Experimental study on catalytic pyrolysis of lignin under char and ZSM-5 for preparation of aromatics
-
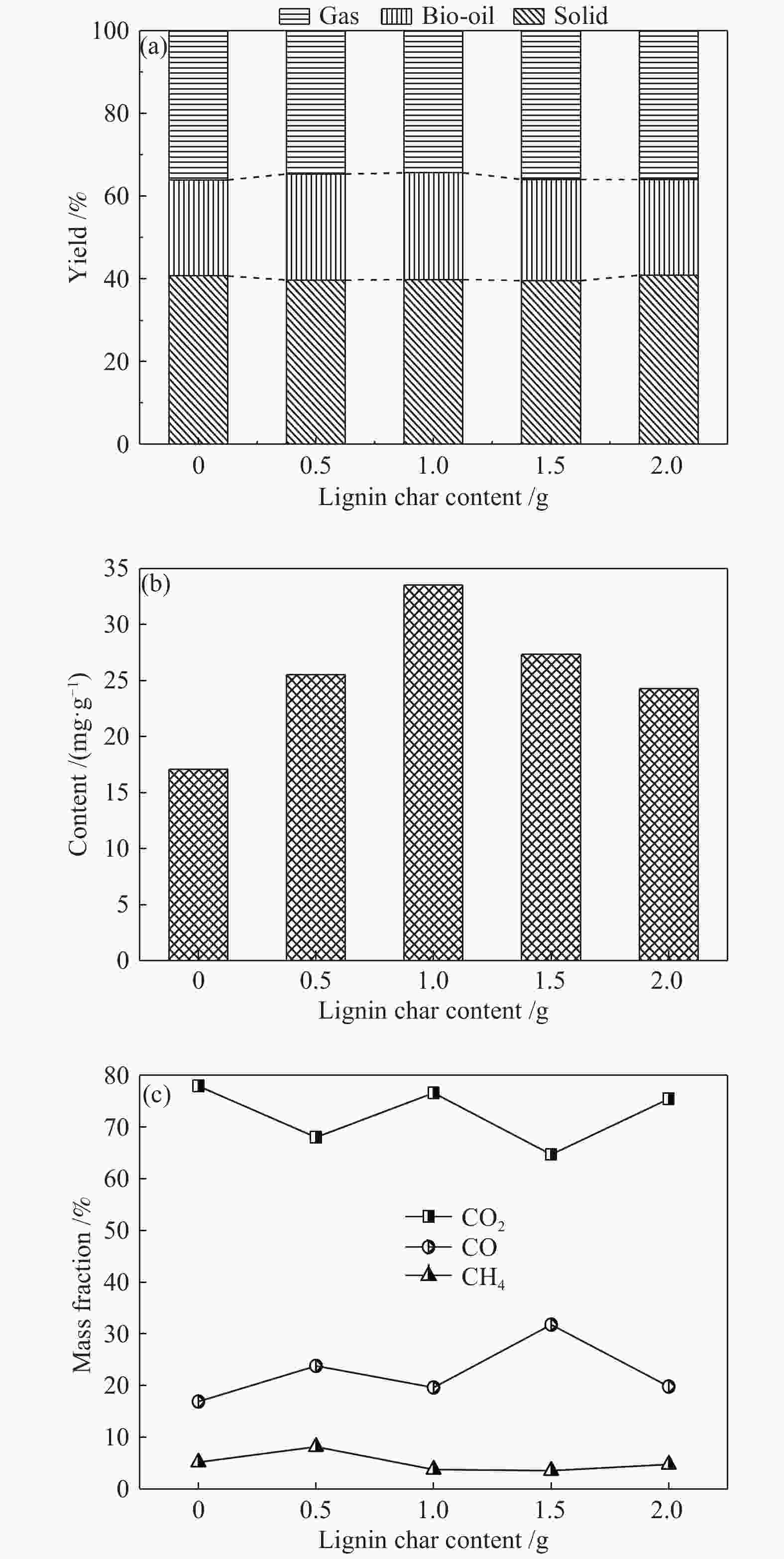
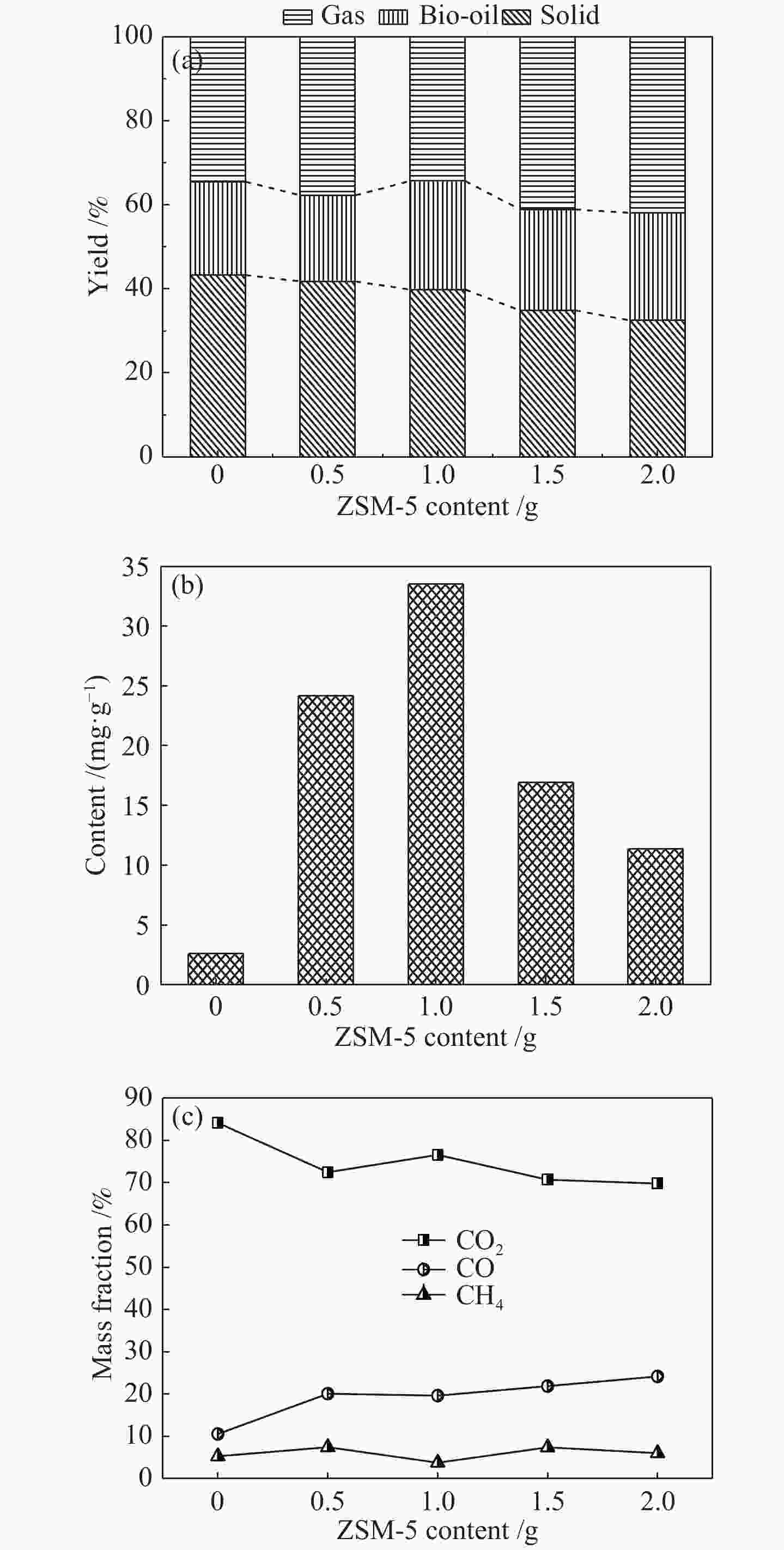
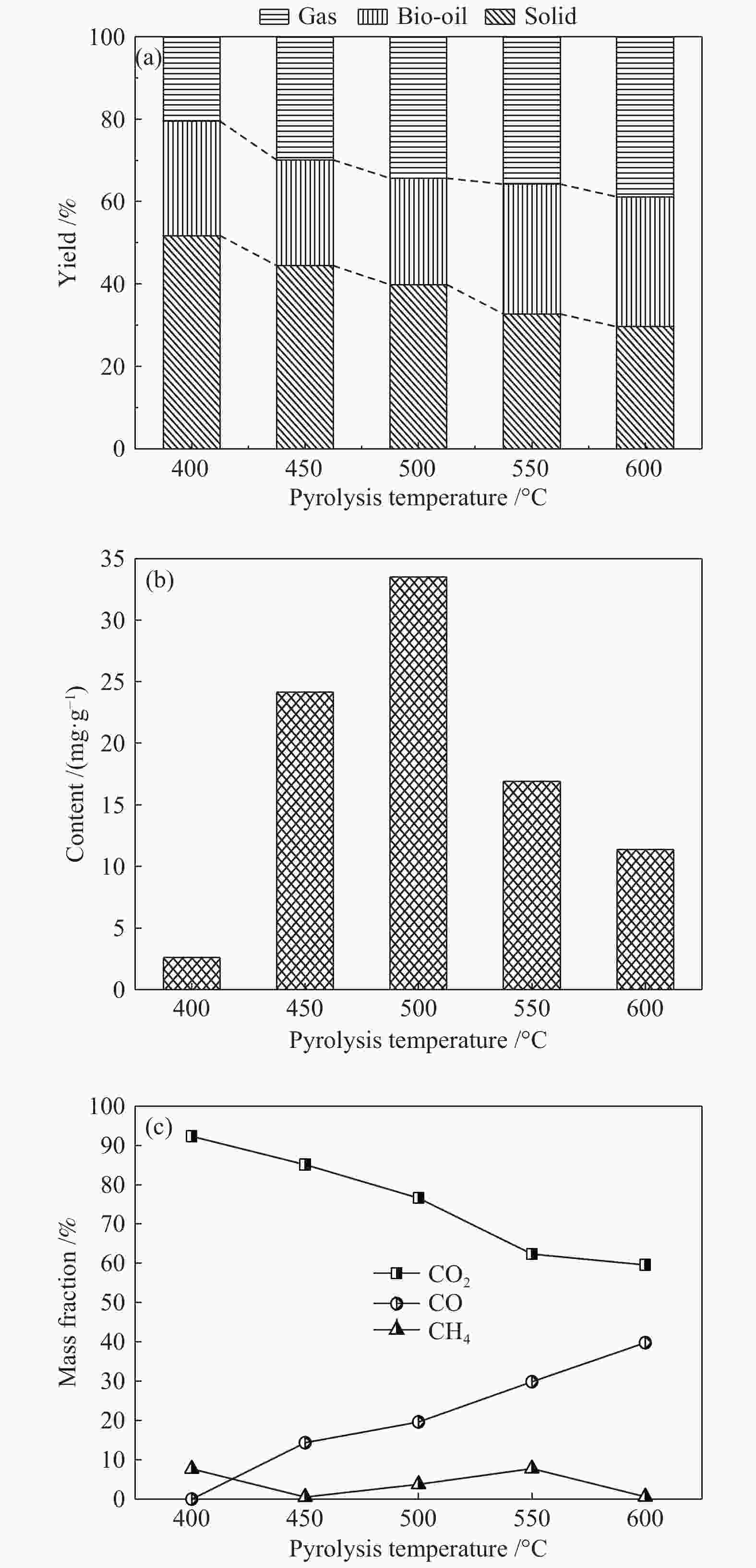
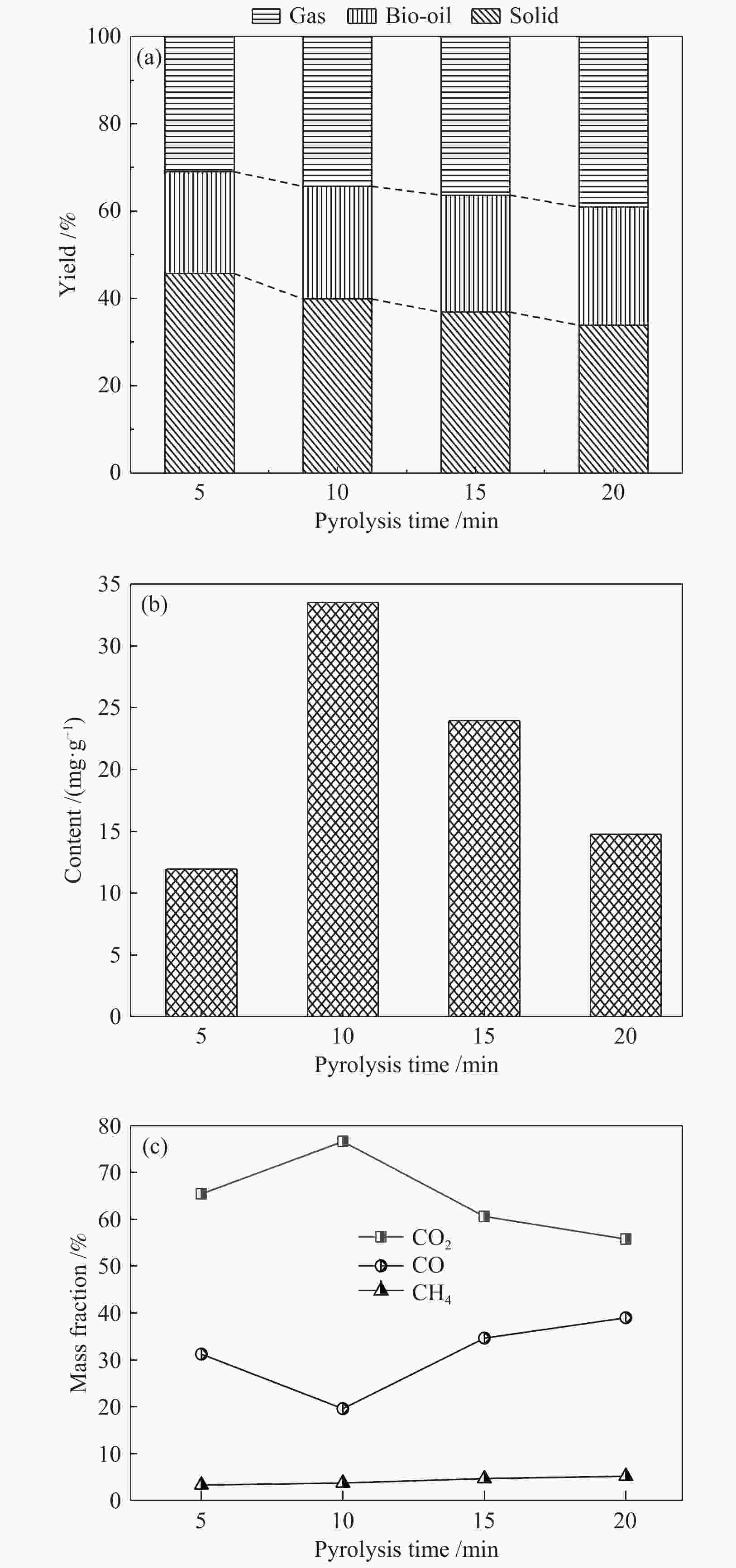
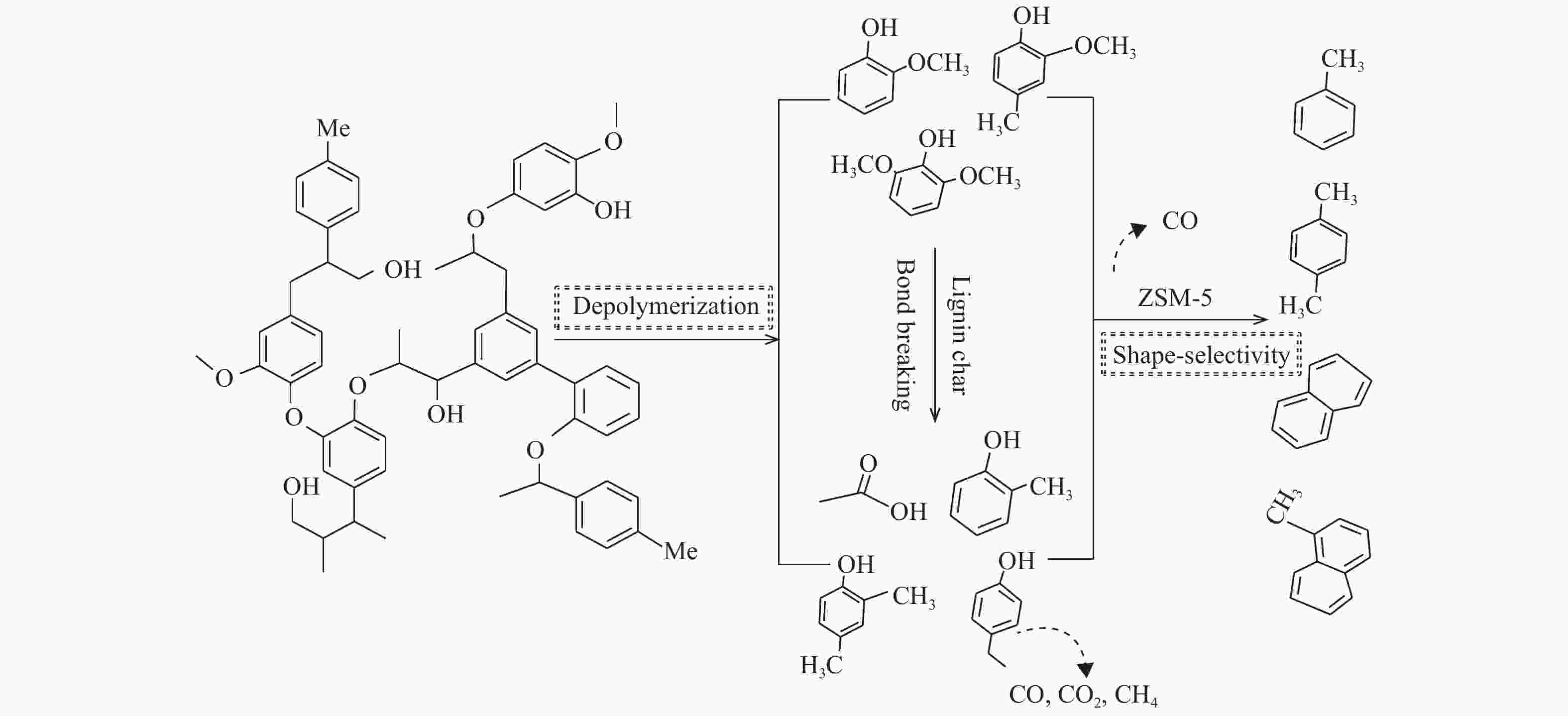
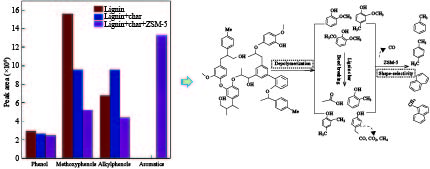
摘要: 为了提高木质素催化热解所得芳烃的产率,本研究以碱木质素为原料,在碱木质素炭和ZSM-5的二元催化体系下进行快速热解实验,选取催化剂比例、热解温度、热解时间等为实验变量,探究碱木质素热解产物中芳烃的变化规律以及碱木质素炭和ZSM-5协同催化作用机理。结果表明,碱木质素催化热解所得芳烃的产量从17 mg/g(未加炭)提高到33 mg/g(炭添加量为1 g),产率增加了近一倍。通过不同工况研究发现,碱木质素快速热解制备芳烃的最佳条件是:碱木质素∶碱木质素炭∶ZSM-5 = 1∶1∶1, 热解温度为500 ℃,热解时间为10 min。机理分析表明,热解过程中碱木质素炭主要起断键作用,而ZSM-5起择形芳构化作用,两者协同作用得到更高的芳烃产率。Abstract: In order to improve yield of aromatics from catalytic pyrolysis of lignin, alkali lignin was used for rapid pyrolysis experiments under a binary catalysis system of alkali lignin char and ZSM-5. Influence of catalyst ratio and pyrolysis temperature as well as pyrolysis time on aromatics quantity and mechanism of synergistic catalysis of alkali lignin char and ZSM-5 were investigated. The results show that quantity of aromatics has increased from 17 mg/g (without char) to 33 mg/g (with char addition of 1 g), which is about doubled. The optimal conditions for preparation of aromatics are under alkali lignin∶alkali lignin char∶ZSM-5=1∶1∶1 at 500℃ for 10 min. Meanwhile, the mechanism analysis shows that alkali lignin char mainly plays a role in bond breaking during pyrolysis, while ZSM-5 can act as selective aromatization to obtain higher aromatics yield, whose synergistic effects result in higher aromatic yield.
-
Key words:
- alkali lignin /
- alkali lignin char /
- binary catalytic system /
- aromatics /
- synergy
-
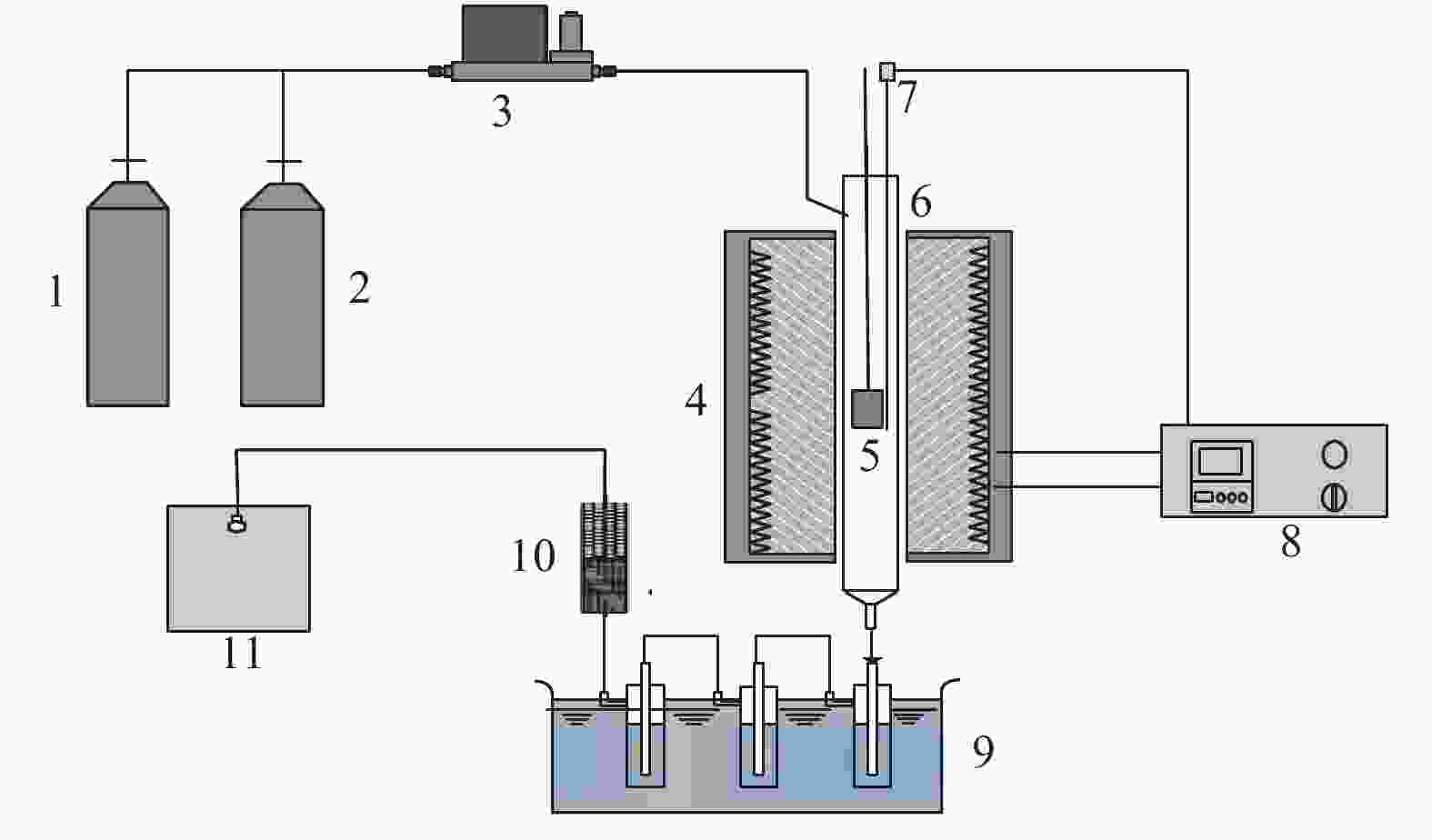
图 1 木质素快速热解实验装置示意图
Figure 1 Schematic diagram of experimental device for fast pyrolysis of lignin
1: nitrogen; 2: air; 3: mass flow controller; 4: furnace; 5: quartz hanging basket; 6: stainless steel tube; 7: thermocouple; 8: temperature controller; 9: ice-bath condenser; 10: drying tube; 11: gas bag
表 1 生物油主要成分的GM-MS分析
Table 1 GM-MS analysis of major compositions of bio-oil
RT.(min) Compound Peak area of compounds/ (× 107) 1∶0∶0 1∶1∶0 1∶1∶1 acid 6.742 acetic acid 0.70 0.78 0.85 phenol 14.019 phenol 2.91 2.67 2.42 methoxy phenol 15.099 phenol,2-methoxy- 3.93 2.51 1.13 17.315 phenol,2-methoxy-4-methyl- 2.77 1.41 0.90 19.192 phenol,4-ethyl-2-methoxy- 2.67 1.78 1.23 20.388 phenol,2-methoxy-4-vinyl 1.96 1.56 0.76 21.498 phenol,2,6-dimethoxy- 2.03 0.98 0.39 21.782 3-methoxy-5-methylphenol 0.41 0.52 − 23.658 phenol,4-methoxy-
3-(methoxymethyl)-1.83 0.84 0.76 Total 15.60 9.60 5.17 alkyl phenol 15.026 phenol,2-methyl- − 0.91 0.63 15.775 phenol,4-methyl- 2.01 1.95 1.61 16.834 phenol,2,4-dimethyl- 0.72 1.16 1.23 17.616 phenol,4-ethyl- 3.17 3.71 − 18.701 phenol,3-ethyl-5-methyl- 0.85 1.28 0.93 20.623 2-Allylphenol − 0.56 − total 6.75 9.57 4.4 aromatics 8.094 toluene − − 1.35 9.996 p-xylene − − 1.86 16.545 naphthalene − − 5.20 19.037 naphthalene,1-methyl- − − 3.91 total − − 12.32 -
[1] 赵宏志. 锰基钙钛矿型氧化物制备及其催化热解甘蔗渣木质素性能研究[D]. 大庆: 东北石油大学, 2019.ZHAO Hong-zhi. Preparation of manganese based perovskite oxide and its catalytic pyrolysis Performance of Bagasse Lignin[D]. Daqing: Northeast Petroleum University, 2019. [2] JOFFRES B, LORENTZ C, VIDALIE M, LAURENTI D, QUOINEAUD A A, CHARON N, DAUDIN A, QUIGNARD A, GEANTET C. Catalytic hydroconversion of a wheat straw soda lignin: Characterization of the products and the lignin residue[J]. Appl Catal B: Environ,2014,145:167−176. [3] 黄煜乾, 吴宇婷, 郑安庆, 赵增立, 李海滨. 基于Py-GC-MS的木质素与褐煤共热解特性研究[J]. 新能源进展,2017,5(5):333−340. doi: 10.3969/j.issn.2095-560X.2017.05.002HUANG Yi-qian, WU Yu-ting, ZHENG An-qing, ZHAO Zeng-li, LI Hai-bin. Co-pyrolysis characteristics of lignin and lignite: Analytical Py-GC-MS study[J]. Adv New Renewable Energy,2017,5(5):333−340. doi: 10.3969/j.issn.2095-560X.2017.05.002 [4] 王霏, 郑云武, 郑志锋. 云南松热解及其热解产物的研究[J]. 生物质化学工程,2015,49(4):14−18. doi: 10.3969/j.issn.1673-5854.2015.04.003WANG Fei, ZHENG Yun-wu, ZHENG Zhi-feng. Yields and compositions of products by pyrolysis of Yunnan pine[J]. Biomass Chem Eng,2015,49(4):14−18. doi: 10.3969/j.issn.1673-5854.2015.04.003 [5] 王则祥, 李航, 谢文銮, 胡斌, 李凯, 陆强. 木质素基本结构、热解机理及特性研究进展[J]. 新能源进展,2020,8(1):6−14. doi: 10.3969/j.issn.2095-560X.2020.01.002WANG Ze-xiang, LI Hang, XIE Wen-luan, HU Bin, LI Kai, LU Qiang. Progress in basic structure, pyrolysis mechanism and characteristics of lignin[J]. Adv New Renewable Energy,2020,8(1):6−14. doi: 10.3969/j.issn.2095-560X.2020.01.002 [6] 李金鑫. Fe基水滑石固体碱制备过程调控及其催化解聚木质素磺酸钙性能研究[D]. 大庆: 东北石油大学, 2019.LI Jin-xin. Regulation on preparation process of Iron based hydrotalcites solid alkali oxides and its catalytic performance for the depolymerization of calcium lignosulfonate[D]. Daqing: Northeast Petroleum University, 2019. [7] MA Z Q, TROUSSARD E, BOKHOVEN J A V. Controlling the selectivity to chemicals from lignin via catalytic fast pyrolysis[J]. Appl Catal A: Gen,2012,423:130−136. [8] 蒋丽群, 郑安庆, 王小波, 赵增立, 李海滨. 生物质定向快速热解制备左旋葡聚糖和芳烃的研究进展[J]. 新能源进展,2018,6(5):402−409. doi: 10.3969/j.issn.2095-560X.2018.05.010JIANG Li-qun, ZHENG An-qing, WANG Xiao-bo, ZHAO Zeng-li, LI Ha-bin. Progress of biomass fast pyrolysis to produce levoglucosan and aromatics[J]. Adv New Renewable Energy,2018,6(5):402−409. doi: 10.3969/j.issn.2095-560X.2018.05.010 [9] 舒日洋, 徐莹, 张琦, 马隆龙, 王铁军. 木质素催化解聚的研究进展[J]. 化工学报,2016,67(11):4523−4532.SHU Ri-yang, XU Ying, ZHANG Qi, MA Long-long, WANG Tie-jun. Progress in catalytic depolymerization of lignin[J]. J Chem Ind Eng,2016,67(11):4523−4532. [10] 郑云武. 木质生物质催化热解重整制备芳烃化合物的研究[D]. 哈尔滨: 东北林业大学, 2017.ZHENG Yun-wu. Study on aromatics production from catalytic pyrolysis upgraded of ligncellulose[D]. Haerbin: Northeast Foresttry University, 2017. [11] ZHANG M, RESENDE F L P, MOUTSOGLOU A. Catalytic fast pyrolysis of aspen lignin via Py-GC/MS[J]. Fuel,2014,116:358−369. [12] 杨海平, 陈汉平, 杜胜磊, 陈应泉, 王贤华, 张世红. 碱金属盐对生物质三组分热解的影响[J]. 中国电机工程学报,2009,29(17):70−75. doi: 10.3321/j.issn:0258-8013.2009.17.012YANG Hai-ping, CHEN Han-ping, DU Sheng-lei, CHEN Ying-quan, WANG Xian-hua, ZHANG Shi-hong. Influence of alkali salts on the pyrolysis of biomass three components[J]. Proc CSEE,2009,29(17):70−75. doi: 10.3321/j.issn:0258-8013.2009.17.012 [13] 刘心明. Ca/Fe强化生物质炭催化重整玉米秸秆挥发分的试验研究[D]. 包头: 内蒙古科技大学, 2019.LIU Xin-ming. Expeimental Study on pyrolysis of volatiles from corn straw by Ca/Fe enhanced biomass carbon catalytic reforming[D]. Baotou: Inner Mongolia University Science and Technology, 2019. [14] KUMAR G R, DUBEY M, KHAREL P, GU Z R, FAN Q H. Biochar activated by oxygen plasma for supercapacitors[J]. J Power Sources,2015,274(C):1300−1305. [15] SUN K, HUANG Q X, CHI Y, YAN J H. Effect of ZnCl2-activated biochar on catalytic pyrolysis of mixed waste plastics for producing aromatic-enriched oil[J]. Waste Manage,2018,81:128−137. [16] NGUYEN H K D, PHAM V V, DO H T. Preparation of Ni/biochar catalyst for hydrotreating of bio-oil from microalgae biomass[J]. Catal Lett,2016,146(11):2381−2391. [17] ZHONG C L, WEI X M. A comparative experimental study on the liquefaction of wood[J]. Energy,2004,29(11):1731−1741. [18] 于树峰, 仲崇立. 农作物废弃物液化的实验研究[J]. 燃料化学学报,2005,33(2):205−210. doi: 10.3969/j.issn.0253-2409.2005.02.016YU Shu-feng, YU Chong-li. Experimental study on liquefaction of agricultural residue[J]. J Fuel Chem Technol,2005,33(2):205−210. doi: 10.3969/j.issn.0253-2409.2005.02.016 [19] LIU S Y, XIE Q L, ZHANG B, CHENG Y L, LIUY H, CHEN P, RUAN R. Fast microwave-assisted catalytic co-pyrolysis of corn stover and scum for bio-oil production with CaO and HZSM-5 as the catalyst[J]. Bioresour Technol,2016,204:164−170. [20] DICKERSON T, SORIA J. Catalytic Fast Pyrolysis: A Review[J]. Energies,2013,6(1):514−538. [21] MULLEN C A, BOATENG A A. Catalytic pyrolysis-GC/MS of lignin from several sources[J]. Fuel Process Technol,2010,91(11):1446−1458. [22] LIU Q Y, HU C S, PENG B, LIU C, LI Z W, WU K, ZHANG H Y, XIAO R. High H2/CO ratio syngas production from chemical looping co-gasification of biomass and polyethylene with CaO/Fe2O3 oxygen carrier[J]. Energy Conv Manag,2019,199:111951. [23] JACKSON M A, COMPTON D L, BOATENG A A. Screening heterogeneous catalysts for the pyrolysis of lignin[J]. J Anal Appl Pyrolysis,2008,85(1):226−230. [24] 李润东, 张杨, 李秉硕, 刘合鑫, 开兴平, 烟征, 杨天华. 玉米秸秆催化液化制备生物油实验研究[J]. 燃料化学学报,2016,44(1):69−75. doi: 10.3969/j.issn.0253-2409.2016.01.010LI Run-dong, ZHANG Yang, LI Bing-shuo, LIU He-xin, KAI Xing-ping, YAN Zheng, YANG Tian-hua. Hydrothe rmal catalytic lique faction of corn stalk for preparation of bio-oil[J]. J Fuel Chem Technol,2016,44(1):69−75. doi: 10.3969/j.issn.0253-2409.2016.01.010 [25] TOOR S S, ROSENDAHL L, RUDOLF A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies[J]. Energy,2011,36(5):2328−2342. [26] IOANNIDOU O, ZABANIOTOU A, ANTONAKOU E V, PAPAZISI K M, LAPPAS A A, ATHANASSIOU C. Investigating the potential for energy, fuel, materials and chemicals production from corn residues (cobs and stalks) by non-catalytic and catalytic pyrolysis in two reactor configurations[J]. Renewable Sustainable Energy Rev,2008,13(4):750−762. [27] JIN S H, LEE H W, RYU C, JEON J K, PARK Y K. Catalytic fast pyrolysis of Geodae-Uksae 1 over zeolites[J]. Energy,2015,81:41−46. [28] LEE H W, KIM Y M, JAE J, SUNG B H, JUNG S C, KIM J K, CHAI J S, PARK Y K. Catalytic pyrolysis of lignin using a two-stage fixed bed reactor comprised of in-situ natural zeolite and ex-situ HZSM-5[J]. J Anal Appl Pyrolysis,2016,122:282−288. -




 下载:
下载: