Adsorption and mass transfer behavior of benzene on hierarchically structured Y by acid-base treatment
-
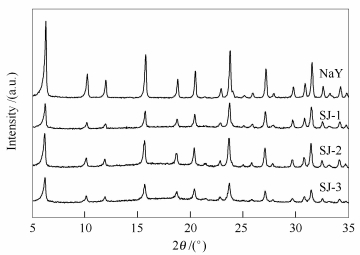
摘要: 利用H4EDTA-NaOH共处理的方法制备了具有不同孔径分布的多级微-介孔NaY分子筛。运用XRD、N2吸附、SEM、TEM对其结构进行了表征。采用频率响应(FR) 和智能重量分析仪(IGA) 技术研究了苯在改性后的多级孔NaY分子筛及微孔NaY分子筛上的吸附和传质性能。结果表明, 适当的酸碱处理不会改变分子筛的晶体结构, 但可调变NaY分子筛的精细结构; 介孔的引入降低了分子在孔道中的扩散阻力, 较大的孔径和较好的孔道贯通性有利于扩散和吸附中心的可接近性; 对于微孔NaY分子筛, 苯在分子筛上的吸附过程为其传质过程的速控步骤, 对于酸碱处理的多级孔NaY分子筛, 分子筛颗粒中微/介孔内的扩散过程及分子筛微-介孔孔道间的分子交换过程是传质过程的速控步骤。Abstract: A series of hierarchical NaY zeolites with different pore size distribution were synthesized by sequential treatment with ethylenediaminetetraacetic acid and NaOH. The structural properties of the as-synthesized hierarchical NaY zeolites were characterized by X-ray diffraction, N2 sorption, SEM and TEM. The adsorption and diffusion performances of benzene on NaY and hierarchical NaY zeolites were studied by frequency response (FR) and intelligent gravimetric analyser (IGA). It was found that the structure of NaY zeolite was remained by suitable acid and base treatment, but the connectivity in the pores was finely tailored. Mesoporous was introduced to reduce the molecular diffusion resistance in the pores, while the larger pore size and connectivity in the mesoporous was more beneficial to diffusion and accessibility of activity site. The adsorption of benzene on NaY was found to be the rate-controlling step. While, the limiting steps for the overall mass transfer processes in the hierarchical zeolites are the diffusion process in the length scale of micropore/mesopore and the mass transfer at the micropore-mesopore molecular exchange rather than the intracrystalline diffusion one in the micropores of the zeolite crystals.
-
Key words:
- hierarchical porous Y /
- benzene /
- frequency response /
- adsorption and diffusion
-
表 1 NaY、SJ-1、SJ-2和SJ-3的织构性质
Table 1 Texture properties data of NaY, SJ-1, SJ-2 and SJ-3 zeolites
Sample daver/nm ABET/(m2·g-1) Ameso/(m2·g-1) Amic/(m2·g-1) vp/(m3·g-1) vmicro/(m3·g-1) vmeso/(m3·g-1) NaY 3.12 647 49 598 0.352 0.313 0.039 SJ-1 8.68 709 147 562 0.666 0.296 0.37 SJ-2 8.54 688 109 579 0.563 0.304 0.259 SJ-3 9.44 556 143 413 0.608 0.217 0.391 表 2 室温下苯分子在NaY、SJ-1、SJ-2和SJ-3分子筛上的扩散时间常数
Table 2 Diffusion time constants of benzene on NaY, SJ-1, SJ-2 and SJ-3 zeolite
D/r2(s-1) NaY SJ-1 SJ-2 SJ-3 6.17×10-7 1.61×10-6 2.07×10-6 1.41×10-6 -
[1] PARK D H, KIM S S, WANG H, PINAVAIA T J, PAPAPETROU M C, LAPPAS A A, TRIANTAFYLLIDIS K S. Selective petroleum refining over a zeolite catalyst with small intracrystal mesopores[J]. Angew Chem Int Ed Eng, 2009, 48(41): 7645-7648. doi: 10.1002/anie.v48:41 [2] FLEISCH T H, MEYERS B L, RAY G J, HALL J B, MARSHALL C L. Hydrothermal dealumination of faujasites[J]. J Catal, 1986, 99(86): 117-125. http://www.sciencedirect.com/science/article/pii/0021951786902058 [3] WANG Q L, GIANNETTO G, TORREALBA M, PEROT G, KAPPENSTEIN C, GUISNET M. Dealumination of zeolites Ⅱ. Kinetic study of the dealumination by hydrothermal treatment of a NH4NaY zeolite[J]. J Catal, 1991, 130(2): 459-470. doi: 10.1016/0021-9517(91)90128-Q [4] 刘兴云, 张旭政, 李宣文. NaY沸石草酸脱铝[J].高等学校化学学报, 1997, 18(3): 342-347. http://www.cnki.com.cn/Article/CJFDTOTAL-GDXH199703039.htmLIU Xing-yun, ZHANG Xu-zheng, LI Xuan-wen. NaY zeolite acid dealumination[J]. Chem Res Chin Univ, 1997, 18(3): 342-347. http://www.cnki.com.cn/Article/CJFDTOTAL-GDXH199703039.htm [5] PU X, SHI L, LIU N W. Acid properties and catalysis of USY zeolite with different extra-framework aluminum concentration[J]. Microporous Mesoporous Mater, 2015, 201: 17-23. doi: 10.1016/j.micromeso.2014.08.056 [6] QIN Z X, SHEN B J, YU Z W, DENG F, ZHAO L, ZHOU S G, YUAN D L, GAO X H, WANG B J, ZHAO H J, LIU H H. A defect-based strategy for the preparation of mesoporous zeolite Y for high-performance catalytic cracking[J]. J Catal, 2013, 298: 102-111. doi: 10.1016/j.jcat.2012.11.023 [7] VERBOEKEND D, KELLER T C, MITCHELL S, PEREZ-RAMIREZ J. Hierarchical FAU-and LTA-type zeolites by post-synthetic design: A new generation of highly efficient base catalysts[J]. Adv Funct Mater, 2013, 23(15): 1923-1934. doi: 10.1002/adfm.v23.15 [8] VERBOEKEND D, VILE G, PEREZ-RAMIREZ J. Hierarchical Y and USY zeolites designed by post-synthetic strategies[J]. Adv Funct Mater, 2012, 22(5): 916-928. doi: 10.1002/adfm.v22.5 [9] TIAN F P, SHEN Q C, FU Z K, WU Y H, JIA C Y. Enhanced adsorption desulfurization performance over hierarchically structured zeolite Y[J]. Fuel Process Technol, 2014, 128: 176-182. doi: 10.1016/j.fuproc.2014.07.018 [10] SUN H Y, SUN L P, LI F, ZHANG L. Adsorption of benzothiophene from fuels on modified NaY zeolites[J]. Fuel Process Technol, 2015, 134: 284-289. doi: 10.1016/j.fuproc.2015.02.010 [11] ZHAO J, YIN Y C, LI Y, CHEN W Y, LIU B J. Synthesis and characterization of mesoporous zeolite Y by using block copolymers as templates[J]. Chem Eng J, 2016, 284: 405-411. doi: 10.1016/j.cej.2015.08.143 [12] KORTUNOV P, VASENKOV S, KARGER J, VALIULLIN R, GOTTSCHALK P, ELIA MF, PEREZ M, STOCKER M, DRESCHER B, MCELHINEY G, BERGER C, GLASER R, WEITKAMP J. The role of mesopores in intracrystalline transport in USY zeolite: PFG NMR diffusion study on various length scales[J]. J Am Chem Soc, 2005, 127(37): 13055-13059. doi: 10.1021/ja053134r [13] QIN Z X, SHEN B J, GAO X H, LIN F, WANG B J, XU C M. Mesoporous Y zeolite with homogeneous aluminum distribution obtained by sequential desilication-dealumination and its performance in the catalytic cracking of cumene and 1, 3, 5-triisopropylbenzene[J]. J Catal, 2011, 278(2): 266-275. doi: 10.1016/j.jcat.2010.12.013 [14] BOURDIN V, GRAY P G, GRENIER P, TERRIER M F. An apparatus for adsorption dynamics studies using infrared measurement of the adsorbent temperature[J]. Rev Sci Instrum, 1998, 69(5): 2130-2136. doi: 10.1063/1.1148911 [15] SONG L J, SUN Z L, REES L V C. Studies of adsorption, diffusion and molecular simulation of cyclic hydrocarbons in MFI zeolites[J]. Stud Surf Sci Catal, 2001, 135: 3064-3072. -





 下载:
下载: