Effect of O2/CO2 combustion atmosphere on the mineral inter-reaction of blended coal ashes
-
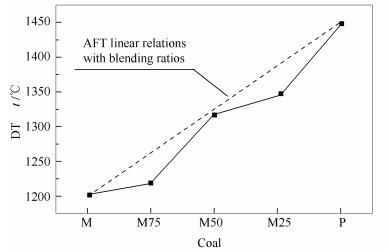
摘要: 对蒙煤与平七煤两种单煤及其按照不同比例组成的混煤,分别在O2/CO2和O2/N2气氛下,采用管式炉燃烧制取灰样;对灰样进行灰熔点、XRD及同步热分析(TG/DSC)测试,并进行相关热力学计算,分析了O2/CO2燃烧方式对混煤灰中矿物质间反应的影响。结果表明,常规灰熔点测试方法测得的两种气氛下的混煤灰熔点没有明显差别。O2/CO2气氛促进了煤灰/混煤灰中钙的碳酸化,且明显抑制了高温下CaCO3的分解。气氛的改变影响了含钙矿物的转化,进而影响了混煤中钙与莫来石反应生成低温共熔物;O2/CO2气氛下钙更易于与莫来石发生反应生成低温共熔物,从而会增加结渣倾向。当混煤中蒙煤比例达到或大于75%时,随着蒙煤比例的逐渐增加,莫来石含量减少,O2/CO2气氛对钙与莫来石之间的反应影响减弱,但对含铁矿物的影响更加明显,使其更易于生成含铁玻璃体,从而也会增加结渣倾向。Abstract: The mineral inter-reaction of blending coals during O2/CO2 combustion was studied. Two coals, Meng coal and Pingqi coal, were blended by certain ratios. The blending coals were burned in a tube furnace with O2/CO2 or O2/N2. Ash fusion temperature test, XRD, TG/DSC and thermodynamic calculation were employed to examine the melting behavior and mineral reactions of blending coal ashes during O2/CO2 and O2/N2 combustion in detail. The results show that:there is no a pronounced difference in the blending coal ash fusion temperature between O2/CO2 and O2/N2 combustion. More CaCO3 produced during O2/CO2 combustion suggests that O2/CO2 atmosphere significantly prevents the decomposition of CaCO3. The changing of atmosphere has an impact on the transformation of Ca-containing minerals, and the reaction between Ca and mullite occurs significantly, which is easier in O2/CO2 combustion to produce more low-melting phase that will aggravate the boiler slagging. When the blending ratio of Meng coal in blends with Pingqi coal is 75% or more, less mullite is present in blending coals, and thus the impact of atmosphere on Ca-mullite reaction is weaker. However, the atmosphere has a more impact on Fe-containing minerals and more Fe-glass phase will be formed during O2/CO2 combustion, which will aggravate the boiler slagging.
-
Key words:
- pulverized coal /
- coal blending /
- O2/CO2 atmosphere /
- mineral transformation /
- boiler slagging
-
表 1 实验用煤的煤质特性
Table 1 Properties of coal samples
Coal sample Ultimate analysis wd/% Proximate analysis w/% Q (net, ar) /(kJ·kg-1) C H N S O Mad Ad Vd FCd Meng coal 71.23 4.80 0.75 0.49 15.19 3.51 7.54 34.07 58.39 21543 Pingqi coal 62.06 4.50 0.87 0.54 10.61 1.42 21.42 30.28 48.30 21505 表 2 煤灰成分分析
Table 2 Ash composition of coal samples
Coal Composition w/% Na2O Al2O3 SiO2 SO3 K2O CaO Fe2O3 MgO TiO2 Meng coal 2.88 13.74 32.03 22.31 0.71 19.83 5.55 0.71 0.95 Pingqi coal 1.47 34.13 50.29 4.27 0.80 3.26 1.97 1.80 1.04 表 3 HSC模拟计算工况
Table 3 Cases of HSC calculation
Case 1 (O2/N2 atmosphere) Case 2(O2/CO2 atmosphere) Reactants SiO2 Al2O3 CaSO4 SiO2 Al2O3 CaCO3 CaSO4 Amount /kmol 5.22 1.32 3.46 5.22 1.32 2.08 1.38 Products 3Al2O3·2SiO2、CaO·Al2O3·2SiO2
2CaO·Al2O3·SiO2、CaO、SO3(g)3Al2O3·2SiO2、CaO·Al2O3·2SiO2
2CaO·Al2O3·SiO2、
CaO、SO3(g)、CO2(g) -
[1] 刘燕华, 葛全胜, 何凡能, 程邦波.应对国际CO2减排压力的途径及我国减排潜力分析[J].地理学报, 2008, 63(7):675-682. doi: 10.11821/xb200807001LIU Yan-hua, GE Quan-sheng, HE Fan-neng, CHENG Bang-bo. Countermeasures against international pressure of reducing CO2 emissions and analysis on China's potential of CO2 emission reduction[J]. Acta Geog Sin, 2008, 63(7):675-682. doi: 10.11821/xb200807001 [2] WALL T, LIU Y, SPERO C, ELLIOTT L, KHARE S, RATHNAM R, ZEENATHAL F, MOGHTADERI B, BUHRE B, SHENG C, GUPTA R, YAMADA T, MAKINO K, YU J. An overview on oxyfuel coal combustion-State of the art research and technology development[J]. Chem Eng Res Des, 2009, 87(8A):1003-1016. http://www.geste.mecanica.ufrgs.br/pss/oxicomb/An%20overview%20on%20oxyfuel%20coal%20combustion%20-%20State%20of%20art%20research%20and%20technology%20development.pdf [3] BUHRE B, ELLIOTT L K, SHENG C D, GUPTA R P, WALL T F. Oxy-fuel combustion technology for coal-fired power generation[J]. Prog Energy Combust Sci, 2005, 31(4):283-307. doi: 10.1016/j.pecs.2005.07.001 [4] SHENG C, LI Y. Experimental study of ash formation during pulverized coal combustion in O2/CO2 mixtures[J]. Fuel, 2008, 87(7):1297-1305. doi: 10.1016/j.fuel.2007.07.023 [5] YU D, LIANG Z, ZHANG Z, CHANG W, XU M, HONG Y. Iron transformation and ash fusibility during coal combustion in air and O2/CO2 medium[J]. Energy Fuels, 2012, 26(6):3150-3155. doi: 10.1021/ef201786v [6] FRYDA L, SOBRINO C, GLAZER M, BERTRAND C, CIEPLIK M. Study of ash deposition during coal combustion under oxyfuel conditions[J]. Fuel, 2012, 92(1):308-317. doi: 10.1016/j.fuel.2011.08.013 [7] YU D, MORRIS W J, ERICKSON R, WENDT J O L, FRY A, SENIOR C L. Ash and deposit formation from oxy-coal combustion in a 100 kW test furnace[J]. Int J Greenhouse Gas Control, 2011, 5(1):S159-S167. https://www.deepdyve.com/lp/elsevier/ash-and-deposit-formation-from-oxy-coal-combustion-in-a-100-kw-test-TT0Sg3UKfm [8] 李意, 盛昌栋. O2/CO2煤粉燃烧时含铁矿物质转化行为的实验研究[J].燃料化学学报, 2008, 36(4):415-420. http://rlhxxb.sxicc.ac.cn/CN/Y2008/V36/I04/415LI Yi, SHENG Chang-dong. Experimental study on transformation behaviors of iron-bearing minerals during O2/CO2 combustion of pulverized coal[J]. J Fuel Chem Technol, 2008, 36(4):415-420. http://rlhxxb.sxicc.ac.cn/CN/Y2008/V36/I04/415 [9] LIM H, SHAGDARSUREN L, KIM S, HOSHINO A, YAMASHITA T, JEON C. The effect of blending of bituminous and sub-bituminous coals on ash fusibility and deposition formation[J]. J Mech Sci Technol, 2016, 30(3):1413-1420. doi: 10.1007/s12206-016-0249-8 [10] BRYANT G W, BROWNING G J, EMANUEL H, GUPTA S K, GUPTA R P, LUCAS J A, WALL T F. The fusibility of blended coal ash[J]. Energy Fuels, 2000, 14(2):316-325. doi: 10.1021/ef990093+ [11] 陈玉爽, 张忠孝, 乌晓江, 李洁, 管嵘清, 闫博.配煤对煤灰熔融特性影响的实验与量化研究[J].燃料化学学报, 2009, 37(5):521-526. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17485.shtmlCHEN Yu-shuang, ZHANG Zhong-xiao, WU Xiao-jiang, LI Jie, GUAN Rong-qing, YAN Bo. Quantum chemistry calculation and experimental study on coal ash fusion characteristics of blend coal[J]. J Fuel Chem Technol, 2009, 37(5):521-526. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17485.shtml [12] 孙青, 张泰, 黄晓宏, 柳朝晖, 刘超, 陈松涛, 郑楚光.富氧燃烧方式下神华煤熔融特性实验研究[J].燃烧科学与技术, 2016, 22(2):179-185. http://industry.wanfangdata.com.cn/yj/Detail/Periodical?id=Periodical_rskxyjs201602015SUN Qing, ZHANG Tai, HUANG Xiao-hong, LIU Zhao-hui, LIU Chao, CHEN Song-tao, ZHENG Chu-guang. Study of ash fusion behavior of shenhua coale during oxygen enriched combustion[J]. J Combust Sci Technol, 2016, 22(2):179-185. http://industry.wanfangdata.com.cn/yj/Detail/Periodical?id=Periodical_rskxyjs201602015 [13] QIU J R, LI F, ZHENG Y, ZHENG C G, ZHOU H C. The influences of mineral behaviour on blended coal ash fusion characteristics[J]. Fuel, 1999, 78(8):963-969. doi: 10.1016/S0016-2361(99)00005-8 [14] MCLENNAN A R, BRYANT G W, BAILEY C W, STANMORE B R, WALL T F. Index for iron-based slagging for pulverized coal firing in oxidizing and reducing conditions[J]. Energy Fuels, 2000, 14(2):349-354. doi: 10.1021/ef990127d [15] 黄芳, 张立麒, 易宝军, 郑楚光. O2/CO2气氛下高温煤灰热行为及其矿物相转化规律[J].煤炭学报, 2015, 40(11):2714-2719. http://bbs.sciencenet.cn/blog-437124-320988.htmlHUANG Fang, ZHANG Li-qi, YI Bao-jun, ZHENG Chu-guang. Thermal and mineral matter transformation behavior of coal ashes in O2/CO2 atmosphere[J]. J China Coal Soc, 2015, 40(11):2714-2719. http://bbs.sciencenet.cn/blog-437124-320988.html [16] 赵永椿. 煤燃烧矿物组合演化及其与重金属相互作用机制的研究[D]. 武汉: 华中科技大学, 2008.ZHAO Yong-chun. Mineral transformation and its interaction mechanism with heavy metals during coal combustion[D]. Wuhan: Huazhong University of Science and Technology, 2008. [17] QUEROL X. The behaviour of mineral matter during combustion of Spanish subbituminous and brown coals[J]. Mineral Mag, 1994, 58(390):119-133. doi: 10.1180/minmag [18] DENG C, ZHANG C, TAN P, FANG Q, CHEN G. The melting and transformation characteristics of minerals during co-combustion of coal with different sludges[J]. Energy Fuels, 2015, 29(10):6758-6767. doi: 10.1021/acs.energyfuels.5b01201 [19] 袁海平, 梁钦锋, 刘海峰, 龚欣. CaCO3对煤灰熔融特性和黏温特性影响的研究[J].中国电机工程学报, 2012, 32(20):49-55. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=sdmtkj201507079YUAN Hai-ping, LIANG Qin-feng, LIU Hai-feng, GONG Xin. Effects of CaCO3 on the fusion characteristic and viscosity-temperature behaviour of coal ashes[J]. Proc CSEE, 2012, 32(20):49-55. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=sdmtkj201507079 [20] MAYORAL M C, IZQUIERDO M T, ANDRES J M, RUBIO B. Aluminosilicates transformations in combustion followed by DSC[J]. Thermochim Acta, 2001, 373(2):173-180. doi: 10.1016/S0040-6031(01)00459-2 [21] 乌晓江, 张忠孝, 周托, 陈玉爽, 陈国艳, 陆成, 黄凤豹.气化条件下混煤灰熔融特性及矿物质演变规律[J].燃烧科学与技术, 2010, 16(6):508-514. http://www.cqvip.com/QK/98306X/201006/37126051.htmlWU Xiao-jiang, ZHANG Zhong-xiao, ZHOU Tuo, CHEN Yu-shuang, CHEN Guo-yan, LU Cheng, HUANG Feng-bao. Ash fusion characteristics and mineral evolvement of blended ash under gasification condition[J]. J Combust Sci Technol, 2010, 16(6):508-514. http://www.cqvip.com/QK/98306X/201006/37126051.html [22] 兰泽全, 曹欣玉, 周俊虎, 赵显桥, 饶甦, 周志军, 刘建忠, 岑可法.炉内灰渣沉积物中矿物元素分布的电子探针分析[J].中国电机工程学报, 2005, 25(2):117-122. http://www.oalib.com/paper/4397437LAN Ze-quan, CAO Xin-yu, ZHOU Jun-hu, ZHAO Xian-qiao, RAO Su, ZHOU Zhi-jun, LIU Jian-zhong, CHEN Ke-fa. The electron probe analysis of mineral elementary distribution at ash deposition from furnace[J]. Proc CSEE, 2005, 25(2):117-122. http://www.oalib.com/paper/4397437 [23] 陶玉洁, 张彦威, 周俊虎, 景雪晖, 李涛, 刘建忠, 岑可法.准东煤在燃烧过程中的矿物演变过程及Na、Ca释放规律[J].中国电机工程学报, 2015, 35(5):1169-1175. http://www.cnki.com.cn/Article/CJFDTotal-ZGDC201505018.htmTAO Yu-jie, ZHANG Yan-wei, ZHOU Jun-hu, JING Xue-hui, LI Tao, LIU Jian-zhong, CHEN Ke-fa. Mineral conversion regularity and release behavior of Na, Ca during Zhundong coal's combustion[J]. Proc CSEE, 2015, 35(5):1169-1175. http://www.cnki.com.cn/Article/CJFDTotal-ZGDC201505018.htm [24] 丰芸, 李寒旭, 丁立明.利用XRD分析高温下淮南煤灰矿物质变化[J].安徽建筑工业学院学报:自然科学版, 2008, 16(5):53-57. http://www.docin.com/p-521584906.htmlFENG Yun, LI Han-xu, DING Li-ming. Study of huainan coal ash mineral variation under high temperature with XRD[J]. J Anhui Inst Archit Ind:Nat Sci, 2008, 16(5):53-57. http://www.docin.com/p-521584906.html [25] 秦娟, 崔崇, 崔晓昱, 马海龙.钙长石晶体的形成机制研究[J].人工晶体学报, 2016, 45(5):1153-1157. http://www.cnki.com.cn/Article/CJFDTotal-RGJT201605002.htmQIN Juan, CUI Cong, CUI Xiao-yu, MA Hai-long. Study on formation mechanism of anorthite crystal[J]. J Synthetic Cryst, 2016, 45(5):1153-1157. http://www.cnki.com.cn/Article/CJFDTotal-RGJT201605002.htm [26] 杜胜磊. 生物质热化学利用过程中无机矿物质转化规律及灰熔融特性研究[D]. 武汉: 华中科技大学, 2014.DU Sheng-lei. Fundamental Study on Transformation behavior of inorganic components during thermochemical conversion of biomassand ash fusion characteristics[D]. Wuhan: Huazhong University of Science and Technology, 2014. -





 下载:
下载: