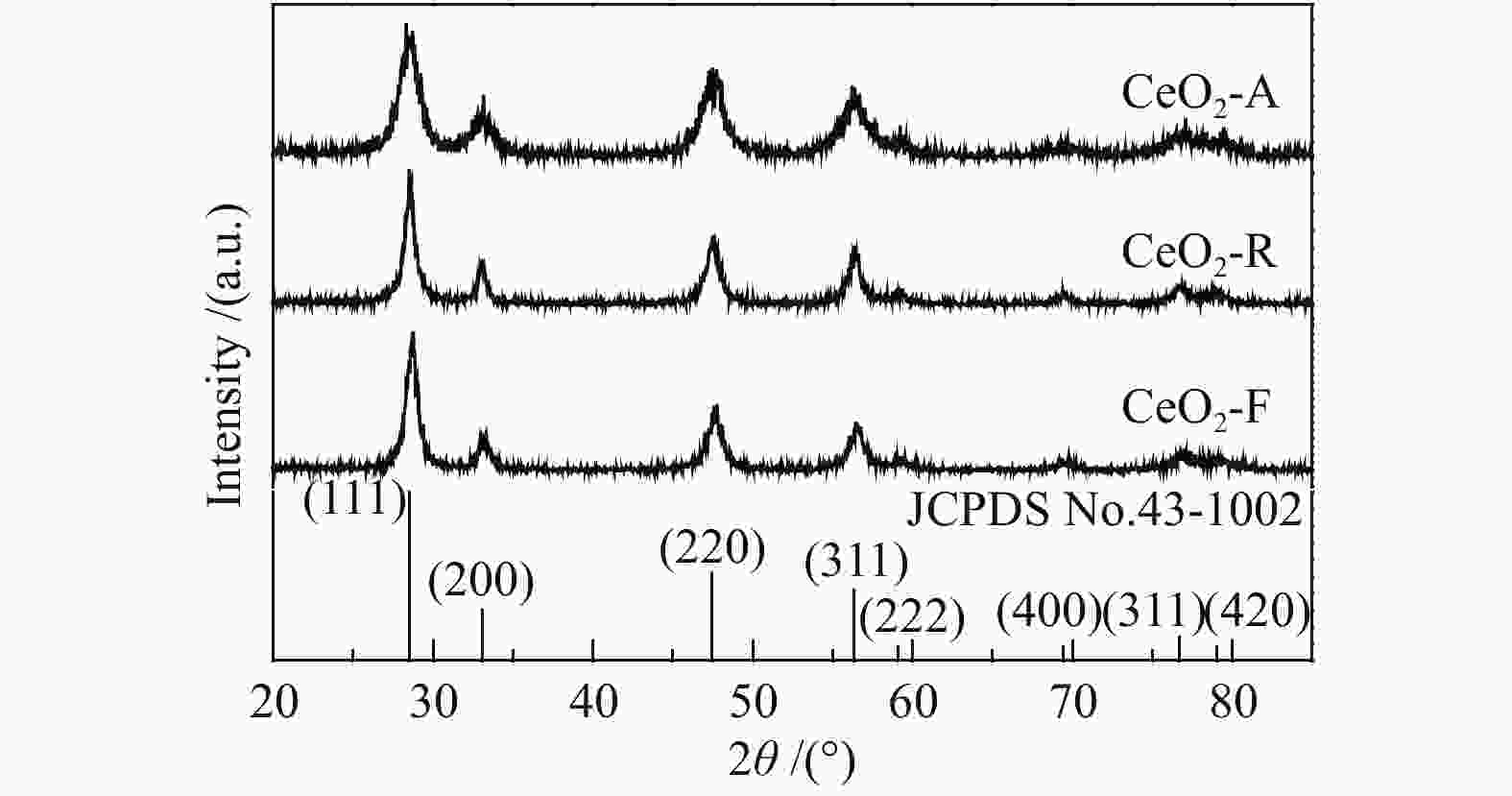
Effect of preparation methods on the structure and catalytic performance of CeO2 for toluene combustion
-
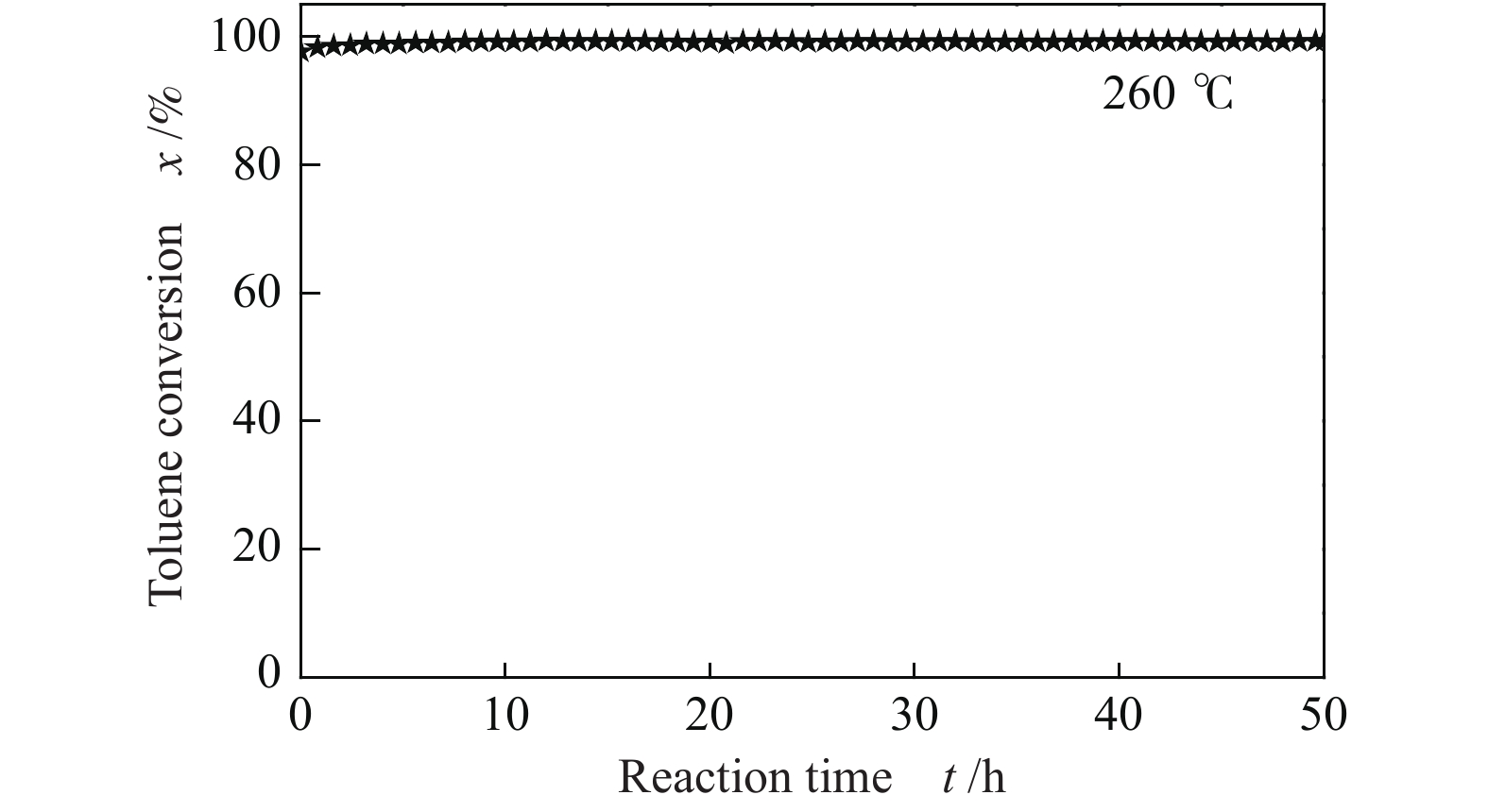
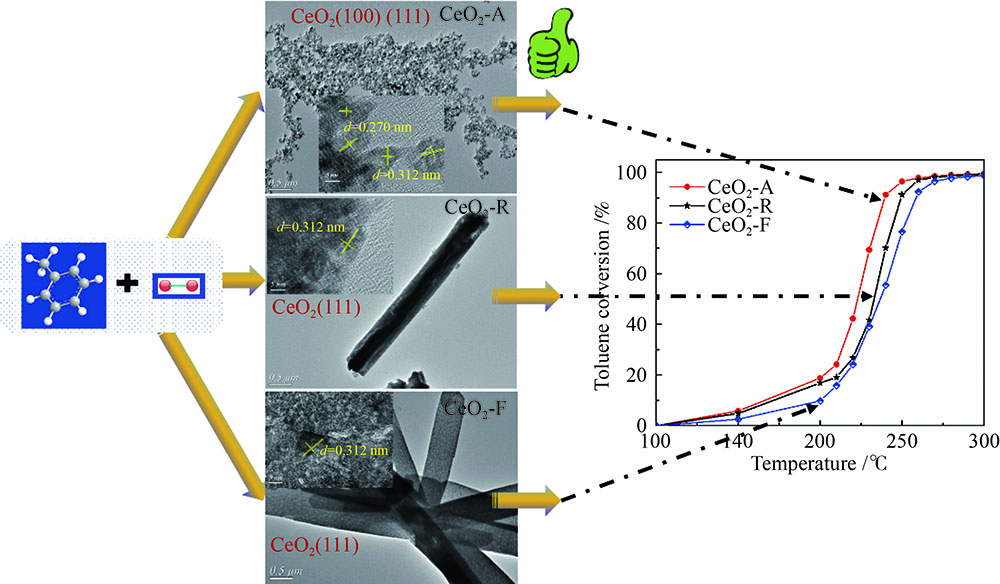
摘要: 采用溶胶-凝胶-超临界干燥法、水热法及共沉淀法分别合成了氧化铈气凝胶(CeO2-A)、纳米棒(CeO2-R)和纳米片(CeO2-F)。考察了不同形貌氧化铈的催化燃烧甲苯性能,通过多种方法分析表征了氧化铈样品的微观结构,讨论了不同方法制得的CeO2形貌结构对催化性能的影响。结果表明,CeO2-R和CeO2-F比表面积较低,并且仅暴露(111)晶面,催化燃烧甲苯活性较低。CeO2-A具有高比表面积和丰富的孔道结构,有利于反应物分子的吸附,而且同时暴露(100)和(111)两种活性晶面,增加了氧空位浓度(Osur/Olatt = 0.25)。此外,CeO2-A由于表面晶格氧移动性较强,有利于Ce3+/Ce4+氧化还原的循环,加快甲苯深度氧化反应的进行。因此,CeO2-A具有更加优异的催化燃烧甲苯活性,t50和t90分别为223 和239 ℃,这主要归因于其大比表面积、高暴露活性晶面以及强晶格氧迁移性。Abstract: CeO2 aerogel (CeO2-A), nanorod (CeO2-R) and nanoflake (CeO2-F) were prepared via sol-gel, hydrothermal and coprecipitation methods, respectively. The effect of morphology and structure of CeO2 on the catalytic performance in toluene combustion reaction was investigated based on structure analysis provided by characterization. The results revealed that the activity of both CeO2-R and CeO2-F was inferior to that of CeO2-A, due to CeO2-R and CeO2-F smaller specific surface area only exposed (111) crystal plane dominantly detected from their TEM images. While, the CeO2-A had a larger specific surface area and more exposed (111) and (100) facet, which contributed to exposure and formation of more oxygen vacancies and further to the adsorption of more gaseous oxygen. In addition, highly mobile lattice oxygen was another critical factor for influencing the catalytic performance of CeO2, which was beneficial to the redox cycle of Ce3+/Ce4+ and could further accelerate the toluene combustion. As a result, the CeO2-A catalyst exhibited the superior performance in toluene catalytic combustion with t50 of 223℃ and t90 of 239℃, respectively, owing to the larger specific surface area, higher exposure of reactive crystal plane and stronger mobility of lattice oxygen.
-
Key words:
- ceria morphology /
- catalytic combustion /
- preparation method /
- crystal face /
- oxygen vacancy
-
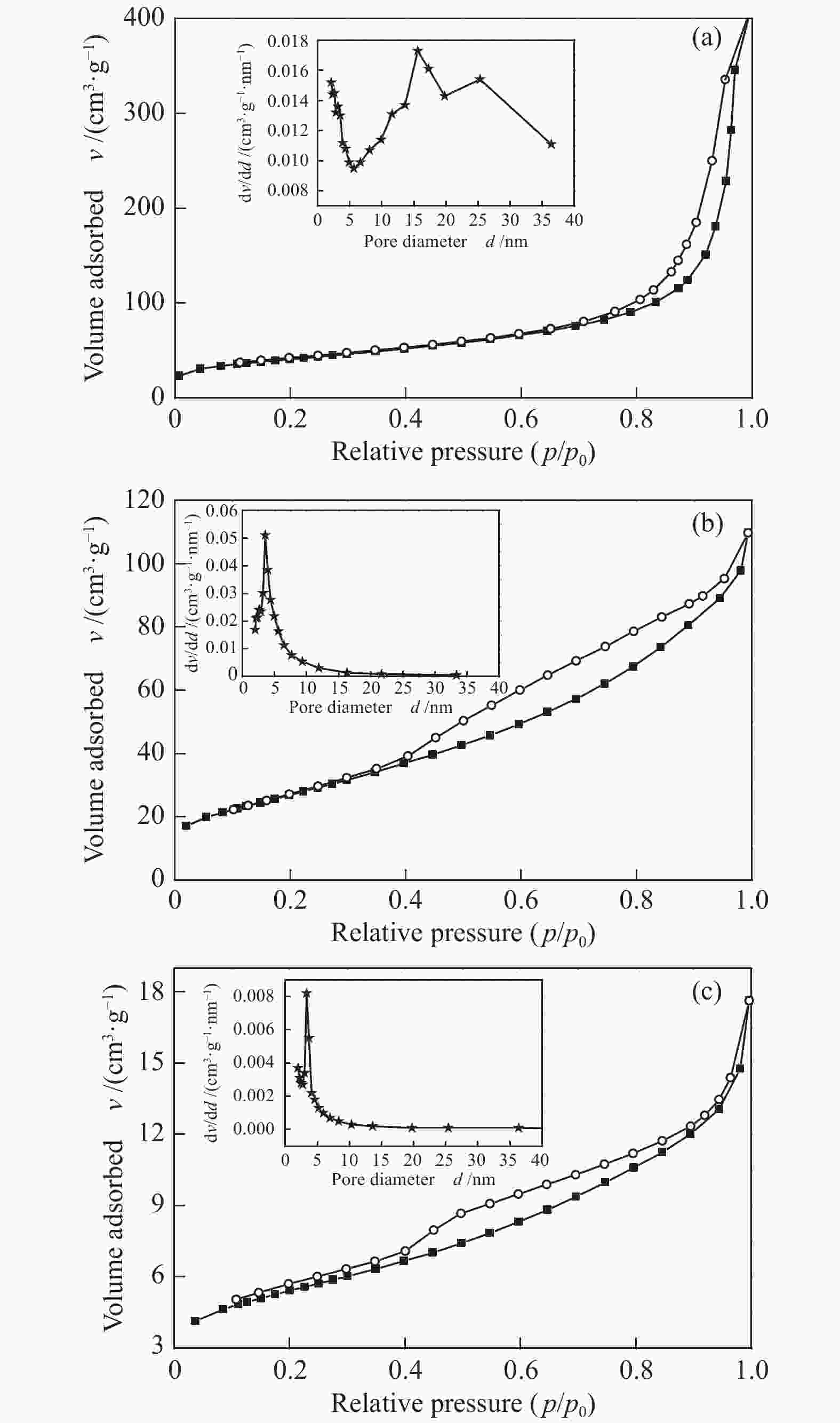
表 1 不同形貌氧化铈催化剂的织构性质
Table 1 Structural properties of CeO2 catalysts with different morphologies
Catalyst SBETa/
(m2·g−1)vtotalb/
(cm3·g−1)Average pore
diameterc d/nmCrystallite
sizesd d/nmCeO2-A 144.0 0.60 15.6 10.6 CeO2-R 99.7 0.20 5.3 16.7 CeO2-F 18.4 0.03 6.0 15.5 a: specific surface area was calculated using Brumauer-Emmett-Teller (BET) modelling;
b: total pore volume was measured by single point adsorption at p/p0 = 0.99;
c: most probable apertures calculated by BJH method;
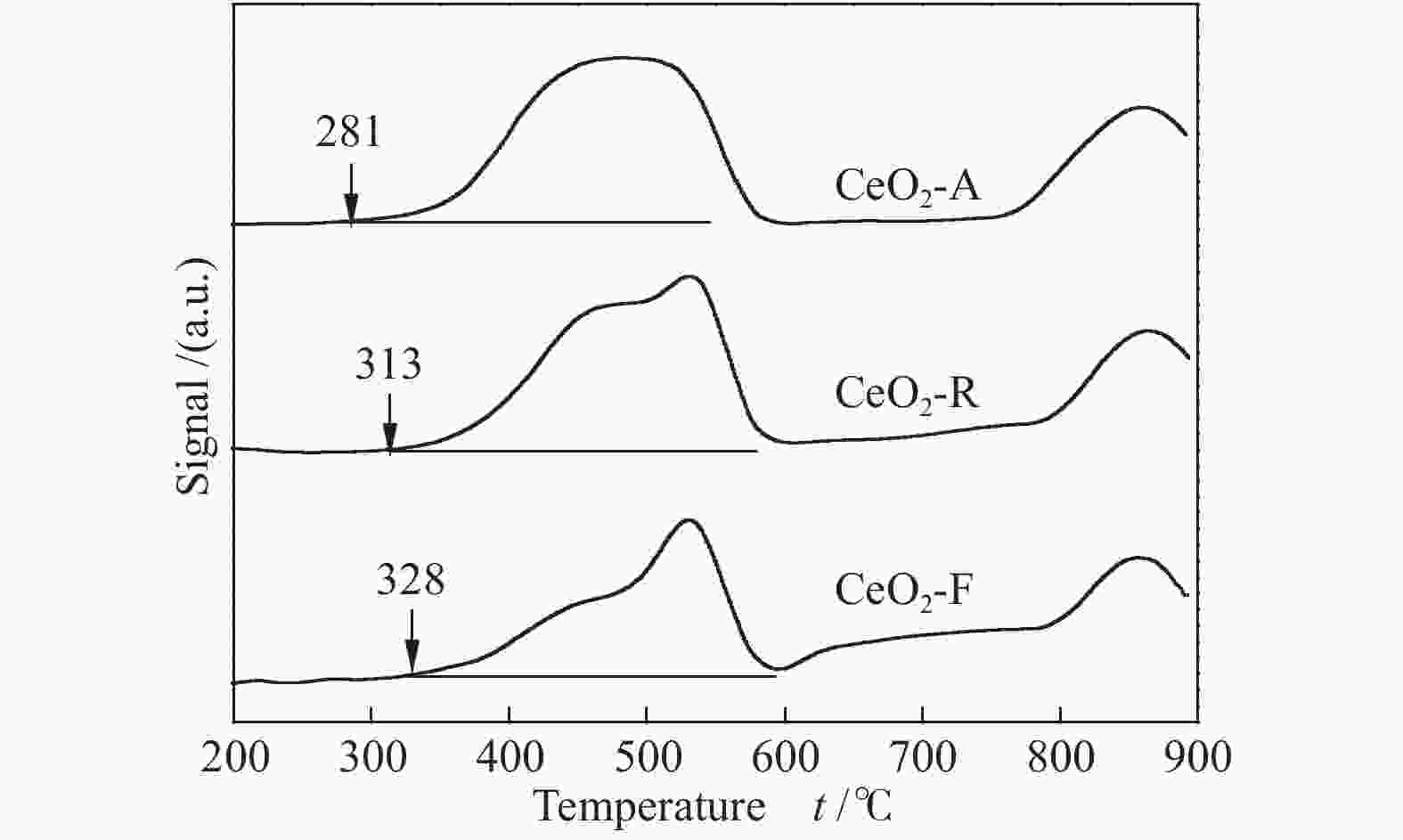
d: calculated by the Scherrer equation applied to the CeO2 (111) peak表 2 不同形貌的氧化铈催化剂TPR表征
Table 2 TPR results of CeO2 catalysts with different morphologies
Catalyst Started reduction
temperature t/℃H2 consumption
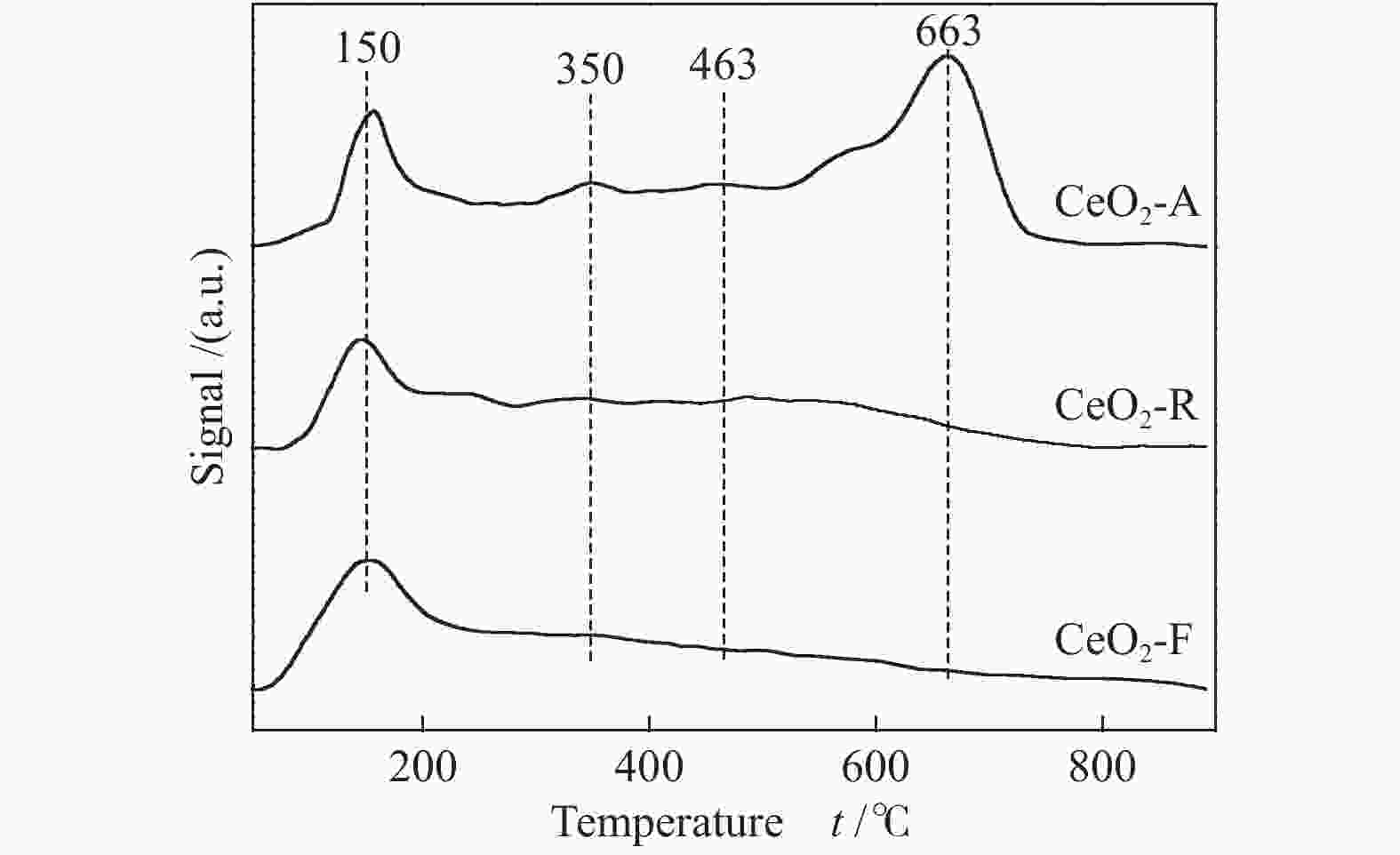
below 600 ℃CeO2-A 281 5020 CeO2-R 313 4292 CeO2-F 328 3100 表 3 不同形貌氧化铈催化剂的表面元素分析
Table 3 Surface element analysis of CeO2 catalysts with different morphologies
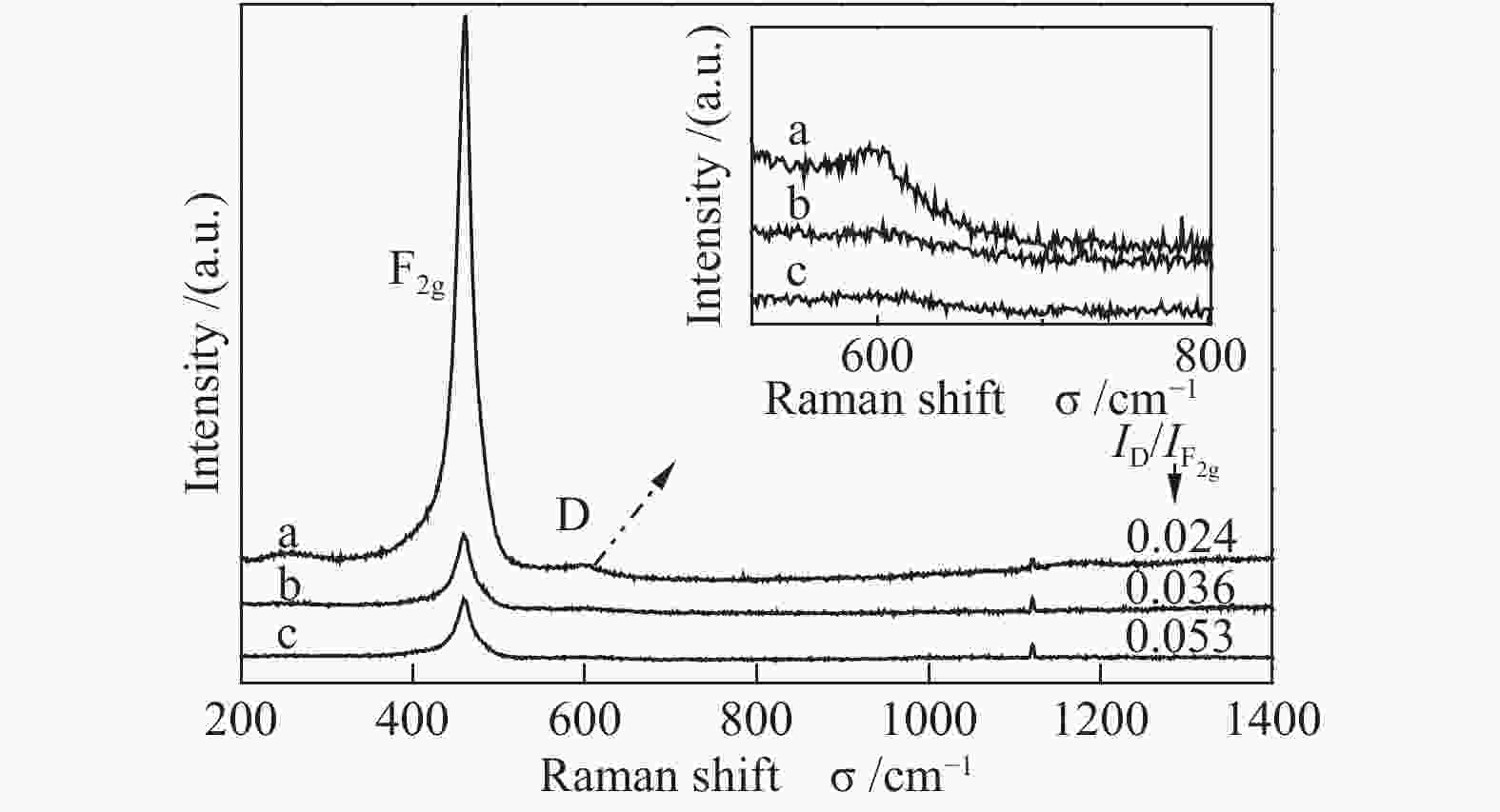
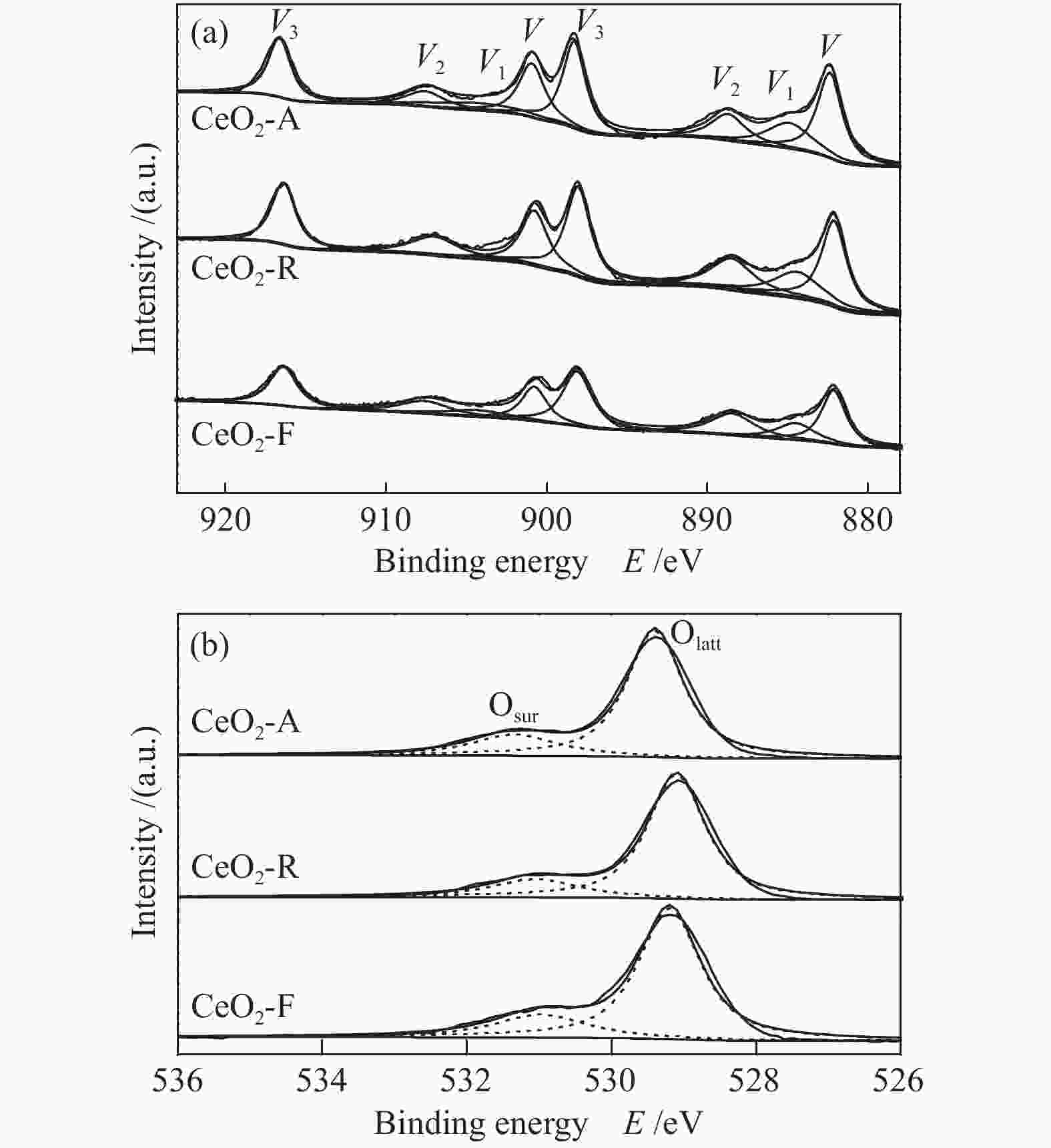
Catalyst Ce3+/Ce4+ Osur/Olatt Ce3+/Ce CeO2-A 0.25 0.25 0.20 CeO2-R 0.23 0.22 0.19 CeO2-F 0.16 0.20 0.14 表 4 不同形貌氧化铈催化剂对甲苯燃烧的活性评价
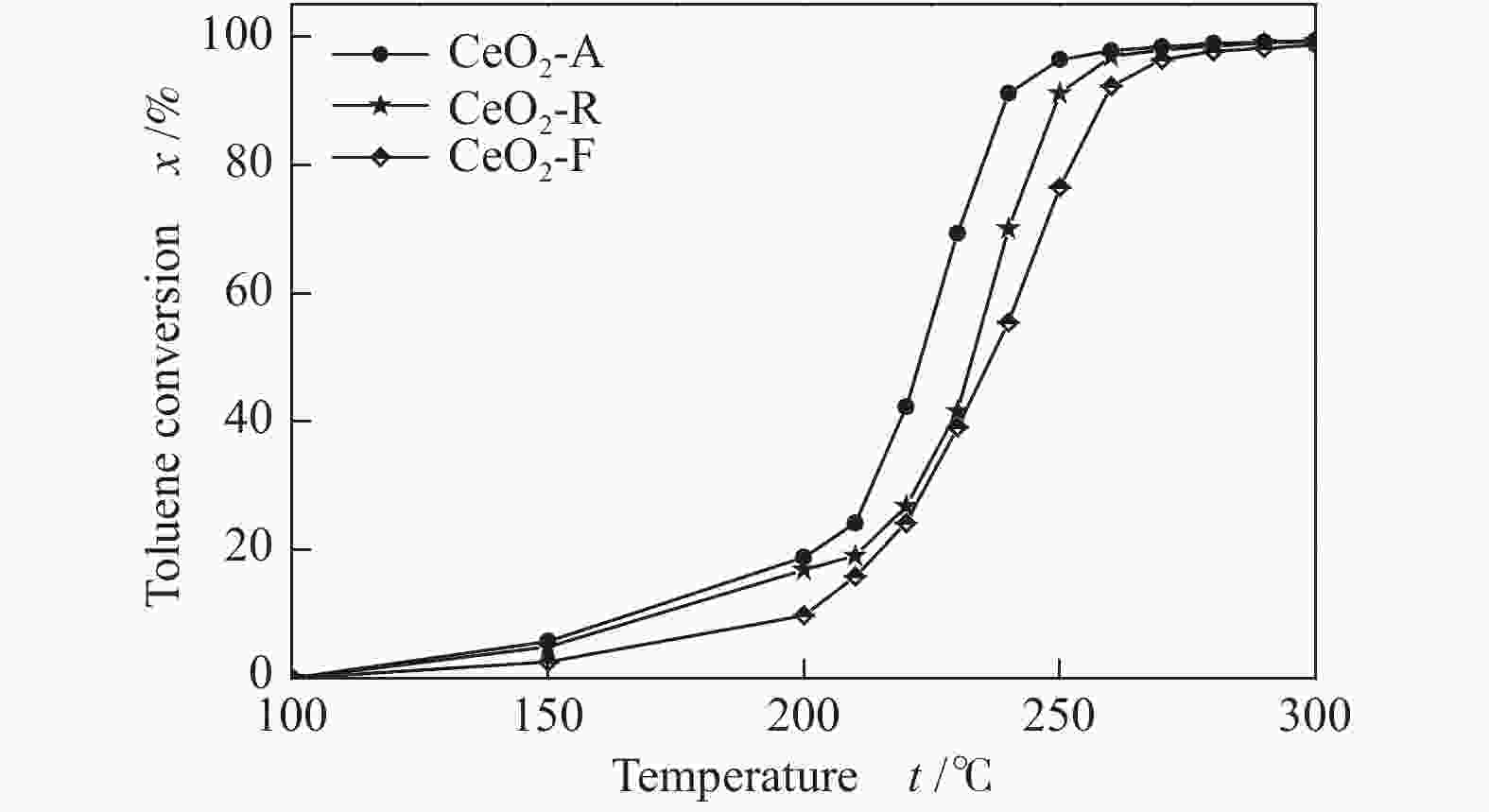
Table 4 Catalytic activity of different morphologies CeO2 catalysts for oxidation of toluene
Sample Preparation method Toluene
concentration/
(g·L−1)WHSV/
(mL·g−1·h−1)t50/
℃t90/
℃CeO2-A sol-gel 1 78000 223 239 CeO2-R hydrothermal 1 78000 233 250 CeO2-F precipitation 1 78000 236 259 -
[1] 张嘉妮. “十三五”挥发性有机物总量控制情景分析[J]. 环境科学,2018,39(8):3544−3551.ZHANG Jia-ni. Scenario analyses of the volatile organic compound emission allowance and allocation in the 13th Five-Year period[J]. Chin J Environ Sci,2018,39(8):3544−3551. [2] 席劲瑛,武俊良,胡洪营,王灿. 工业VOCs气体处理技术应用状况调查分析[J]. 中国环境科学,2012,32(11):1955−1960. doi: 10.3969/j.issn.1000-6923.2012.11.005XI Jin-ying, WU Jun-liang, HU Hong-ying, WANG Can. Application status of industrial VOCs gas treatment techniques[J]. China Environ Sci,2012,32(11):1955−1960. doi: 10.3969/j.issn.1000-6923.2012.11.005 [3] EVERAERT K, BAEYENS J. Catalytic combustion of volatile organic compounds[J]. J Hazard Mater,2004,109(1/3):113−139. doi: 10.1016/j.jhazmat.2004.03.019 [4] LU H, ZHOU Y, HUANG H, ZHANG B, CHEN Y. In-situ synthesis of monolithic Cu-Mn-Ce/cordierite catalysts towards VOCs combustion[J]. J Rare Earth,2011,29(9):855−860. doi: 10.1016/S1002-0721(10)60555-8 [5] ZHANG P, LU H, ZHOU Y, ZHANG L, WU Z, YANG S, SHI H, ZHU Q, CHEN Y, DAI S. Mesoporous MnCeOx solid solutions for low temperature and selective oxidation of hydrocarbons[J]. Nat Commun,2015,6:8446−8455. doi: 10.1038/ncomms9446 [6] LIAN W, YU Y, HE H, ZHANG Y, QIN X, WANG B. Oxygen vacancy clusters essential for the catalytic activity of CeO2 nanocubes for o-xylene oxidation[J]. Sci Rep,2017,1(7):12845−12850. [7] PENG R, SUN X, LI S, CHEN L, FU M, WU J, YE D. Shape effect of Pt/CeO2 catalysts on the catalytic oxidation of toluene[J]. Chem Eng J,2016,306:1234−1246. doi: 10.1016/j.cej.2016.08.056 [8] FENG Z, REN Q, PENG R, MO S, ZHANG M, FU M, CHEN L, YE D. Effect of CeO2 morphologies on toluene catalytic combustion[J]. Catal Today,2019,332:177−182. doi: 10.1016/j.cattod.2018.06.039 [9] FENG Z, ZHANG M, REN Q, MO S, PENG R, YAN D, FU M, CHEN L, WU J, YE D. Design of 3-dimensionally self-assembled CeO2 hierarchical nanosphere as high efficiency catalysts for toluene oxidation[J]. Chem Eng J,2019,369:18−25. doi: 10.1016/j.cej.2019.03.051 [10] LIAO Y, HE L, MAN C, CHEN L, FU M, WU J, YE D, HUANG B. Diameter-dependent catalytic activity of ceria nanorods with various aspect ratios for toluene oxidation[J]. Chem Eng J,2014,256:439−447. doi: 10.1016/j.cej.2014.07.014 [11] LV J, SHEN Y, PENG L, GUO X, DING W. Exclusively selective oxidation of toluene to benzaldehyde on ceria nanocubes by molecular oxygen[J]. Chem Commun (Camb),2010,46(32):5909−5911. doi: 10.1039/c0cc00777c [12] HUANG X, SUN H, WANG L, LIU Y, FAN K, CAO Y. Morphology effects of nanoscale ceria on the activity of Au/CeO2 catalysts for low-temperature CO oxidation[J]. Appl Catal B: Environ,2009,90(1/2):224−232. doi: 10.1016/j.apcatb.2009.03.015 [13] PAN C, ZHANG D, SHI L. CTAB assisted hydrothermal synthesis, controlled conversion and CO oxidation properties of CeO2 nanoplates, nanotubes, and nanorods[J]. J Solid State Chem,2008,181(6):1298−1306. doi: 10.1016/j.jssc.2008.02.011 [14] 闫宁, 周安宁, 张亚刚, 杨志远, 贺新福, 张亚婷. CeO2的形貌特征对Ni/CeO2 催化剂CO甲烷化性能的影响[J]. 燃料化学学报,2020,48(4):466−475. doi: 10.3969/j.issn.0253-2409.2020.04.010YAN Ning, ZHOU An-ning, ZHANG Ya-gang, YANG Zhi-yuan, HE Xin-fu, ZHANG Ya-ting. Morphologic effect of CeO2 on the catalytic performance of Ni/CeO2 in CO methanation[J]. J Fuel Chem Technol,2020,48(4):466−475. doi: 10.3969/j.issn.0253-2409.2020.04.010 [15] 李健. 富氢中一氧化碳选择氧化的铈基气凝胶催化剂的研究[D]. 内蒙古: 内蒙古工业大学, 2007.LI Jian. Studies on CeO2-based aerogel catalysts used for preferential oxidation of carbon monoxide in excess hydrogen[D]. Inner Mongolia: Inner Mongolia University of Technology, 2007. [16] KEMPAIAH D M, YIN S, SATO T. A facile and quick solvothermal synthesis of 3D microflower CeO2 and Gd: CeO2 under subcritical and supercritical conditions for catalytic applications[J]. CrystEngComm,2011,13(3):741−746. doi: 10.1039/C0CE00611D [17] TROVARELLI A, LLORCA J. Ceria catalysts at nanoscale: How do crystal shapes shape catalysis?[J]. ACS Catal,2017,7(7):4716−4735. doi: 10.1021/acscatal.7b01246 [18] BAI J, XU Z, ZHENG Y, YIN H. Shape control of CeO2 nanostructure materials in microemulsion systems[J]. Mater Lett,2006,60(9/10):1287−1290. doi: 10.1016/j.matlet.2005.11.016 [19] WU K. SUN L, YAN C. Recent progress in well-controlled synthesis of ceria-based nanocatalysts towards enhanced catalytic performance[J]. Adv Energy Mater,2016,6(17):1600501. doi: 10.1002/aenm.201600501 [20] ZHANG M, GUO S, ZHENG L, ZHANG G, HAO Z, KANG L, LIU Z. Preparation of NiMn2O4 with large specific surface area from an epoxide-driven sol-gel process and its capacitance[J]. Electrochim Acta,2013,87:546−553. doi: 10.1016/j.electacta.2012.09.085 [21] RAO R, ZHANG Q, LIU H, YANG M, LING Q, ZHANG A. Formaldehyde-assisted hydrothermal synthesis of one-dimensional CeO2 and their morphology-dependent properties[J]. CrystEngComm,2012,14(18):5929−5936. doi: 10.1039/c2ce25644d [22] DAI Q, ZHANG Z, YAN J, WU J, JOHNSON G, SUN W, WANG X, ZHANG S, ZHAN W. Phosphate-functionalized CeO2 nanosheets for efficient catalytic oxidation of dichloromethane[J]. Environ Sci Technol,2018,52(22):13430−13437. doi: 10.1021/acs.est.8b05002 [23] YAN D, MO S, SUN Y, REN Q, FENG Z, CHEN P, WU J, FU M, YE D. Morphology-activity correlation of electrospun CeO2 for toluene catalytic combustion[J]. Chemosphere,2020,247:125860. doi: 10.1016/j.chemosphere.2020.125860 [24] JIANG Y, GAO J, ZHANG Q, LIU Z, FU M, WU J, HU Y, YE D. Enhanced oxygen vacancies to improve ethyl acetate oxidation over MnOx-CeO2 catalyst derived from MOF template[J]. Chem Eng J,2019,371:78−87. doi: 10.1016/j.cej.2019.03.233 [25] CHOWDHURY M B, SUI R, LUCKY R A, CHARPENTIER P A. One-pot procedure to synthesize high surface area alumina nanofibers using supercritical carbon dioxide[J]. Langmuir,2010,26(4):2707−2713. doi: 10.1021/la902738y [26] WANG Y, XUE Y, ZHAO C, ZHAO D, LIU F, WANG K, DIONYSIOU D D. Catalytic combustion of toluene with La0.8Ce0.2MnO3 supported on CeO2 with different morphologies[J]. Chem Eng J,2016,300:300−305. doi: 10.1016/j.cej.2016.04.007 [27] YANG P, YANG S, SHI Z, MENG Z, ZHOU R. Deep oxidation of chlorinated VOCs over CeO2-based transition metal mixed oxide catalysts[J]. Appl Catal B: Environ,2015,162:227−235. doi: 10.1016/j.apcatb.2014.06.048 [28] YAN Y, WANG L, ZHANG H. Catalytic combustion of volatile organic compounds over Co/ZSM-5 coated on stainless steel fibers[J]. Chem Eng J,2014,255:195−204. doi: 10.1016/j.cej.2014.05.141 [29] LI J, ZUO S, YANG P, QI C. Study of CeO2 modified AlNi mixed pillared clays supported palladium catalysts for benzene adsorption/desorption catalytic combustion[J]. Materials,2017,10(8):949. [30] WANG W, ZHU Q, DAI Q, WANG X. Fe doped CeO2 nanosheets for catalytic oxidation of 1,2-dichloroethane: Effect of preparation method[J]. Chem Eng J,2017,307:1037−1046. doi: 10.1016/j.cej.2016.08.137 [31] TANG X, XU Y, SHEN W. Promoting effect of copper on the catalytic activity of MnOx-CeO2 mixed oxide for complete oxidation of benzene[J]. Chem Eng J,2008,144(2):175−180. doi: 10.1016/j.cej.2008.01.016 [32] 张新华. Ce和Mn金属氧化物低温催化燃烧DCM及其性能调控[D]. 杭州: 浙江工业大学, 2019.Zhang Xin-hua. Low-temperature catalytic combustion DCM on Ce and Mn oxides and performances tuning[D]. Hangzhou: Zhejiang University of Technology, 2019. [33] HU F, CHEN J, PENG Y, SONG H, LI K, LI J. Novel nanowire self-assembled hierarchical CeO2 microspheres for low temperature toluene catalytic combustion[J]. Chem Eng J,2018,331:425−434. doi: 10.1016/j.cej.2017.08.110 [34] LI P, HE C, CHENG J, MA C, DOU B, HAO Z. Catalytic oxidation of toluene over Pd/Co3AlO catalysts derived from hydrotalcite-like compounds: Effects of preparation methods[J]. Appl Catal B: Environ,2011,101(3/4):570−579. doi: 10.1016/j.apcatb.2010.10.030 [35] CHEN X, CHEN X, YU E, CAI S, JIA H, CHEN J, LIANG P. In situ pyrolysis of Ce-MOF to prepare CeO2 catalyst with obviously improved catalytic performance for toluene combustion[J]. Chem Eng J,2018,344:469−479. doi: 10.1016/j.cej.2018.03.091 [36] HAN W, ZHAO H, DONG F, TANG Z. Morphology-controlled synthesis of 3D, mesoporous, rosette-like CeCoOx catalysts by pyrolysis of Ce[Co(CN)6] and application for the catalytic combustion of toluene[J]. Nanoscale,2018,10(45):21307−21319. doi: 10.1039/C8NR07882C [37] FENG X, GUO J, WEN X, XU M, CHU Y, YUAN S. Enhancing performance of Co/CeO2 catalyst by Sr doping for catalytic combustion of toluene[J]. Appl Surface Sci,2018,445:145−153. doi: 10.1016/j.apsusc.2018.03.070 [38] CHANG S, LI M, HUA Q, ZHANG L, MA Y, YE B, HUANG W. Shape-dependent interplay between oxygen vacancies and Ag-CeO2 interaction in Ag/CeO2 catalysts and their influence on the catalytic activity[J]. J Catal,2012,293:195−204. doi: 10.1016/j.jcat.2012.06.025 [39] SAKTHIVEL T S, REID D L, BHATTA U M, MÖBUS G, SAYLE D C, SEAL S. Engineering of nanoscale defect patterns in CeO2 nanorods via ex situ and in situ annealing[J]. Nanoscale,2015,7(12):5169−5177. doi: 10.1039/C4NR07308H [40] LIU X, ZHOU K, WANG L, WANG B, LI Y. Oxygen vacancy clusters promoting reducibility and activity of ceria nanorods[J]. J Am Chem Soc,2009,131(9):3140−3141. doi: 10.1021/ja808433d [41] LUO Y, LIN D, ZHENG Y, FENG X, CHEN Q, ZHANG K, WANG X, JIANG L. MnO2 nanoparticles encapsuled in spheres of Ce-Mn solid solution: Efficient catalyst and good water tolerance for low-temperature toluene oxidation[J]. Appl Surf Sci,2020,504:144481. doi: 10.1016/j.apsusc.2019.144481 [42] HE H, LIN X, LI S, WU Z, GAO J, WU J, WEN W, YE D, FU M. The key surface species and oxygen vacancies in MnOx(0.4)-CeO2 toward repeated soot oxidation[J]. Appl Catal B: Environ,2018,223:134−142. doi: 10.1016/j.apcatb.2017.08.084 [43] LIOTTA L F, OUSMANE M, DI CARLO G, PANTALEO G, DEGANELLO G, BOREAVE A, GIROIR-FENDLER A. Catalytic removal of toluene over Co3O4-CeO2 mixed oxide catalysts: comparison with Pt/Al2O3[J]. Catal Lett,2008,127(3/4):270−276. [44] LIAO Y, FU M, CHEN L, WU J, HUANG B, YE D. Catalytic oxidation of toluene over nanorod-structured Mn-Ce mixed oxides[J]. Catal Today,2013,216:220−228. doi: 10.1016/j.cattod.2013.06.017 [45] LI Y, SHEN W. Morphology-dependent nanocatalysis on metal oxides[J]. Sci China Chem,2012,55(12):2485−2496. doi: 10.1007/s11426-012-4565-2 [46] GAO Y, WANG W, CHANG S, HUANG W. Morphology effect of CeO2 support in the preparation, metal-support interaction, and catalytic performance of Pt/CeO2 catalysts[J]. ChemCatChem,2013,5(12):3610−3620. doi: 10.1002/cctc.201300709 -





 下载:
下载: