Effect of citric acid addition on MoO3/CeO2-Al2O3 catalyst for sulfur-resistant methanation
-
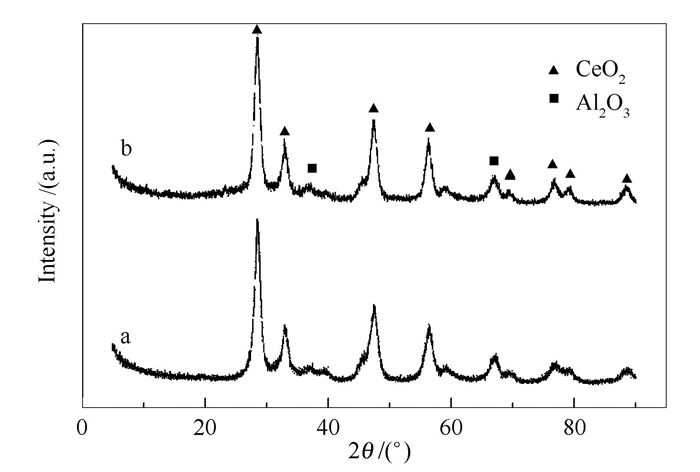
摘要: 将柠檬酸(CA)作为络合剂添加至CeO2-Al2O3复合载体中,并考察了CA对MoO3/CeO2-Al2O3催化剂耐硫甲烷化性能的影响。活性评价结果显示,催化剂活性随柠檬酸添加量的增大而增大,当n(CA)/n(Ce)为3时,CO转化率可达60%。催化剂BET、XRD、H2-TPR及XPS等表征结果表明,在CeO2-Al2O3复合载体中加入CA,可以增大载体及催化剂的比表面积,使Mo物种分散性提高。同时,CA对Ce物种起络合作用,致使催化剂表面Ce元素含量明显增加,进而减弱了活性组分Mo物种与载体间相互作用力,并最终导致了催化剂活性的提升。Abstract: Citric acid hold great promise to improve the Mo-based catalyst performance for hydrogenation reaction applications. MoO3/CeO2-Al2O3 catalysts were prepared by impregnation method with adding citric acid into CeO2-Al2O3 composite supports and tested for sulfur resistant methanation. The syngas methanation activity increased with the increase of citric acid additive amount, and CO conversion could reach up 60% when the molar ratio of citric acid to Ce was 3. The prepared catalysts were characterized by BET, H2-TPR, XRD and XPS. The increased catalytic performance was mainly attributed to the increased amount of Ce species on the surface of catalysts which could decreased the interaction force between MoO3 and CeO2-Al2O3 supports. Additionally, the increased specific surface of CeO2-Al2O3 composite support was also in favor of catalytic performance.
-
Key words:
- synthetic natural gas /
- methanation /
- CeO2 /
- citric acid
-
Table 1 Textural properties of CeAl supports with/without the addition of citric acid and their corresponding catalysts
Support BET surface area
A/(m2·g-1)Pore size d/nm CeO2 diameter
D/nmCatalyst BET surface area
A/(m2·g-1)Pore size d/nm CeAl 97 13.6 7.7 Mo/CeAl 76 13.0 CeAl-1CA 117 9.6 7.5 Mo/CeAl-1CA 89 10.2 -
[1] KUSTOV A L,FREY A M,LARSEN K E,JOHANNESSEN T,NØRSKOV J K,CHRISTENSEN C H.CO methanation over supported bimetallic Ni-Fe catalysts:From computational studies towards catalyst optimization[J].Appl Catal A:Gen,2007,320(3):98-104. http://www.sciencedirect.com/science/article/pii/S0926860X06008969 [2] KOPYSCINSKI J,SCHILDHAUER T J,BIOLLAZ S M A.Production of synthetic natural gas (SNG) from coal and dry biomass-A technology review from 1950 to 2009[J].Fuel,2010,89(8):1763-1783. doi: 10.1016/j.fuel.2010.01.027 [3] HACATOGLU K,JAMES MCLELLAN P,LAYZELL D B.Production of bio-synthetic natural gas in Canada[J].Environ Sci Technol,2010,44(6):2183-2188. doi: 10.1021/es901561g [4] ZAHRADNIK R L,GLENN R A.Direct methanation of coal[J].Fuel,1971,50(1):77-90. doi: 10.1016/S0016-2361(71)81022-0 [5] MOELLER F W,ROBERTS H,BRITZ B.Methanation of coal gas for SNG[J].Hydrocarbon Process,1974,53(4):69-74. https://www.researchgate.net/publication/283158569_METHANATION_OF_COAL_GAS_FOR_SNG [6] VEEN G V,KRUISSINK E C,DOESBURG E B M,ROSS J R H,REIJEN L L V.The effect of preparation conditions on the activity and stability of copreciptitated Ni/Al2O3 catalysts for the methanation of carbon monoxide[J].React Kinet Catal Lett,1978,9(2):143-148. doi: 10.1007/BF02068914 [7] GALLAGHER J E,EUKER C A.Catalytic coal gasification for SNG manufacture[J].Int J Energy Res,1980,4(2):137-147. doi: 10.1002/(ISSN)1099-114X [8] FRANK A J,DICK H A,GORAL J,NELSON A J,GRÄTZEL M.MoS2-catalyzed methanation of CO with H2S[J].J Catal,1990,126(2):674-676. doi: 10.1016/0021-9517(90)90030-N [9] CHRISTENSEN J M,MORTENSEN P M,TRANE R,JENSEN P A,JENSEN A D.Effects of H2S and process conditions in the synthesis of mixed alcohols from syngas over alkali promoted cobalt-molybdenum sulfide[J].Appl Catal A:Gen,2009,366(1):29-43. doi: 10.1016/j.apcata.2009.06.034 [10] KIM M Y,HA S B,DONG J K,BYUN C,PARK E D.CO methanation over supported Mo catalysts in the presence of H2S[J].Catal Commun,2013,35(17):68-71. [11] SASAKI T,SUZUKI T.Sulfide molybdenum catalysts for water-gas shift reaction:Influence of the kind of promoters and supports to generate MoS2[J].Appl Catal A:Gen,2014,484(10):79-83. [12] WANG B W,DING G Z,SHANG Y G,LV J,WANG H Y,WANG E D,LI Z H,MA X B,QIN S D,SUN Q.Effects of MoO3 loading and calcination temperature on the activity of the sulphur-resistant methanation catalyst MoO3/γ-Al2O3[J].Appl Catal A:Gen,2012,431-432:144-150. doi: 10.1016/j.apcata.2012.04.029 [13] TRIKI M,KSIBI Z,GHORBEL A,MEDINA F.Preparation and characterization of CeO2-Al2O3 aerogels supported ruthenium for catalytic wet air oxidation of p-hydroxybenzoic acid[J].J Sol-Gel Sci Technol,2011,59(1):1-6. doi: 10.1007/s10971-011-2452-5 [14] DAMYANOVA S,PEREZ C A,SCHMAL M,BUENO J M C.Characterization of ceria-coated alumina carrier[J].Appl Catal A:Gen,2002,234(1/2):271-282. https://www.researchgate.net/profile/JMC_Bueno/publication/229168026_Characterization_of_ceria-coated_alumina_carrier/links/00b7d52bcc8d13e338000000.pdf?inViewer=true&disableCoverPage=true&origin=publication_detail [15] YAO H C,YAO Y F Y.Ceria in automotive exhaust catalysts:I.Oxygen storage[J].J Catal,1984,86(2):254-265. doi: 10.1016/0021-9517(84)90371-3 [16] ZHUANG Q,QIN Y,CHANG L.Promoting effect of cerium oxide in supported nickel catalyst for hydrocarbon steam-reforming[J].Appl Catal,1991,70(1):1-8. doi: 10.1016/S0166-9834(00)84149-4 [17] WANG B W,SHANG Y G,DING G Z,LÜ J,WANG H Y,WANG E D,LI Z H,MA X B,QIN S D,SUN Q.Effect of the ceria-alumina composite support on the Mo-based catalyst's sulfur-resistant activity for the synthetic natural gas process[J].Reac Kinet,Mech Cat,2012,106(2):495-506. doi: 10.1007/s11144-012-0452-2 [18] CATTANEO R,WEBER T,SHIDO T,PRINS R.A quick EXAFS study of the sulfidation of NiMo/SiO2 hydrotreating catalysts prepared with chelating ligands[J].J Catal,2000,191(1):225-236. doi: 10.1006/jcat.1999.2784 [19] IWAMOTO R,KAGAMI N,ⅡNO A.Effect of polyethylene glycol addition on hydrodesulfurization activity over CoO-MoO3/Al2O3 catalyst[J].J Jpn Pet Inst,2005,48(4):237-242. doi: 10.1627/jpi.48.237 [20] RINALDI N,KUBOTA T,OKAMOTO Y.Effect of citric acid addition on Co-Mo/B2O3/Al2O3 catalysts prepared by a post-treatment method[J].Ind Eng Chem Res,2009,48(23):10414-10424. doi: 10.1021/ie9008343 [21] LI H F,LI M F,CHU Y,LIU F,NIE H.Essential role of citric acid in preparation of efficient NiW/Al2O3 HDS catalysts[J].Appl Catal A:Gen,2011,403(1/2):75-82. http://www.doc88.com/p-1931584301169.html [22] CASTILLO-VILLALÓN P,RAMIREZ J,VARGAS-LUCIANO J A.Analysis of the role of citric acid in the preparation of highly active HDS catalysts[J].J Catal,2014,320(1):127-136. https://www.researchgate.net/publication/267456000_Analysis_of_the_role_of_citric_acid_in_the_preparation_of_highly_active_HDS_catalysts [23] BERGWERFF J A,JANSEN M,LELIVELD B G,VISSER T,JONG K P D,WECKHUYSEN B M.Influence of the preparation method on the hydrotreating activity of MoS2/Al2O3 extrudates:A Raman microspectroscopy study on the genesis of the active phase[J].J Catal,2006,243(2):292-302. doi: 10.1016/j.jcat.2006.07.022 [24] RINALDI N,KUBOTA T,OKAMOTO Y.Effect of citric acid addition on the hydrodesulfurization activity of MoO3/Al2O3 catalysts[J].Appl Catal A:Gen,2010,374(1/2):228-236. [25] WANG R,SMITH K J.Hydrodesulfurization of 4,6-dimethyldibenzothiophene over high surface area metal phosphides[J].Appl Catal A:Gen,2009,361(1):18-25. https://www.researchgate.net/publication/239691379_Hydrodesulfurization_of_46-dimethyldibenzothiophene_over_high_surface_area_metal_phosphides [26] SURESH R,PONNUSWAMY V,MARIAPPAN R.Effect of annealing temperature on the microstructural,optical and electrical properties of CeO2 nanoparticles by chemical precipitation method[J].Appl Surf Sci,2013,273(273):457-464. https://www.researchgate.net/publication/257032897_Effect_of_annealing_temperature_on_the_microstructural_optical_and_electrical_properties_of_CeO2_nanoparticles_by_chemical_precipitation_method [27] LIANG C X,LI X Y,QU Z P,TADE M,LIU S M.The role of copper species on Cu/γ-Al2O3 catalysts for NH3-SCO reaction[J].Appl Surf Sci,2012,258(8):3738-3743. doi: 10.1016/j.apsusc.2011.12.017 [28] MESTL G,SRINIVASAN T K K,KNOEZINGER H.Mechanically activated MoO3.3.Characterization by vibrational spectroscopy[J].Langmuir,2002,11(10):3795-3804. [29] MESTL G,SRINIVASAN T K K.Raman spectroscopy of monolayer-type catalysts:Supported molybdenum oxides[J].Cat Rev,1998,40(4):451-570. doi: 10.1080/01614949808007114 [30] ABERUAGBA F,KUMAR M,MURALIDHAR G,SHARMA L D.Characterization of Al2O3-ZrO2 mixed oxide supported Mo hydrotreating catalyst[J].Pet Sci Technol,2004,22(9):1287-1298. [31] ZAKI M I,VIELHABER B,KNOEZINGER H.Low-temperature carbon monoxide adsorption and state of molybdena supported on alumina,titania,ceria,and zirconia.An infrared spectroscopic investigation[J].J Phys Chem,1986,90(14):3176-3183. doi: 10.1021/j100405a026 [32] BERIT H,NØRSKOV J K,HENRIK T E.A density functional study of the chemical differences between Type I and Type Ⅱ MoS2-based structures in hydrotreating catalysts[J].J Phys Chem B,2005,109(6):2245-2253. doi: 10.1021/jp048842y -





 下载:
下载: