Effect of flotation fractions of long-flame coal on regulation of sulfur and coke reactivity during pyrolysis of high-sulfur coking coal
-
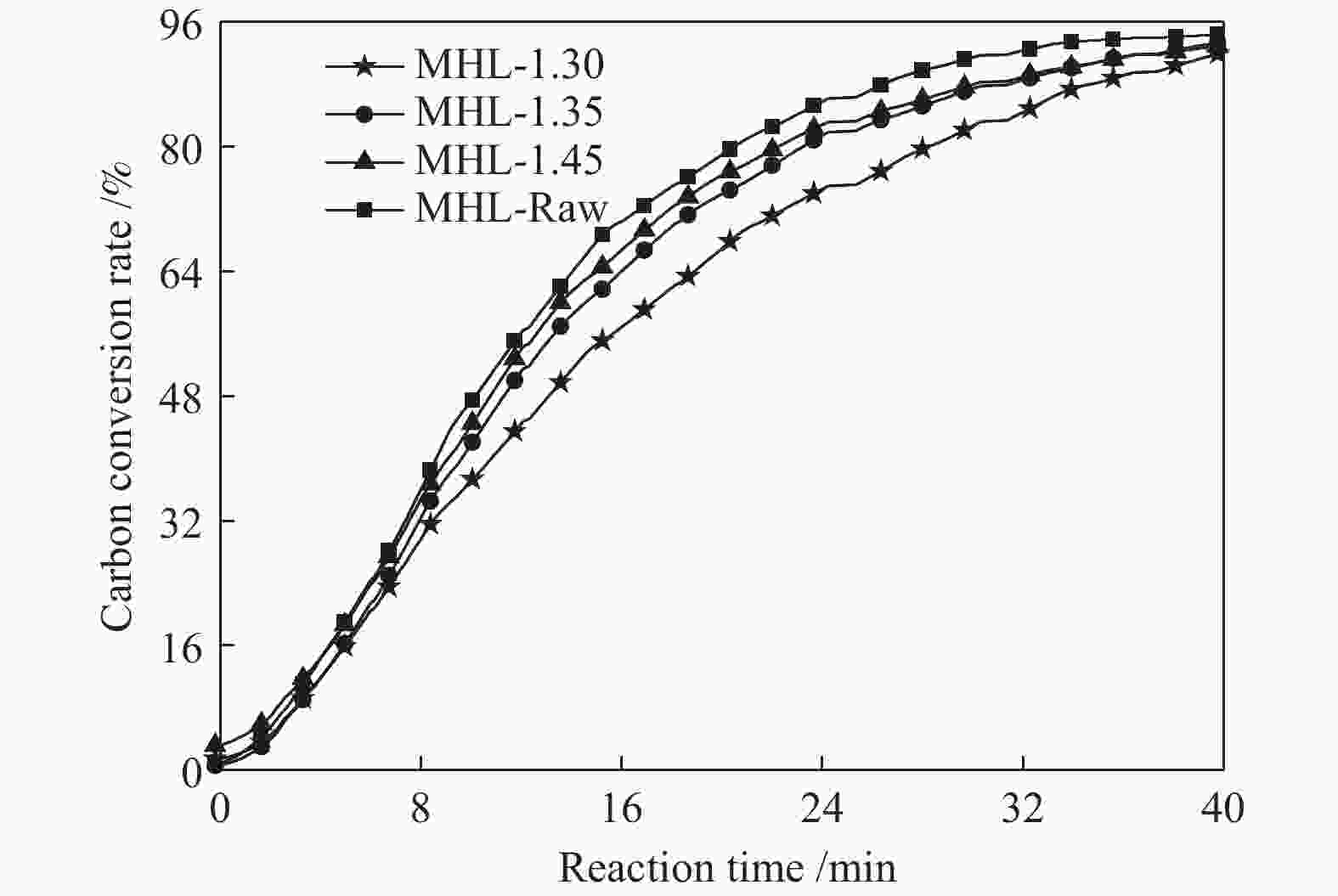
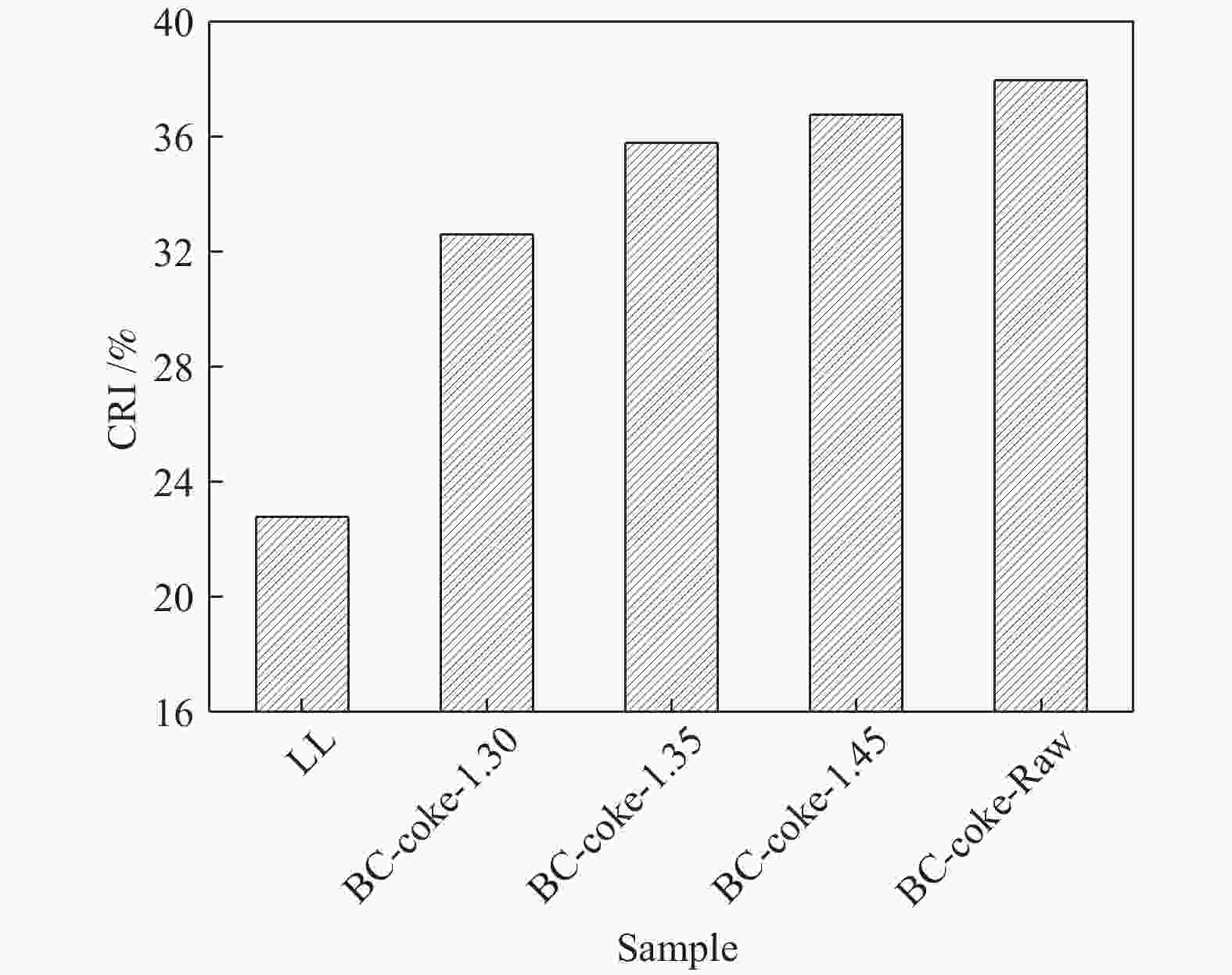
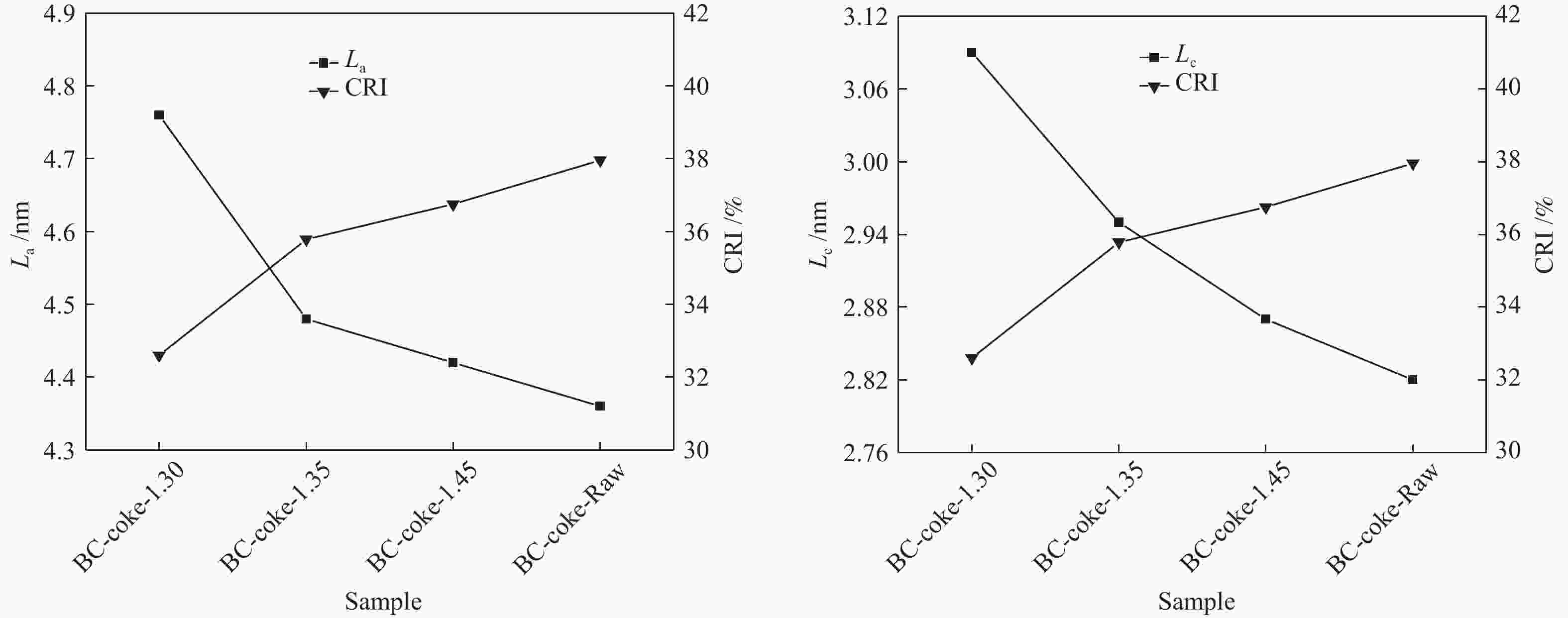
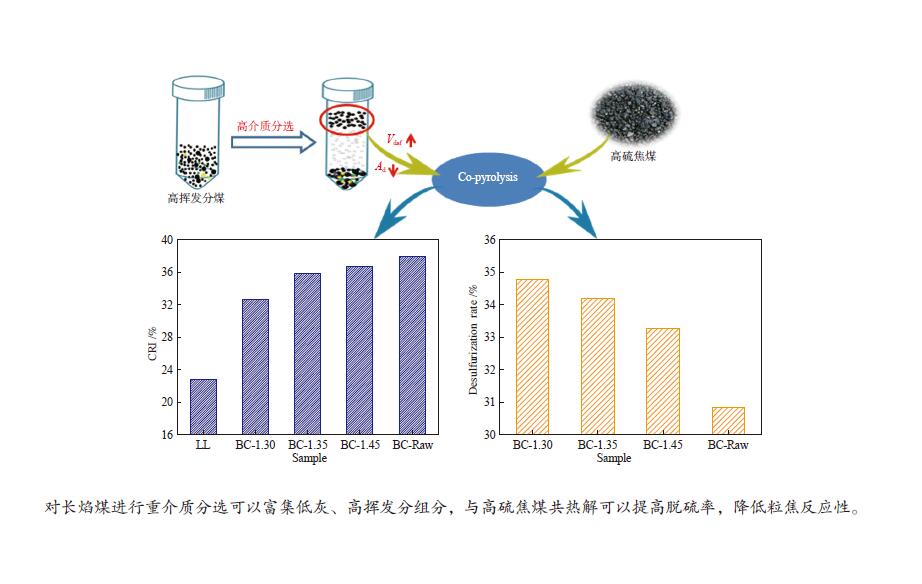
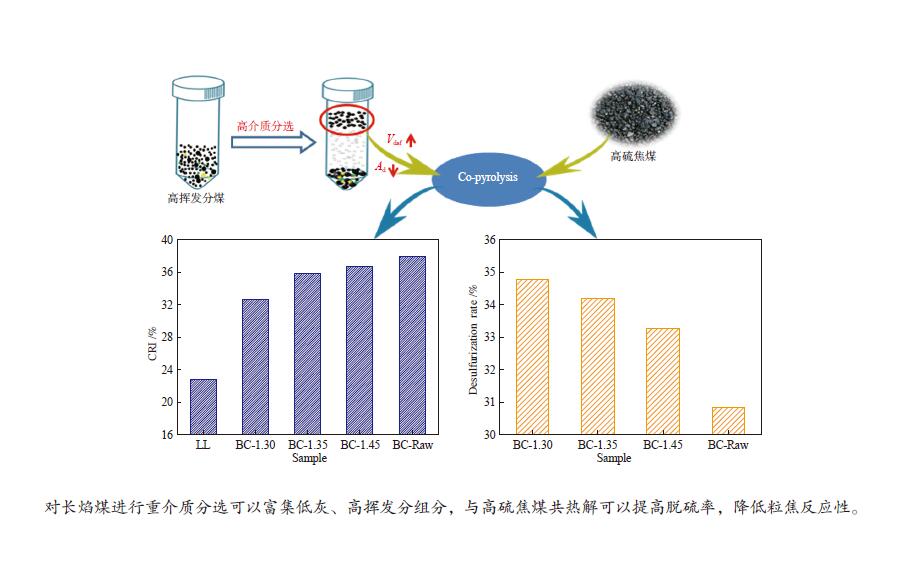
摘要: 采用重介质分选法得到长焰煤不同密度级分选组分,利用红外、拉曼、热重、基式流动度、静态氮吸附仪、X射线衍射仪等手段分析研究了不同分选组分对高硫炼焦煤热解过程中硫变迁及焦反应性的影响。结果表明,低密度组分含有较多的脂肪侧链,结构有序度低,而矿物质和惰质组则富集于高密度组分中。低密度级组分由于碱性矿物质含量少,脂肪侧链多,与高硫炼焦煤共热解的脱硫率明显高于高密度组分。低密度组分中的中等分子量组分对胶质体的性质影响较小,高密度组分的矿物质和惰性组分对胶质体的劣化作用更加明显,同时使焦样的微晶结构有序度降低,缺陷位增多,粒焦的反应性升高。Abstract: The flotation fractions of a long-flame coal were obtained by heavy medium separation method, and its effect on regulation of sulfur and coke reactivity during pyrolysis of high-sulfur coking coal were investigated by FT-IR, Raman, TG, Gieseler fluidity, N2 adsorption, XRD. The results show that the low density fractions contain more aliphatic side chains and unstable aliphatic structure, while the high-density fractions show higher amount of minerals and inert components. Low density fractions have the highest sulfur removal rate due to lower content of alkaline minerals and more aliphatic side chains. The medium molecular weight component in the low density fractions has little effect on properties of metaplast. Higher minerals and inert components in high-density fractions deteriorate the metaplast more obviously. Meanwhile, order of the coke's microcrystalline structure is reduced and the defect sites increase, and consequently, reactivity of the coke increases.
-
Key words:
- long-flame coal /
- flotation fraction /
- pyrolysis /
- coke reactivity /
- sulfur content
-
表 1 实验用煤的分析数据
Table 1 Analysis parameter of coal samples
Sample Proximate analysis w/% Ultimate analysis w/% G Y/mm Mad Ad Vdaf Cdaf Hdaf Ndaf Sd O* LL 0.20 9.76 21.50 88.49 4.69 1.42 1.94 3.25 85.00 16.50 MHL 3.54 4.90 37.63 81.61 4.99 1.07 0.34 11.97 10.00 − note: ad: air dried basis; d: dry basis; daf: dry and ash-free basis; *: by difference 表 2 实验用煤的灰成分分析
Table 2 Ash composition of coal samples
Sample Ash composition w/% R SiO2 Al2O3 Fe2O3 CaO MgO TiO2 SO3 K2O Na2O P2O5 LL 48.70 38.96 4.79 1.80 0.18 1.52 1.16 0.16 0.22 0.72 0.08 MHL 26.58 13.26 11.27 21.76 3.13 0.50 19.61 0.21 0.31 0.07 0.92 表 3 MHL分选组分的基本分析数据及其收率
Table 3 Analysis parameter and yield of MHL flotation fractions
Sample Proximate analysis w/% Ultimate analysis w/% MCI/% Yield/% Mad Ad Vdaf Cdaf Hdaf Ndaf Sd O* MHL-1.30 2.38 0.90 39.62 81.66 5.28 1.16 0.16 11.73 0.64 54.40 MHL-1.35 2.34 1.12 38.87 81.96 5.21 1.16 0.16 11.50 0.71 79.56 MHL-1.45 2.26 1.84 37.54 82.16 5.14 1.13 0.17 11.40 1.07 90.88 MHL-Raw 3.54 4.90 37.63 81.61 4.99 1.07 0.34 11.97 4.83 100.00 note: ad: air dried basis; d: dry basis; daf: dry and ash-free basis; *: by difference 表 4 MHL分选组分的煤岩分析
Table 4 Petrographic analysis of MHL flotation fractions
Sample Vitrinite
/%Liptinite
/%Inertinite
/%Mineral
/%Rmax MHL-1.30 83.81 0.45 15.59 0.15 0.53 MHL-1.35 73.76 0.30 25.64 0.30 0.60 MHL-1.45 65.31 0.27 33.74 0.68 0.56 MHL-Raw 60.34 0.26 38.11 1.29 0.51 note: Rmax: mean maximum vitrinite reflectance 表 5 MHL分选组分红外和拉曼结构参数
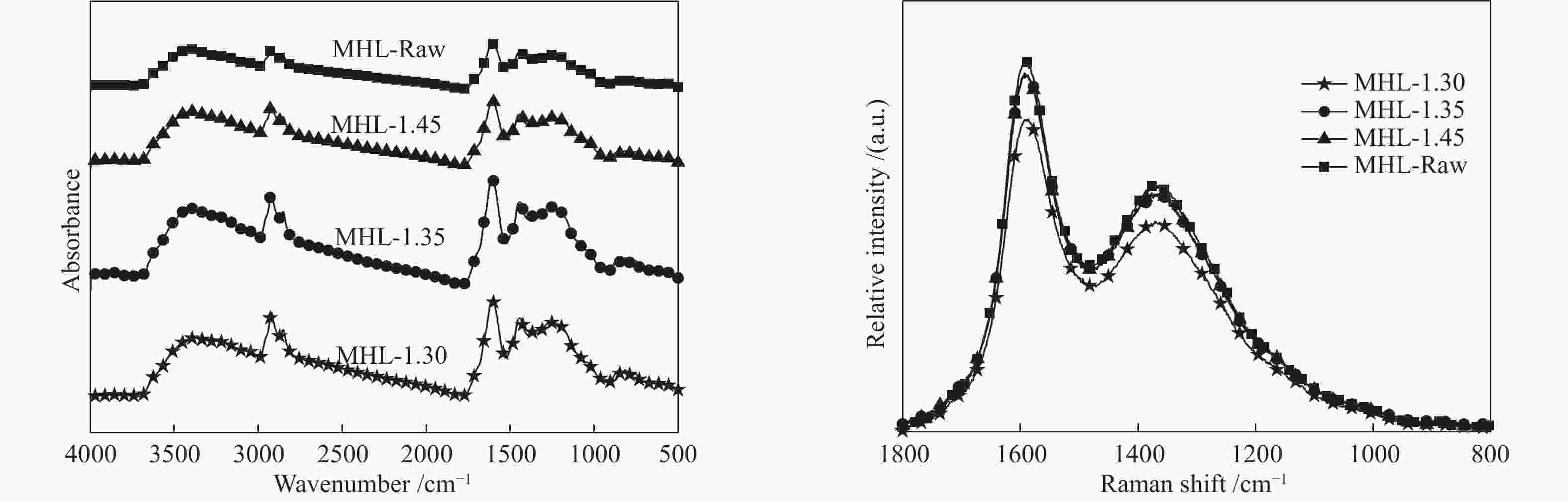
Table 5 Structural parameters of FT-IR and Raman spectra of MHL flotation fractions
Sample fa I1 I2 I(Gr+Vl+Vr)/ID MHL-1.30 0.71 0.48 1.67 4.07 MHL-1.35 0.72 0.52 1.57 3.37 MHL-1.45 0.73 0.53 1.32 3.28 MHL-Raw 0.74 0.54 1.19 2.50 表 6 MHL分选组分与LL配煤的基氏流动度参数
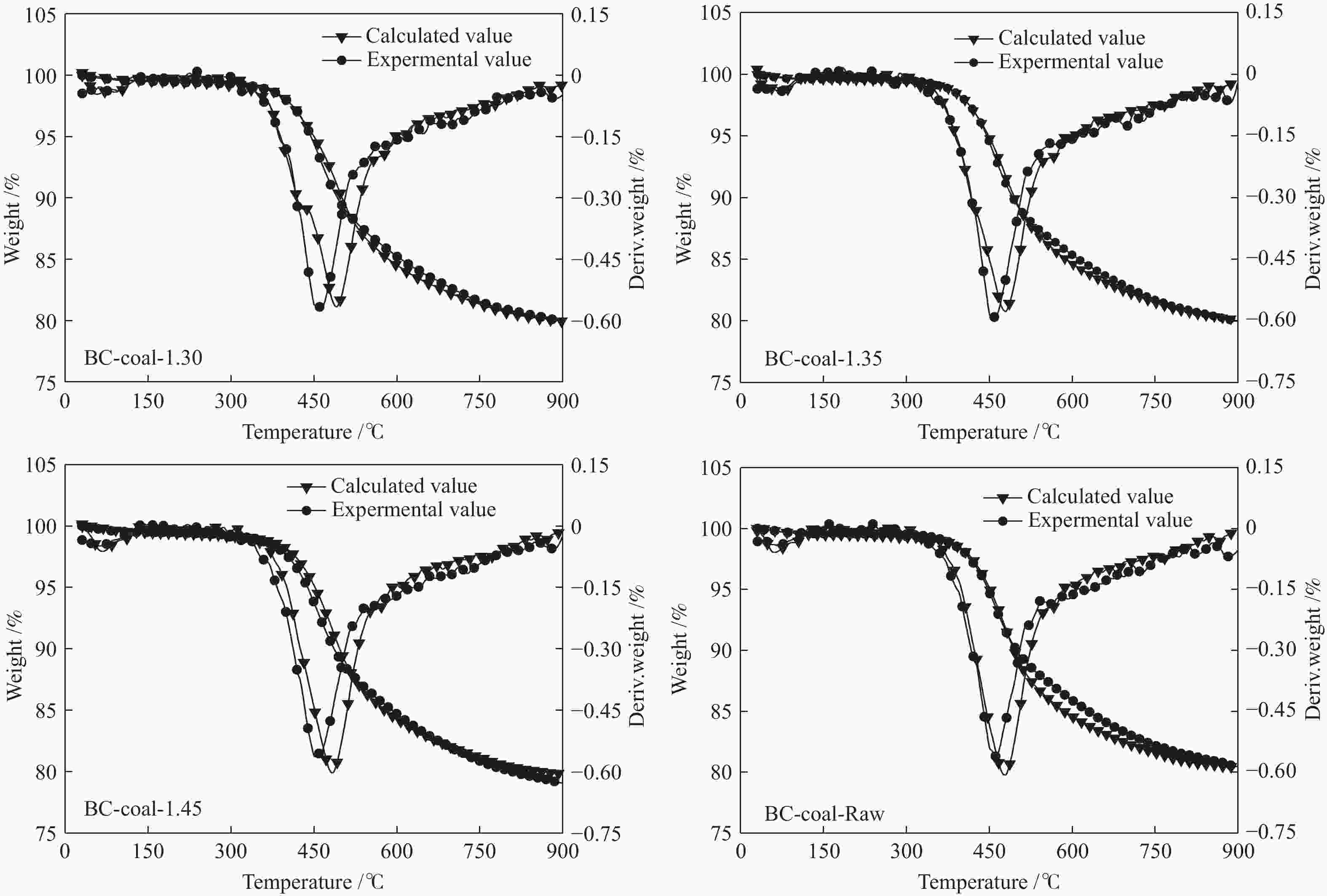
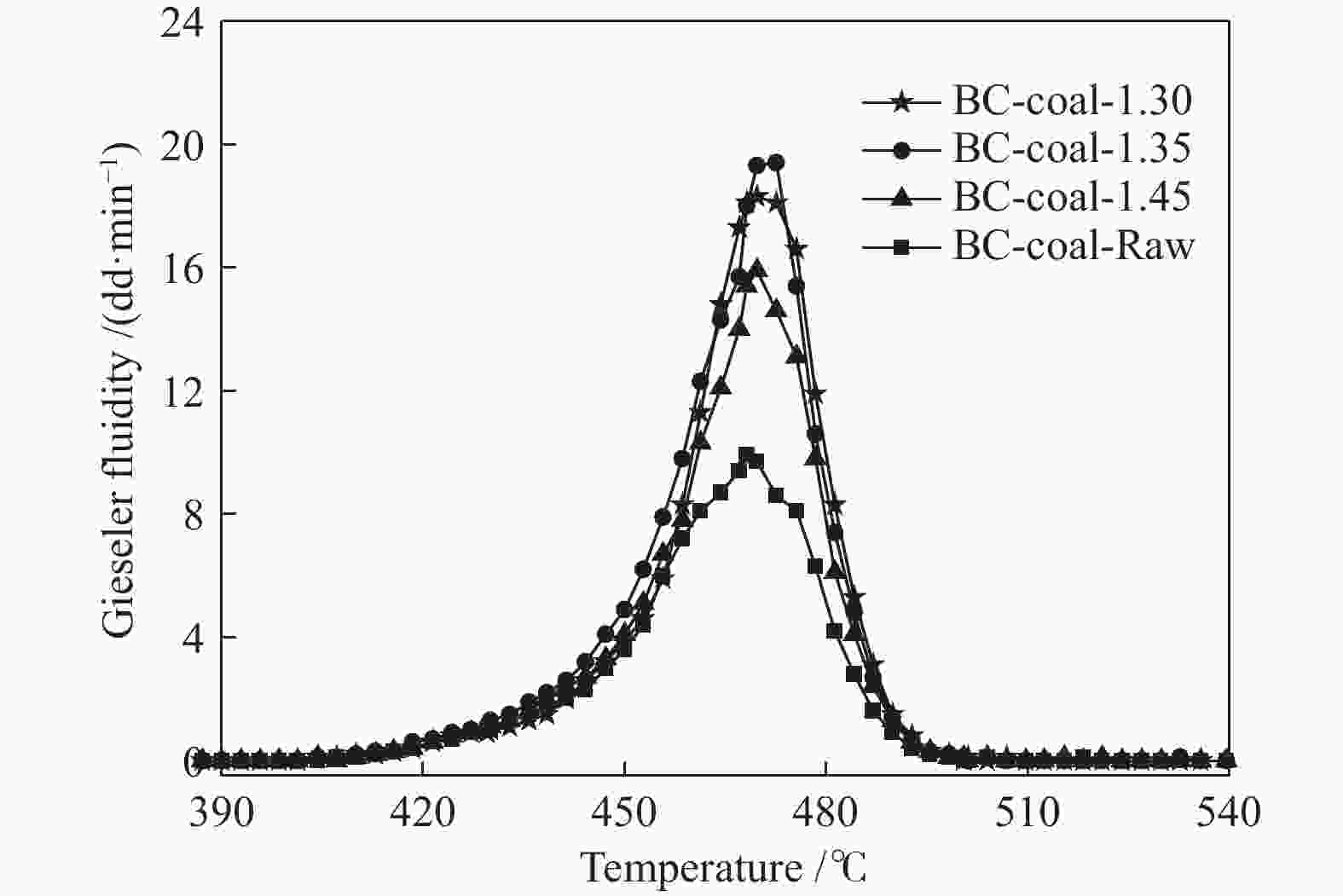
Table 6 Gieseler fluidity parameters of coal blends of MHL flotation fractions and LL coal
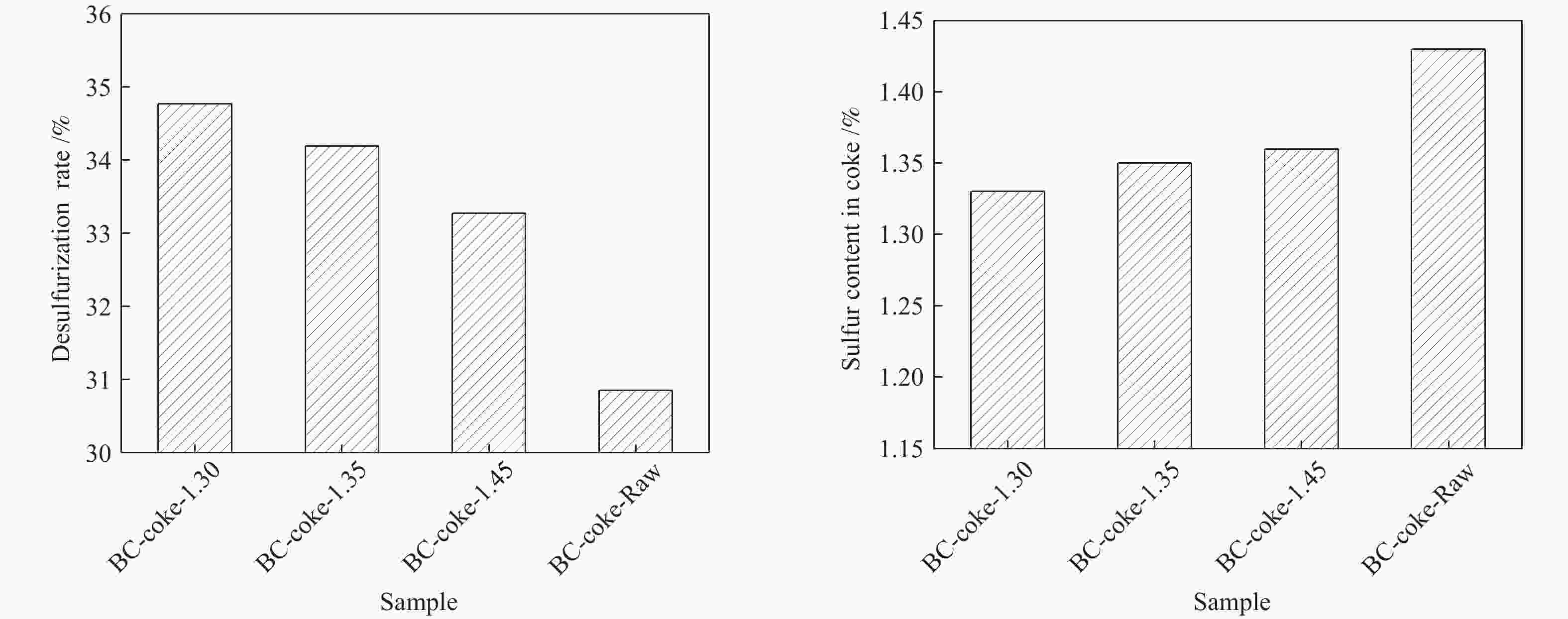
Sample t1/℃ t2/℃ t3/℃ Δt/℃ Fmax/(dd·min−1) LL 432.1 474.9 511.5 79.4 244.7 BC-coal-1.30 432.1 472.9 502.0 69.9 18.3 BC-coal-1.35 427.3 474.3 504.8 76.5 19.4 BC-coal-1.45 428.1 471.4 501.9 73.8 15.9 BC-coal-Raw 429.5 468.2 500.5 71.0 9.9 note: t1: softening temperature; t2: max fluidity temperature; t3: resolidification temperature; Δt: plastic range; Fmax: maximum fluidity 表 7 MHL不同分选组分与LL煤配煤焦的气孔结构参数
Table 7 Pore structure parameters of coal blend cokes from pyrolysis of LL coal and MHL flotation fractions
Sample BC-coke-
1.30BC-coke-
1.35BC-coke-
1.45BC-coke-
RawSBET/(m2·g−1) 4.26 6.82 4.95 3.16 rBJH/nm 5.47 4.81 5.52 6.76 vBJH/(mm3·g−1) 1.79 1.77 2.32 1.79 -
[1] WANG H, FENG Y, ZHANG X, LIN W, ZHAO Y. Study of coal hydropyrolysis and desulfurization by ReaxFF molecular dynamics simulation[J]. Fuel,2015,145:241−248. doi: 10.1016/j.fuel.2014.12.074 [2] LIAO H, LI B, ZHANG B. Pyrolysis of coal with hydrogen-rich gases. 2 desulfurization and denitrogenation in coal pyrolysis under coke-oven gas and synthesisgas[J]. Fuel,1998,77:1643−1646. doi: 10.1016/S0016-2361(98)00076-3 [3] GUO Z, TANG H, LIU J. Desulfurization of coke by recycling COG in coking process[J]. Fuel,2005,84:893−901. doi: 10.1016/j.fuel.2004.11.020 [4] LIU Q, HU H, ZHU S, ZHOU Q, Li W, WEI X, XIE K. Desulfurization of coal by pyrolysis and hydropyrolysis with addition of KOH/NaOH[J]. Energy Fuels,2005,19:1673−1678. doi: 10.1021/ef0497053 [5] 周仕学, 聂西文, 王荣春, 刘泽常. 高硫强粘结性煤与生物质共热解的研究[J]. 燃料化学学报,2000,28(3):294−297. doi: 10.3969/j.issn.0253-2409.2000.04.002ZHOU Shi-xue, NIE Xi-wen, WANG Rong-chun, LIU Ze-chang. Study on co-pyrolysis of high sulfur and strongly caking coal with biomass[J]. J Fuel Chem Technol,2000,28(3):294−297. doi: 10.3969/j.issn.0253-2409.2000.04.002 [6] 么秋香, 杜美利, 张锦仁. 高硫煤与生物质共热解脱有机硫研究[J]. 煤炭转化,2014,37:15−19. doi: 10.3969/j.issn.1004-4248.2014.01.004YAO Qiu-xiang, DU Mei-li, ZHANG Jin-ren. Organic sulfur removal from high sulfur coal during co-pyrolysis with biomass[J]. Coal Convers.,2014,37:15−19. doi: 10.3969/j.issn.1004-4248.2014.01.004 [7] GUAN R, LI W, LI B. Effects of Ca-based additives on desulfurization during coal pyrolysis[J]. Fuel,2003,82:1961−1966. [8] IBARRA J V, PALACIOS J M, MOLINER R, BONET A J. Evidence of the reciprocal organic matter-pyrite interactions affecting sulfur removal during coal pyrolysis[J]. Fuel,1993,72(5):697−698. [9] WANG M, LIU L, WANG J, CHANG L, WANG H, HU Y. Sulfur K-edge XANES study of sulfur transformation during pyrolysis of four coals with different ranks[J]. Fuel Process Technol,2015,131:262−269. doi: 10.1016/j.fuproc.2014.10.038 [10] SHEN Y, WANG M, WU Y, HU Y, KONG J, DUAN X, WANG J, CHANG L, BAO W. Role of gas coal in directional regulation of sulfur during coal blending coking of high organic-sulfur coking coal[J]. Energy Fuels,2020,34(3):2757−2764. doi: 10.1021/acs.energyfuels.9b03737 [11] 张有芝. 瘦煤和长焰煤在捣固焦炉配煤中的优化配合[J]. 山西冶金,2006,(3):41−42. doi: 10.3969/j.issn.1672-1152.2006.03.016ZHANG You-zhi. Optimum blending of carbonaceous coal and long flame coal on stamt-charging coke oven[J]. Shanxi Metall,2006,(3):41−42. doi: 10.3969/j.issn.1672-1152.2006.03.016 [12] SHAO Y. Preliminary study on weakly caking coal blending coking and production practice[J]. Clean Coal Technol,2005,11(3):41−44. [13] 杨俊和, 杜鹤桂, 钱湛芬, 崔平. 焦炭的粒焦反应性[J]. 东北大学学报,1999,20(3):286.YANG Jun-he, DU He-gui, QIAN Zhan-feng, CUI Ping. Reactivity of particulate coke[J]. J Dongbei Univ,1999,20(3):286. [14] ORREGO-RUIZ J A, CABANZO R, MEJIA-OSPINO E. Study of Colombian coals using photoacoustic Fourier transform infrared spectroscopy[J]. Int J Coal Geol,2011,85(3):307−310. [15] HE X, LIU X, NIE B, SUN D. FT-IR and Raman spectroscopy characterization of functional groups in various rank coals[J]. Fuel,2017,206(15):555−563. [16] LI K, KHANNA R, ZHANG J, BARATI M, LIU Z, XU T, YANG T, SAHAJWALLA V. Comprehensive investigation of various structural features of bituminous coals using advanced analytical techniques[J]. Energy Fuels,2015,29(11):7178−7189. doi: 10.1021/acs.energyfuels.5b02064 [17] BAYSAL M, YURUM A, YILDIZ B, YURUM Y. Structure of some western Anatolia coals investigated by FT-IR, Raman, 13C solid state NMR spectroscopy and X-ray diffraction[J]. Int J Coal Geol,2016,163:166−176. doi: 10.1016/j.coal.2016.07.009 [18] WANG M, TIAN J, ROBERTS D G, CHANG L, XIE K. Interactions between corncob and lignite during temperature-programmed co-pyrolysis[J]. Fuel,2015,142:102−108. doi: 10.1016/j.fuel.2014.11.003 [19] JIAO H, WANG M, KONG J, YAN D, GUO J, CHANG L. Contribution of single coal property to the changes of structure and reactivity of chars from blending coking[J]. J Anal Appl Pyrolysis,2018,134:114−121. doi: 10.1016/j.jaap.2018.05.016 [20] 高岩, 鲁光辉. 煤与生物质共热解的协同特性研究[J]. 洁净煤技术,2013,19(3):53−56.GAO Yan, LU Guang-hui. Collaborative characteristics of coal and biomass co-pyrolysis[J]. Clean Coal Technol,2013,19(3):53−56. [21] MONDRAGON F, JARAMILLO A, SALDARRIAGA F, QUINTERO G, FERNANDEZ J, RUIZ W, HALL P. The effects of morphological changes and mineral matter on H2S evolution during coal pyrolysis[J]. Fuel,1999,78(15):1841−1846. doi: 10.1016/S0016-2361(99)00096-4 [22] 忻仕河, 徐振刚. 大同煤不同显微组分富集物焦与CO2反应性研究[J]. 煤炭转化,2004,27(4):13−16. doi: 10.3969/j.issn.1004-4248.2004.04.003XIN Shi-he, XU Zhen-gang. Research into reactivity of char from coal maceral concentrates during gasification with CO2[J]. Coal Convers,2004,27(4):13−16. doi: 10.3969/j.issn.1004-4248.2004.04.003 [23] 谢克昌. 煤的结构与反应性[M]. 北京: 科学出版社, 2002.XIE Ke-chang. Structure and Reactivity of Coal[M]. Beijing: Science of Press, 2002. [24] XIE W, STANGER R, TRAN Q A, SMITH N, WALL T, LUCAS J. Impact of coal pyrolysis products as a rheological additive on thermoplasticity of a coking coal[J]. Energy Fuels,2018,32:4382−4390. doi: 10.1021/acs.energyfuels.7b03232 [25] CASCAL M, DIAZ-FAES E, ALVAREZ R. Influence of the permeability of the coal plastic layer on coking pressure[J]. Fuel,2006,85(3):281−288. doi: 10.1016/j.fuel.2005.06.009 [26] 杨志荣, 孟庆岩, 黄戒介, 王志青, 李春玉, 房倚天. 神木煤与不同黏结煤共热解交互作用规律的研究[J]. 燃料化学学报,2018,46(6):641−648. doi: 10.3969/j.issn.0253-2409.2018.06.001YANG Zhi-rong, MENG Qing-yan, HUANG Jie-jie, WANG Zhi-qing, LI Chun-yu, FANG Yi-tian. Interaction between Shenmu coal and different caking coals during co-pyrolysis[J]. J Fuel Chem Technol,2018,46(6):641−648. doi: 10.3969/j.issn.0253-2409.2018.06.001 [27] DIEZ M A, BARRIOCANAL C, ALVAREZ R. Plastic wastes as modifiers of the thermoplasticity of coal[J]. Energy Fuels,2005,19(6):2304−2316. doi: 10.1021/ef0501041 -




 下载:
下载: