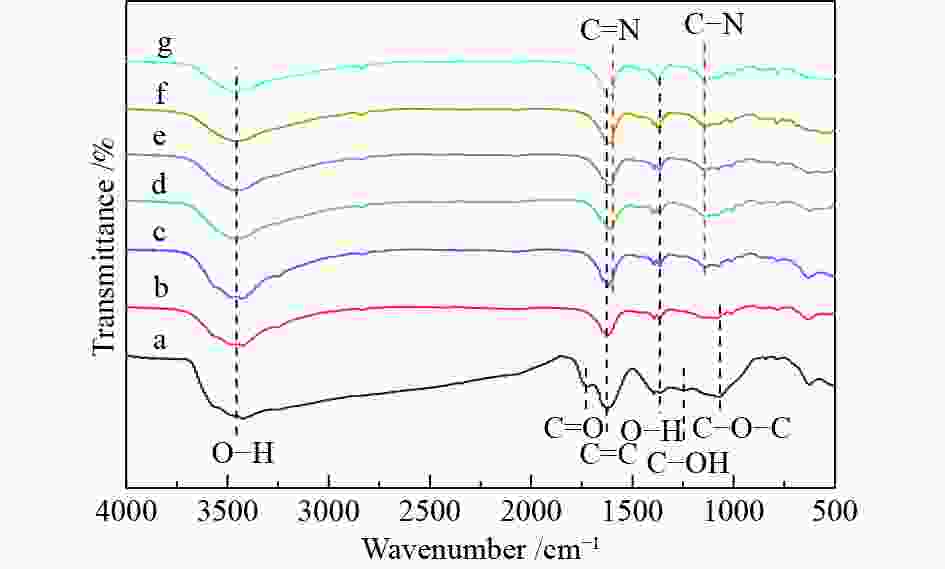
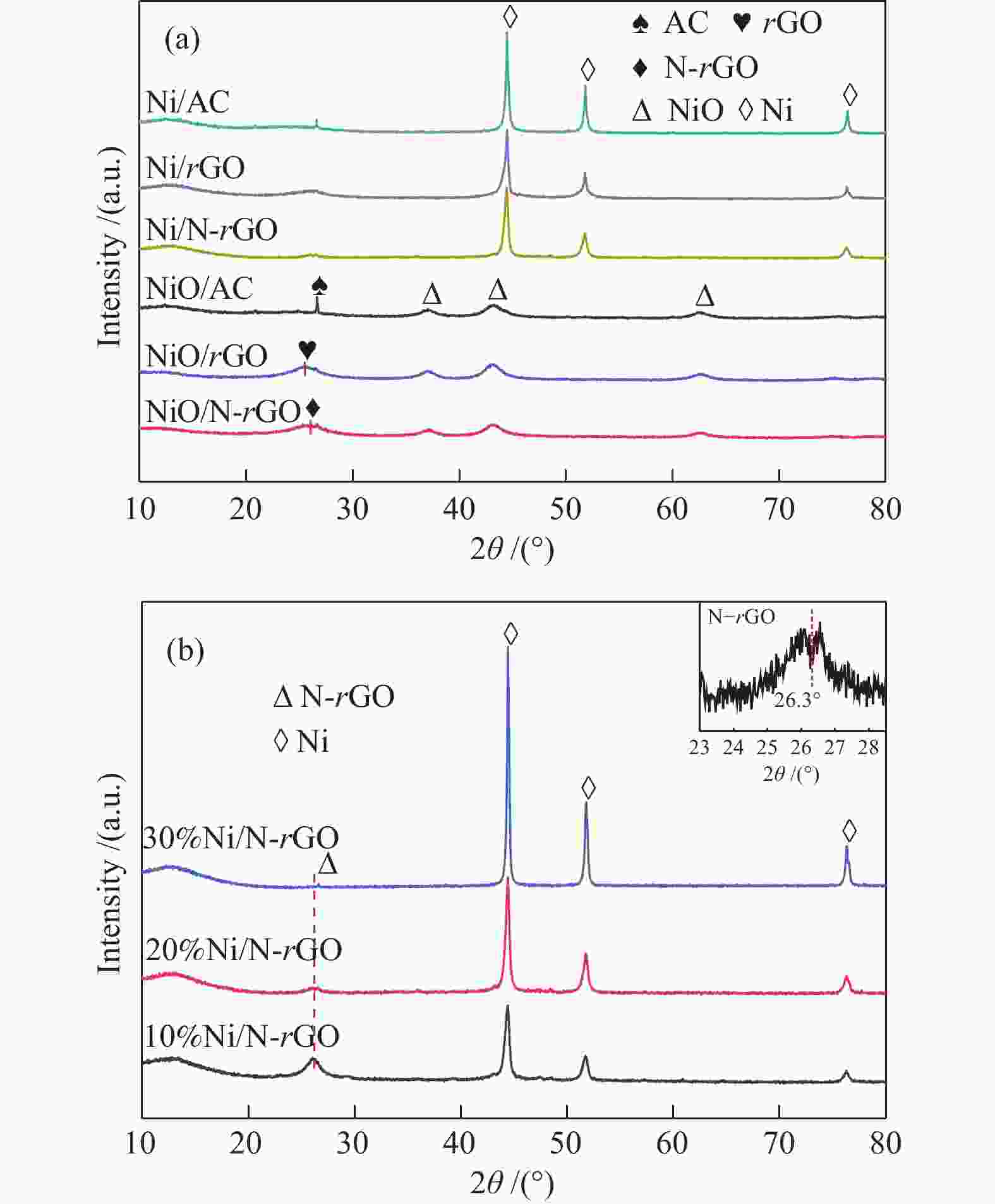
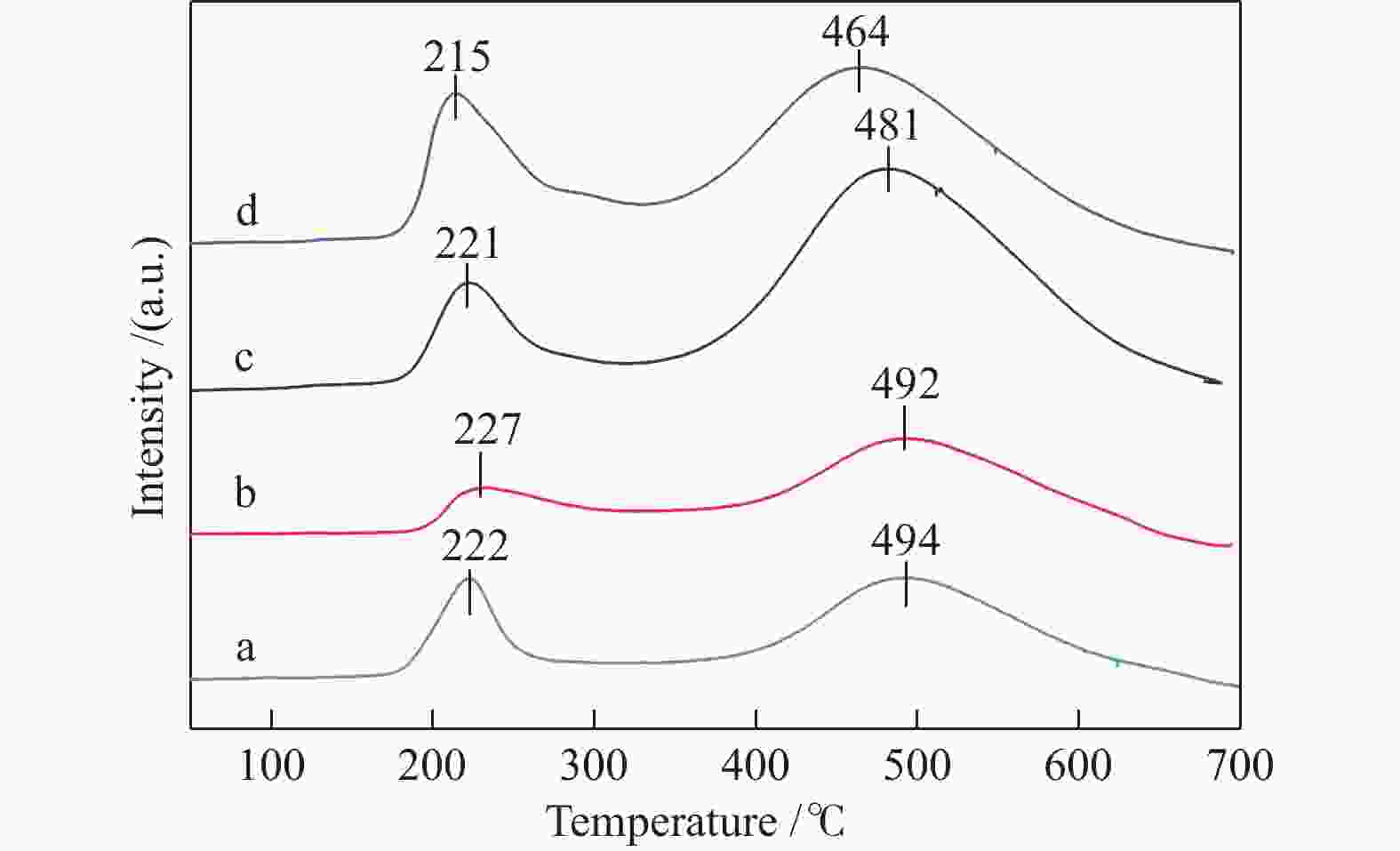
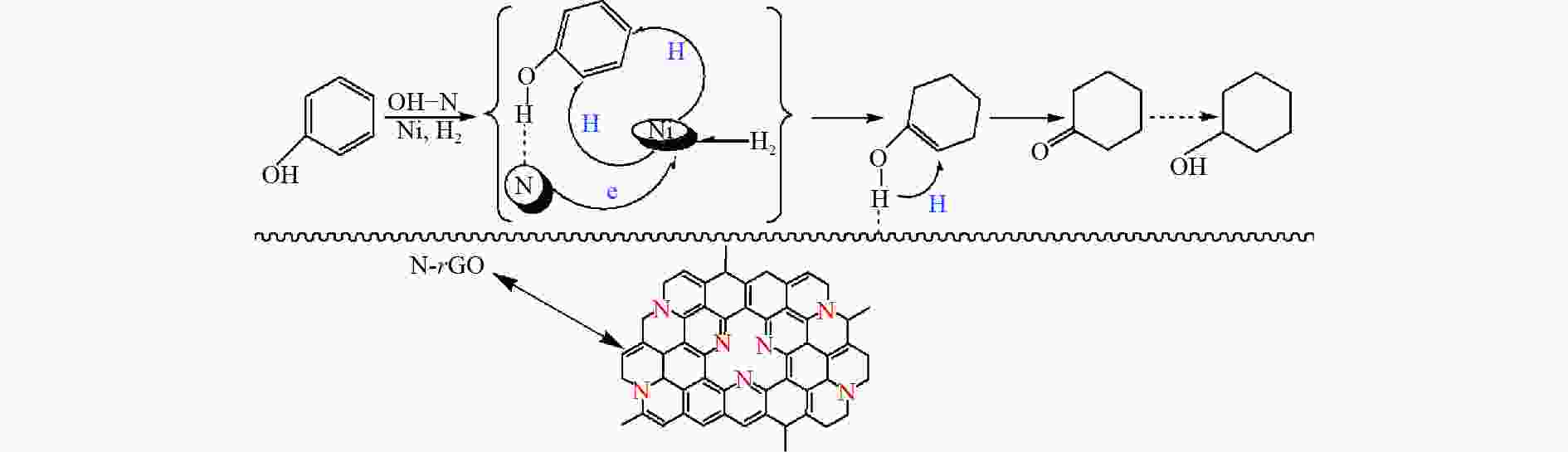
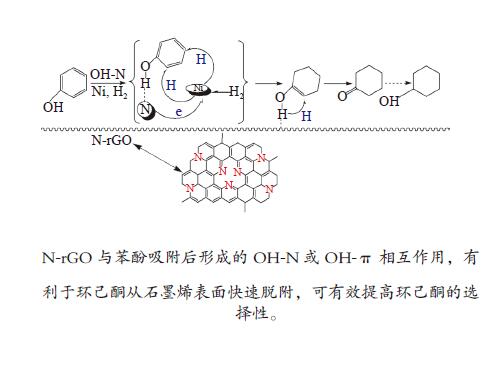
Characterization of Ni/N-rGO catalyst and study on the hydrogenation performance of phenol
-
摘要: 以325目鳞片石墨为原料,采用改进Hummers法合成氧化石墨(GO),再以尿素为氮源,通过水热法制备了氮掺杂还原氧化石墨烯(N-rGO)载体,然后通过浸渍法制备出负载型Ni/N-rGO催化剂。通过N2吸附-脱附、XRD、SEM、H2-TPR等测试方法对所制备的催化剂进行表征分析。以苯酚选择性加氢制备环己酮为反应体系,在反应温度150 ℃、压力0.4 MPa、时间2.0 h的条件下,对比了N-rGO、还原氧化石墨烯(rGO)、活性炭(AC)等载体负载Ni催化剂的加氢性能。结果表明,N-rGO载体不但具有较大的比表面积和适宜的孔结构,而且N-rGO和金属镍之间有较好的协同作用,Ni/N-rGO催化剂展现出优异的催化活性和选择性。Abstract: Using 325 mesh flake graphite as raw material, graphite oxide (GO) was synthesized by improved Hummers method. Nitrogen-doped reduced graphene oxide (N-rGO) carrier was prepared by hydrothermal method using urea as nitrogen source, and then supported Ni/N-rGO catalyst was prepared by impregnation method. The prepared catalysts were characterized and analyzed by N2 adsorption-desorption, XRD, SEM, H2-TPR and other methods. The hydrogenation performance of Ni catalysts supported by N-rGO, reduced graphene oxide (rGO) and activated carbon (AC) were compared under the reaction conditions of 150 ℃, 0.4 MPa and 2.0 h with selective hydrogenation of phenol to cyclohexanone as probe reaction. The results show that the N-rGO support not only has a large specific surface area and suitable pore structure, but also has good synergistic effect with nickel metal, and the Ni/N-rGO catalyst thus shows excellent catalytic activity and selectivity.
-
Key words:
- N-rGO carrier /
- catalytic hydrogenation /
- phenol /
- cyclohexanone /
- reaction mechanism
-
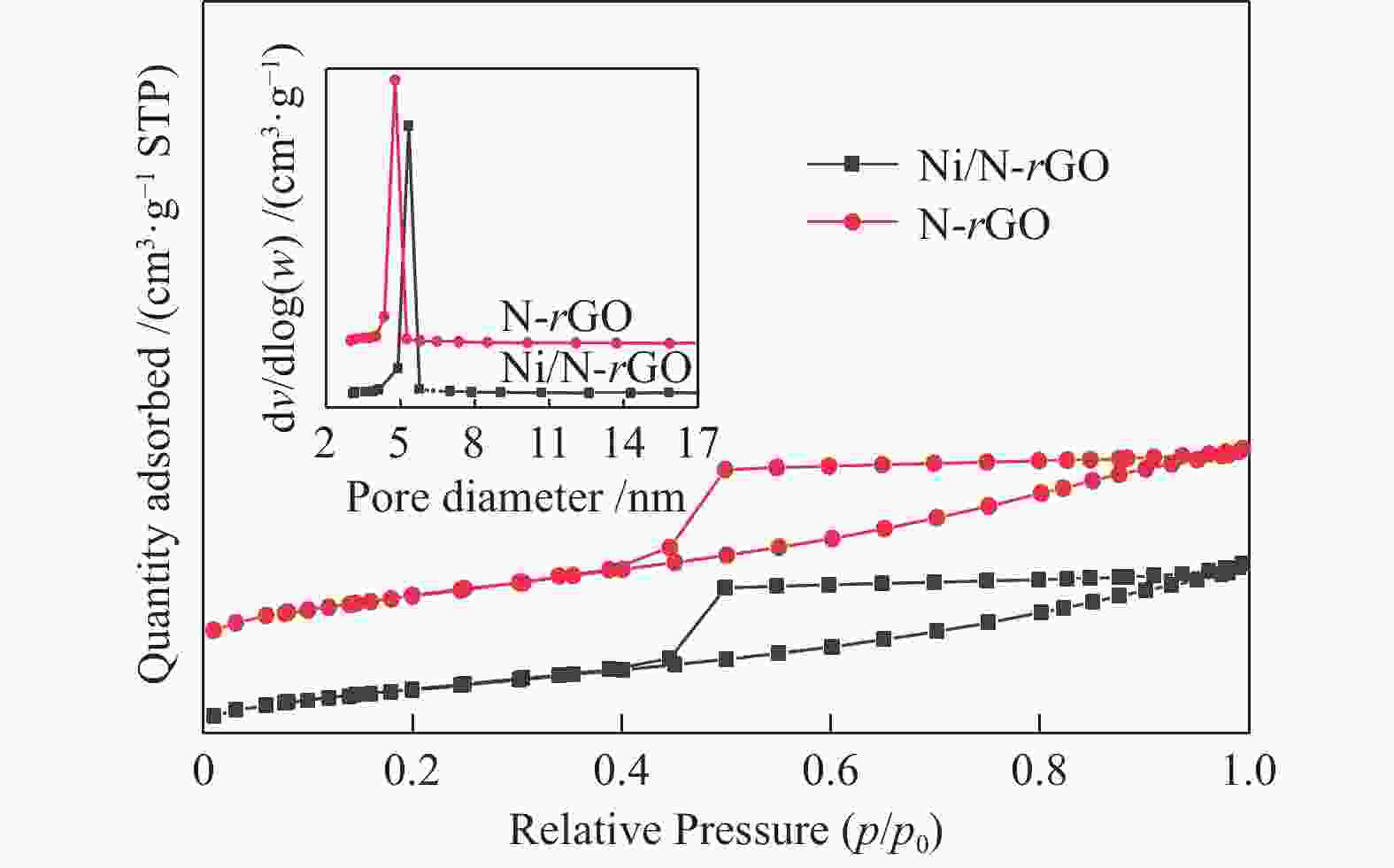
表 1 各催化剂的物理结构性质
Table 1 Physical structural properties of each catalyst
Sample Specific surface
area/(m2·g−1)Average pore size/nm Pore volume
/(cm3·g−1)GO 70 2.0 0.03 N-rGO 352 4.8 0.42 rGO 315 4.5 0.40 AC 483 1.9 0.33 Ni/N-rGO 273 5.4 0.37 Ni/rGO 215 5.1 0.35 Ni/AC 348 2.1 0.27 表 2 不同镍基催化剂的活性和选择性
Table 2 Activity and selectivity of different nickel-based catalysts
Sample Phenol conversion rate/% Cyclohexanone selectivity/% rGO 48.1 30.6 N(30)-rGO 59.7 39.9 10%Ni/N(30)-rGO 81.2 47.3 20%Ni/N(30)-rGO 91.9 61.8 30%Ni/N(30)-rGO 86.2 55.2 20%Ni/rGO 82.7 45.4 20%Ni/AC 67.2 32.6 -
[1] KONG X Q, GONG Y T, MAO S J, WANG Y. Selective hydrogenation of phenol[J]. Chem Nano Mater,2018,4:1−20. doi: 10.1002/cnma.201700346 [2] ALIAHMADI M, DAVOUDI M, KHARAT A N. Selective hydrogenation of phenol to cyclohexanone catalyzed by palladium nanoparticles supported on alumina/lanthanide oxides[J]. React Kinet Mech Catal,2020,11:1−10. [3] 温昕, 董洁, 舍添添, 白国义. 苯酚加氢研究进展[J]. 河北大学学报(自然科学版),2018,38(3):239−247.WEN Xin, DONG Jie, SHE Tian-tian, BAI Guo-yi. Progress in phenol hydrogenation[J]. J Hebei Univ (Nat Sci Ed),2018,38(3):239−247. [4] CHEN Y Z, KONG X Q, MAO S J, WANG Z, GONG Y T, WANG Y. Study of the role of alkaline sodium additive in selective hydrogenation of phenol[J]. Chin J Catal,2019,40(10):1516−1524. doi: 10.1016/S1872-2067(19)63386-3 [5] RAUT A N, NANDANWAR S U, SURYAWANSHI Y R, CHAKRABORTY M, JAUHARI S, MUKHOPADHYAY S, SHENOY K T, BAJAJ H C. Liquid phase selective hydrogenation of phenol to cyclohexanone over Ru/Al2O3 nanocatalyst under mild conditions[J]. Kinet Catal,2016,57(1):39−46. doi: 10.1134/S0023158416010110 [6] TELES C A, RABELO-NETO R C, LIMA J R D, MATTOS L V, RESASCO D E, F B. The effect of metal type on hydrodeoxygenation of phenol over silica supported catalysts[J]. Catal Lett,2016,146(10):1−10. [7] 徐海升, 黄国强, 何丽娟. 酚类化合物加氢脱氧催化剂的研究进展[J]. 现代化工,2020,40(10):66−70.XU Hai-sheng, HUANG Guo-qiang, HE Li-juan. Advances in research of catalysts for hydrodeoxygenation of phenolic compounds[J]. Mod Chem Ind,2020,40(10):66−70. [8] 彭鹏, 刘洪涛, 武斌, 汤庆鑫, 刘云圻. 氮掺杂石墨烯的p型场效应及其精细调控(英文)[J]. 物理化学学报,2019,35(11):1282−1290. doi: 10.3866/PKU.WHXB201903002PENG Peng, LIU Hong-tao, WU Bin, TANG Qing-xin, LIU Yun-qi. Nitrogen doped graphene with a p-type field-effect and its fine modulation[J]. Acta Phys-Chim Sin,2019,35(11):1282−1290. doi: 10.3866/PKU.WHXB201903002 [9] 徐海升, 王 豪, 何丽娟. Ni2P/rGO-Al2O3催化剂的表征及加氢脱氧性能[J]. 石油学报(石油加工),2019,35(1):35−39.XU Hai-sheng, WANG Hao, HE Li-juan. Characterization of Ni2P/rGO-Al2O3 catalyst and its catalytic performance in hydrodeoxygenation[J]. Acta Pet Sin (Pet Process Sect),2019,35(1):35−39. [10] EMTSEV K V, BOSTWICK A, HORN K, JOBST J, KELLOGG G L, LEY L, MCCHESNEY J L, OHTA T, RESHANOV S A. Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide[J]. Nat Mater,2009,8(3):203−207. doi: 10.1038/nmat2382 [11] WANG Y, RONG X M, WANG T, WU S, QU J. Inflfluence of graphene surface chemistry on Ir-catalyzed hydrogenation of p-chloronitrobenzene and cinnamaldehyde: Weak molecule-support interactions[J]. J Catal,2019,377:524−533. doi: 10.1016/j.jcat.2019.07.061 [12] 潘若飞. 石墨烯基催化剂在芳环选择性加氢中的应用研究[D]. 杭州: 浙江大学, 2016.PAN Ruo-fei. Application of graphene-based catalysts in selective hydrogenation of aromatic compounds[D]. Hangzhou: Zhejiang University, 2016. [13] 李孟辉, 袁鸣蔚, 黄佳, 吴鸿杰, 李进军, 游志雄. 石墨烯及其衍生物在催化领域的应用[J]. 分子催化,2019,33(2):190−200.LI Meng-hui, YUAN Ming-wei, HUANG Jia, WU Hong-jie, LI Jin-jun, YOU Zhi-xiong. Application of graphene and its derivatives in catalysis[J]. J Mol Catal,2019,33(2):190−200. [14] ZHANG C H, YANG S Z, WU J J, LIU M J. Electrochemical CO2 reduction with atomic iron-dispersed on nitrogen-doped graphene[J]. Adv Energy Mater,2018,8(19):1703487−1903496. doi: 10.1002/aenm.201703487 [15] HUMMERS W S, OFFEMAN R E. Preparation of graphitic oxide[J]. J Am Chem Soc,1958,80(6):1339. doi: 10.1021/ja01539a017 [16] 苏鹏, 郭慧林, 彭三, 宁生科. 氮掺杂石墨烯的制备及其超级电容性能[J]. 物理化学学报,2012,28(11):2745−2753. doi: 10.3866/PKU.WHXB201208221SU Peng, GUO Hui-lin, PENG San, NING Sheng-ke. Preparation of nitrogen-doped graphene and its supercapacitive properties[J]. Acta Phys-Chim Sin,2012,28(11):2745−2753. doi: 10.3866/PKU.WHXB201208221 [17] 胡克文, 李贺军, 齐乐华, 罗俊, 连洪程. 氮掺杂石墨烯墨水的制备及理化特性(英文)[J]. 新型炭材料,2020,35(4):444−451. doi: 10.1016/S1872-5805(20)60502-9HU Ke-wen, LI He-jun, QI Le-hua, LUO Jun, LIAN Hong-hua. Preparation and physicochemical properties of nitrogen-doped graphene inks[J]. New Carbon Mater,2020,35(4):444−451. doi: 10.1016/S1872-5805(20)60502-9 [18] YANG J, WU M, CHEN F, FEI Z D, ZHONG M Q. Preparation, characterization, and supercritical carbon dioxide foaming of polystyrene/graphene oxide composites[J]. J Supercrit Fluid,2011,56(2):201−207. doi: 10.1016/j.supflu.2010.12.014 [19] GAIDUKEVIC J, BARKAUSKAS J, MALAIKA A, PAULINA R G, ALEKSANDRA M, VITALIJA J. Modified graphene-based materials as effective catalysts for transesterification of rapeseed oil to biodiesel fuel[J]. Chin J Catal,2018,39(10):1633−1645. doi: 10.1016/S1872-2067(18)63087-6 [20] LE M C, VAN K L, NGUYEN T H T, HA N N. The impact of Ce-Zr addition on nickel dispersion and catalytic behavior for CO2 methanation of Ni/AC catalyst at low temperature[J]. J Chem,2017,1−11. [21] MICHAL K, MIETEK J. Gas adsorption characterization of ordered organic-inorganic nanocomposite materials[J]. Chem Mater,2001,13:3169−3183. doi: 10.1021/cm0101069 [22] 张荣斌, 仝塞, 杨金美, 唐纤秾, 黄传庆, 王学文, 冯 刚, 蔡建信. 石墨烯负载镍催化CO2加氢甲烷化[J]. 高等学校化学学报,2017,38(12):2255−2261. doi: 10.7503/cjcu20170348ZHANG Rong-bin, TONG Sai, YANG Jin-mei, TANG Xian-nong, HUANG Chuan-qing, WANG Xue-wen, FENG Gang, CAI Jian-xin. Graphene supported nickel catalyst for methanation of carbon dioxide[J]. Chem J Chin Univ,2017,38(12):2255−2261. doi: 10.7503/cjcu20170348 [23] RIDZUAN N D M, SHAHARUN M S, LEE K M, DIN I U, PUSPITASARI P. Influence of nickel loading on reduced graphene oxide-based nickel catalysts for the hydrogenation of carbon dioxide to methane[J]. Catalysts,2020,10(5):471−486. doi: 10.3390/catal10050471 [24] 徐海升, 何丽娟, 黄国强. Ni2P/Fe-HAP催化剂的表征及其苯酚加氢表面作用机理[J]. 精细化工,2020,37(6):1227−1232.XU Hai-sheng, HE Li-juan, HUANG Guo-qiang. Characterization of Ni2P/Fe-HAP catalyst and its surface interaction mechanism of phenol hydrogenation[J]. Fine Chem,2020,37(6):1227−1232. [25] CHEN J Z, ZHANG W, CHEN L M, MA L L. Direct selective hydrogenation of phenol and derivatives over polyaniline-functionalized carbon-nanotube-supported palladium[J]. Chem Plus Chem,2013,78(2):142−148. doi: 10.1002/cplu.201200276 [26] WANG Y, YAO J, LI H, SU D S, ANTONIETTI M. Highly selective hydrogenation of phenol and derivatives over a Pd@carbon nitride catalyst in aqueous media[J]. J Am Chem Soc,2011,133(8):2362−2365. doi: 10.1021/ja109856y -




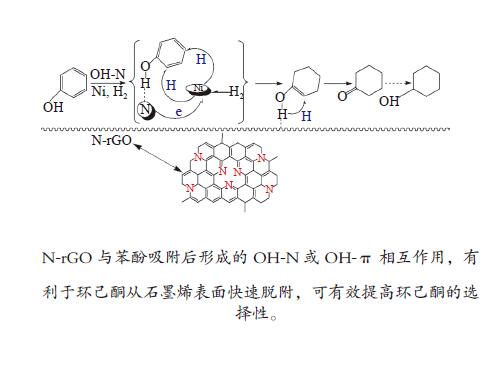
 下载:
下载: