eur J, 2012, 18(17):5256-5260. doi: 10.1002/chem.v18.17 [6] PEDERSEN A T, RINGBORG R, GROTKJÆR T, PEDERSEN S, WOODLEY J M. Synthesis of 5-hydroxymethylfurfural (HMF) by acid catalyzed dehydration of glucose-fructose mixtures[J]. Chem Eng J, 2015, 273(1):455-464. http://orbit.dtu.dk/en/publications/synthesis-of-5hydroxymethylfurfural-hmf-by-acid-catalyzed-dehydration-of-glucosefructose-mixtures(440000fd-fdda-4f99-98d7-c7ede5e41607).html [7] ZHANG J, YU X, ZOU F, ZHONG Y, DU N, HUANG X. Room-temperature ionic liquid system converting fructose into 5-hydroxymethylfurfural in high efficiency[J]. ACS Sustainable Chem Eng, 2015, 3(12):3338-3345. doi: 10.1021/acssuschemeng.5b01015 [8] CHOUDHARY V, MUSHRIF S H, HO C, ANDERKO A, NIKOLAKIS V, MARINKOVIC N S, FRENKEL A I, SANDLER S I, VLACHOS D G. Insights into the interplay of Lewis and Bronsted acid catalysts in glucose and fructose conversion to 5-(hydroxymethyl) furfural and levulinic acid in aqueous media[J]. J Am Chem Soc, 2013, 135(10):3997-4006. doi: 10.1021/ja3122763 [9] KRUGER J S, NIKOLAKIS V, VLACHOS D G. Aqueous-phase fructose dehydration using Brønsted acid zeolites:Catalytic activity of dissolved aluminosilicate species[J]. Appl Catal A:Gen, 2014, 469(2):116-123. http://www.osti.gov/scitech/biblio/1160562-aqueous-phase-fructose-dehydration-using-brnsted-acid-zeolites-catalytic-activity-dissolved-aluminosilicate-species [10] SAMPATH G, KANNAN S. Fructose dehydration to 5-hydroxymethylfurfural:Remarkable solvent influence on recyclability of Amberlyst-15 catalyst and regeneration studies[J]. Catal Commun, 2013, 37(13):41-44. [11] WANG F, WU H Z, LIU C L, YANG R Z, DONG W S. Catalytic dehydration of fructose to 5-hydroxymethylfurfural over Nb2O5 catalyst in organic solvent[J]. Carbohydr Res, 2013, 368(10):78-83. https://www.researchgate.net/profile/Wen-Sheng_Dong/publication/235370674_Catalytic_dehydration_of_fructose_to_5-hydroxymethylfurfural_over_Nb2O5_catalyst_in_organic_solvent/links/54263ca40cf238c6ea778b83.pdf?origin=publication_detail [12] ZAREYEE D, ALIZADEH P, GHANDALI M S, KHALILZADEH M A. Solvent-free acetylation and tetrahydropyranylation of alcohols catalyzed by recyclable sulfonated ordered nanostructured carbon[J]. Chem Pap, 2013, 67(7):713-721. https://www.researchgate.net/publication/257909116_Solvent-free_acetylation_and_tetrahydropyranylation_of_alcohols_catalyzed_by_recyclable_sulfonated_ordered_nanostructured_carbon [13] GALHARDO T S, SIMONE N, GONCALVES M, FIGUEIREDO F C A, MANDELLI D, CARVALHO W A. Preparation of sulfonated carbons from rice husk and their application in catalytic conversion of glycerol[J]. ACS Sustainable Chem Eng, 2013, 1(11):1381-1389. doi: 10.1021/sc400117t [14] YAN L, LIU N, WANG Y, MACHIDA H, QI X. Production of 5-hydroxymethylfurfural from corn stalk catalyzed by corn stalk-derived carbonaceous solid acid catalyst[J]. Bioresour Technol, 2014, 173(22):462-466. https://www.researchgate.net/profile/Xinhua_Qi3/publication/269176565_Production_of_5-hydroxymethylfurfural_from_corn_stalk_catalyzed_by_corn_stalk-derived_carbonaceous_solid_acid_catalyst/links/5485ade90cf24356db610c45.pdf?inViewer=true&disableCoverPage=true&origin=publication_detail [15] KARIMI B, MIRZAEI H M, BEHZADNIA H, VALI H. Novel ordered mesoporous carbon based sulfonic acid as an efficient catalyst in the selective dehydration of fructose into 5-HMF:The role of solvent and surface chemistry[J]. ACS Appl Mater Inter, 2015, 7(34):19050-19059. doi: 10.1021/acsami.5b03985 [16] HU L, TANG X, WU Z, LIN L, XU J, XU N, DAI B. Magnetic lignin-derived carbonaceous catalyst for the dehydration of fructose into 5-hydroxymethylfurfural in dimethylsulfoxide[J]. Chem Eng J, 2015, 263(2):299-308. https://www.researchgate.net/publication/268576456_Magnetic_lignin-derived_carbonaceous_catalyst_for_the_dehydration_of_fructose_into_5-hydroxymethylfurfural_in_dimethylsulfoxide [17] ZHAO J, ZHOU C, HE C, DAI Y, JIA X, YANG Y. Efficient dehydration of fructose to 5-hydroxymethylfurfural over sulfonated carbon sphere solid acid catalysts[J]. Catal Today, 2015, 264(4):123-130. https://www.researchgate.net/publication/281105087_Efficient_dehydration_of_fructose_to_5-hydroxymethylfurfural_over_sulfonated_carbon_sphere_solid_acid_catalysts [18] ORDOMSKY V V, SCHOUTEN J C, SCHAAF J V D, NIJHUIS T A. Foam supported sulfonated polystyrene as a new acidic material for catalytic reactions[J]. Chem Eng J, 2012, 207-208(5):218-225. https://www.researchgate.net/publication/257566004_Foam_supported_sulfonated_polystyrene_as_a_new_acidic_material_for_catalytic_reactions [19] XIA Y, YANG Z, GOU X, ZHU Y. A simple method for the production of highly ordered porous carbon materials with increased hydrogen uptake capacities[J]. Int J Hydrogen Energy, 2013, 38(12):5039-5052. doi: 10.1016/j.ijhydene.2013.02.037 [20] YUN Y S, PARK D J, MIN J J, JIN H J. 3-D ordered bimodal porous carbon/nickel oxide hybrid electrodes for supercapacitors[J]. Synth Met, 2013, 177(8):105-109. https://www.researchgate.net/publication/270949057_3-D_ordered_bimodal_porous_carbonnickel_oxide_hybrid_electrodes_for_supercapacitors [21] ZHANG T, ZHANG Q, GE J, GOEBL J, SUN M, YAN Y, LIU Y S, CHANG C, GUO J, YIN Y. A self-templated route to hollow silica microspheres[J]. J Phys Chem C, 2008, 113(8):3168-3175. http://www.wenkuxiazai.com/doc/4efb3fafd1f34693daef3e1f.html [22] ZHENG F C, CHEN Q W, HU L, YAN N, KONG X K. Synthesis of sulfonic acid-functionalized Fe3O4@C nanoparticles as magnetically recyclable solid acid catalysts for acetalization reaction[J]. Dalton Trans, 2014, 43(3):1220-1227. doi: 10.1039/C3DT52098F [23] ZHAO Q, WANG X, LIU J, WANG H, ZHANG Y, GAO J, LU Q, ZHOU H. Design and synthesis of three-dimensional hierarchical ordered porous carbons for supercapacitors[J]. Electrochim Acta, 2015, 154(4):110-118. https://www.researchgate.net/publication/270007967_Design_and_synthesis_of_three-dimensional_hierarchical_ordered_porous_carbons_for_supercapacitors [24] XUE C, TU B, ZHAO D. Facile fabrication of hierarchically porous carbonaceous monoliths with ordered mesostructure via an organic organic self-assembly[J]. Nano Res, 2009, 2(3):242-253. doi: 10.1007/s12274-009-9022-y [25] ZHOU W, YOSHINO M, HIDETOSHI KITA H, OKAMOTO K. Carbon molecular sieve membranes derived from phenolic resin with a pendant sulfonic acid group[J]. Ind Eng Chem Res, 2001, 40(22):4801-4807. doi: 10.1021/ie010402v [26] CEPEDA-JIMENEZ C M, PASTOR-BLAS M M, FERRANDIZ-GOMEZ T P, MARTIN-MARTINNEZ J M. Influence of the styrene content of thermoplastic styrene-butadiene rubbers in the effectiveness of the treatment with sulfuric acid[J]. Int J Adhes Adhes, 2001, 21(2):161-172. doi: 10.1016/S0143-7496(00)00048-8 [27] OKAMURA M, TAKAGAKI A, TODA M, KONDO J N, DOMEN K, TATSUMI T, HARA M, HAYASHI S. Acid-catalyzed reactions on flexible polycyclic aromatic carbon in amorphous carbon[J]. Chem Mater, 2006, 18(13):3039-3045. doi: 10.1021/cm0605623 [28] SHIMIZU K I, UOZUMI R, SATSUMA A. Enhanced production of hydroxymethylfurfural from fructose with solid acid catalysts by simple water removal methods[J]. Catal Commun, 2009, 10(14):1849-1853. doi: 10.1016/j.catcom.2009.06.012 [29] AMARASEKARA A S, WILLIAMS L T D, EBEDE C C. Mechanism of the dehydration of D-fructose to 5-hydroxymethylfurfural in dimethyl sulfoxide at 150 degrees C:An NMR study[J]. Carbohydr Res, 2008, 343(18):3021-3024. doi: 10.1016/j.carres.2008.09.008 -





 下载:
下载: