Study on the transformation characteristic of heteroatoms during liquefaction of Naomaohu coal
-
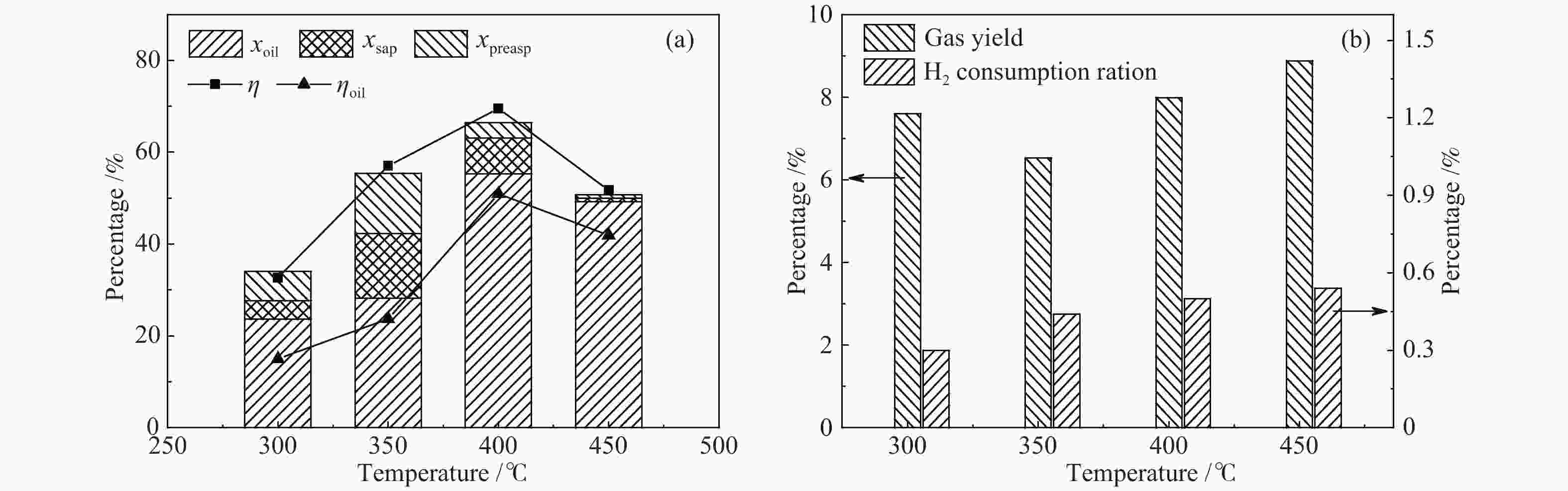
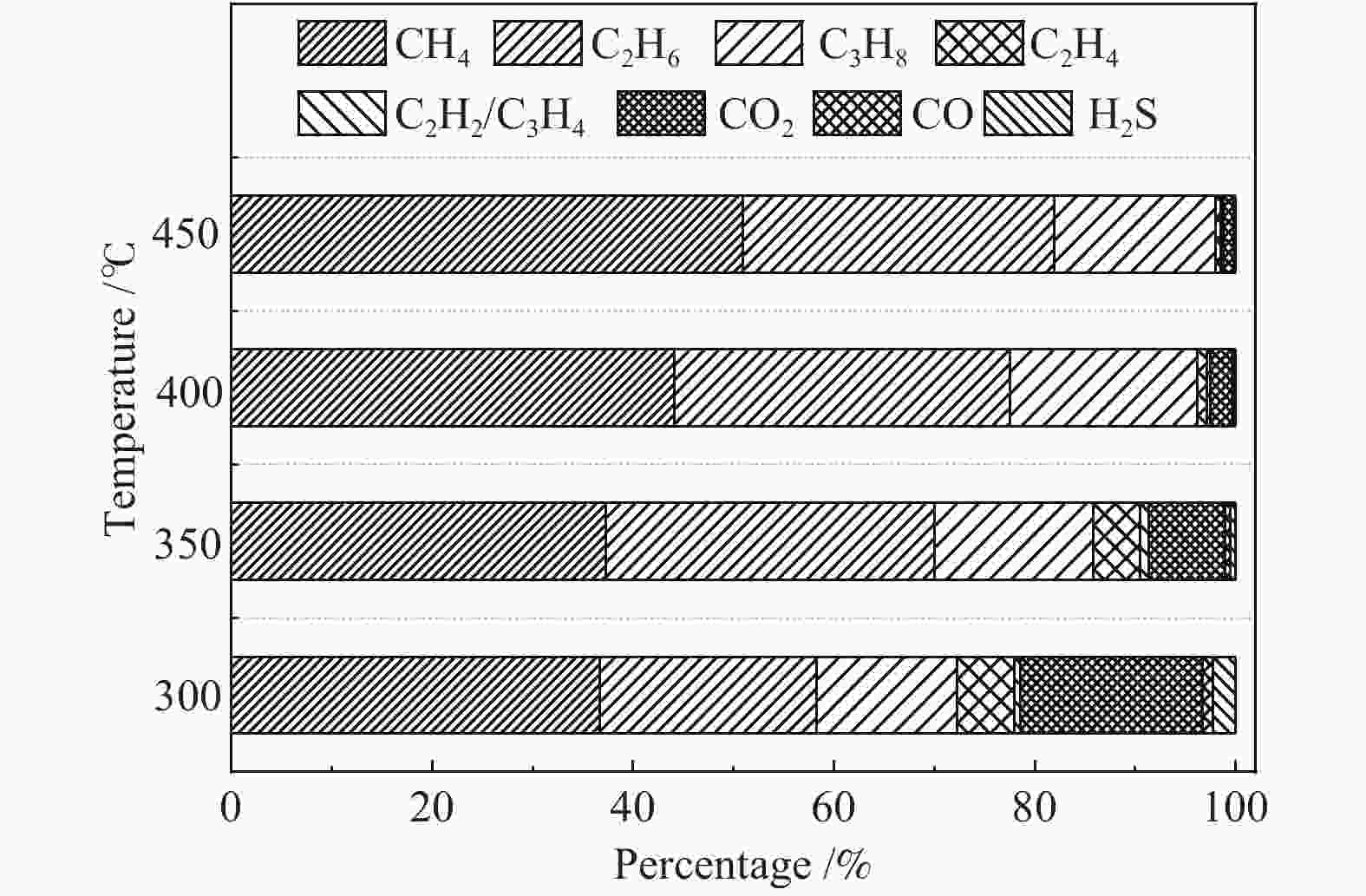
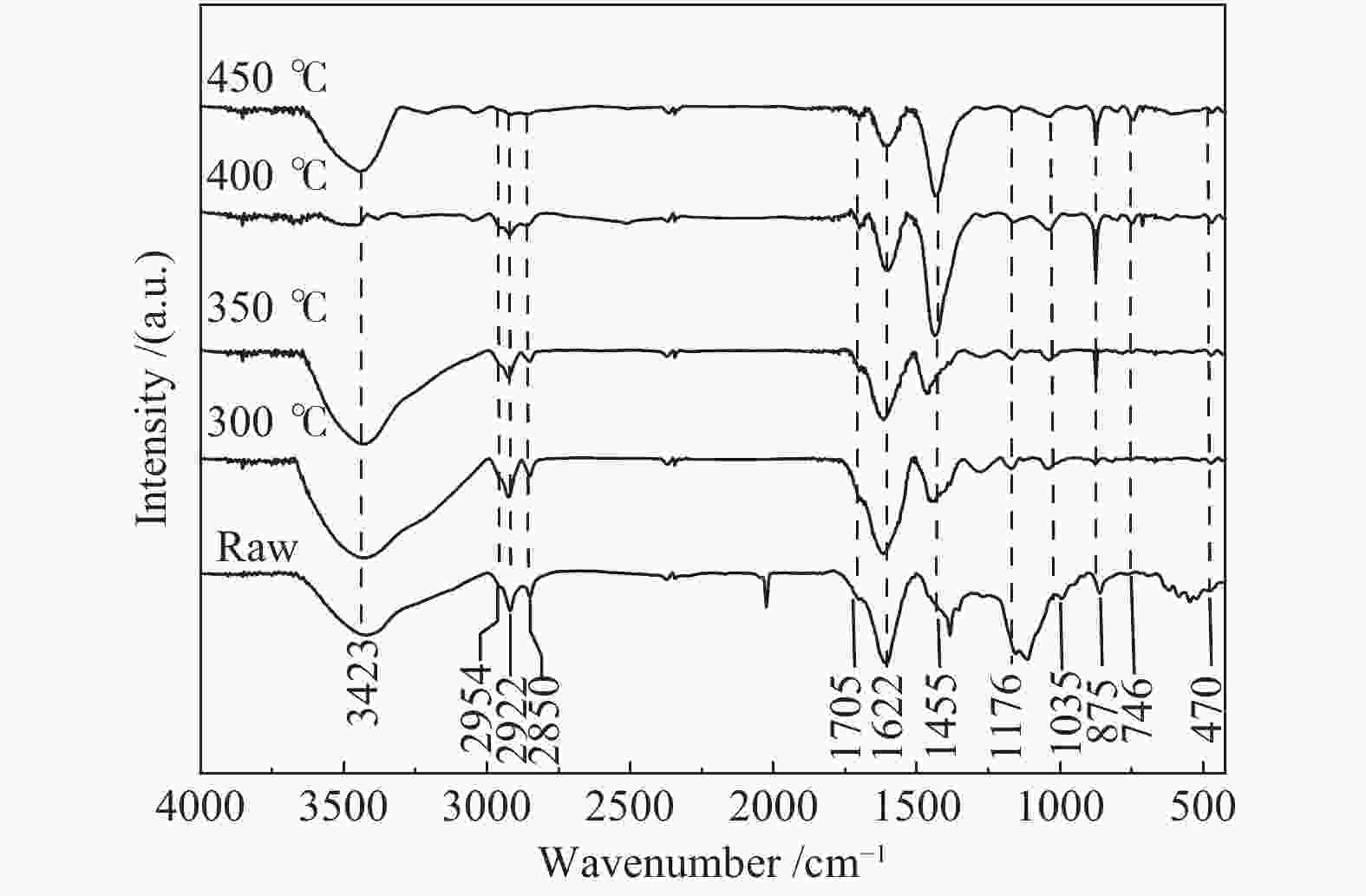
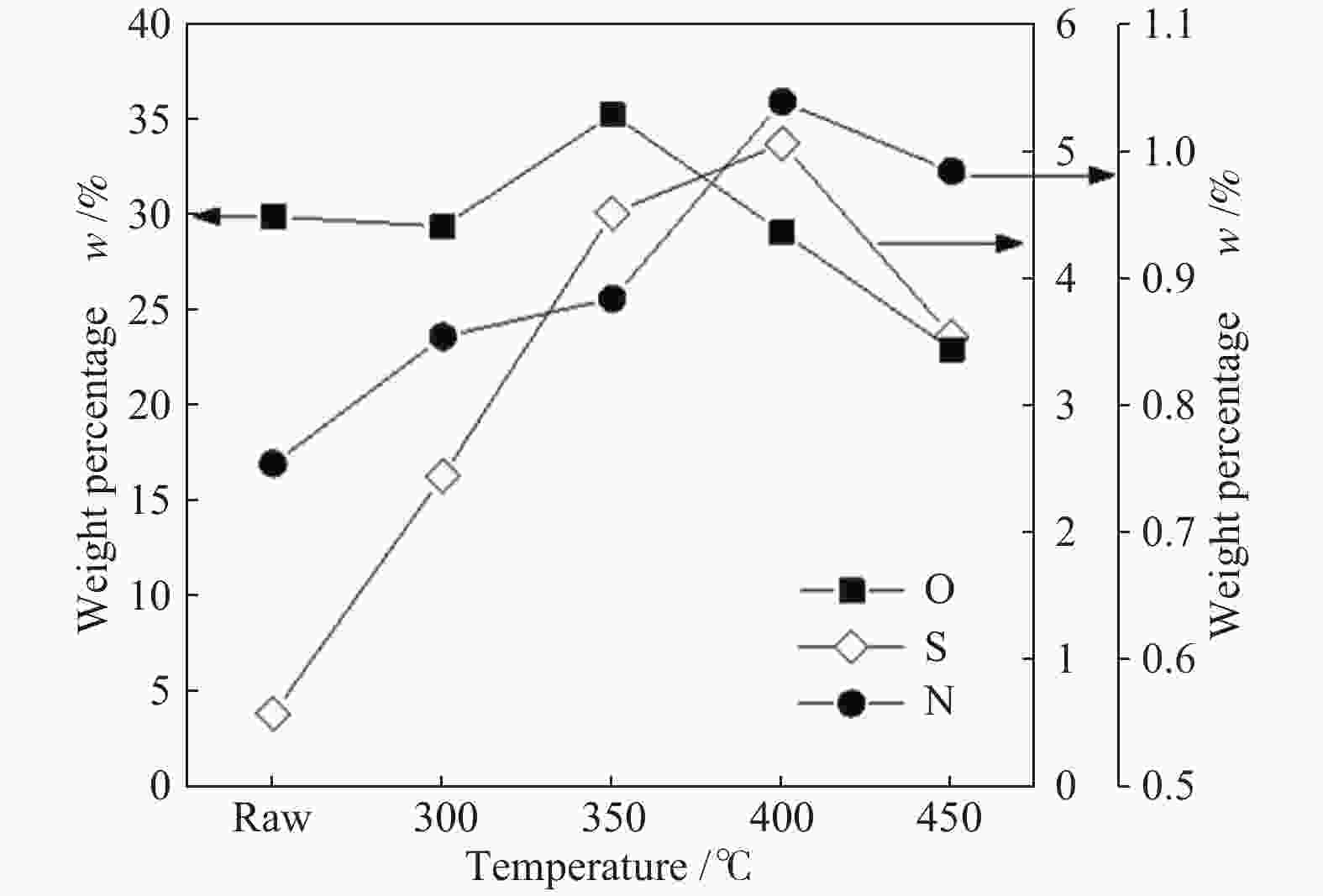
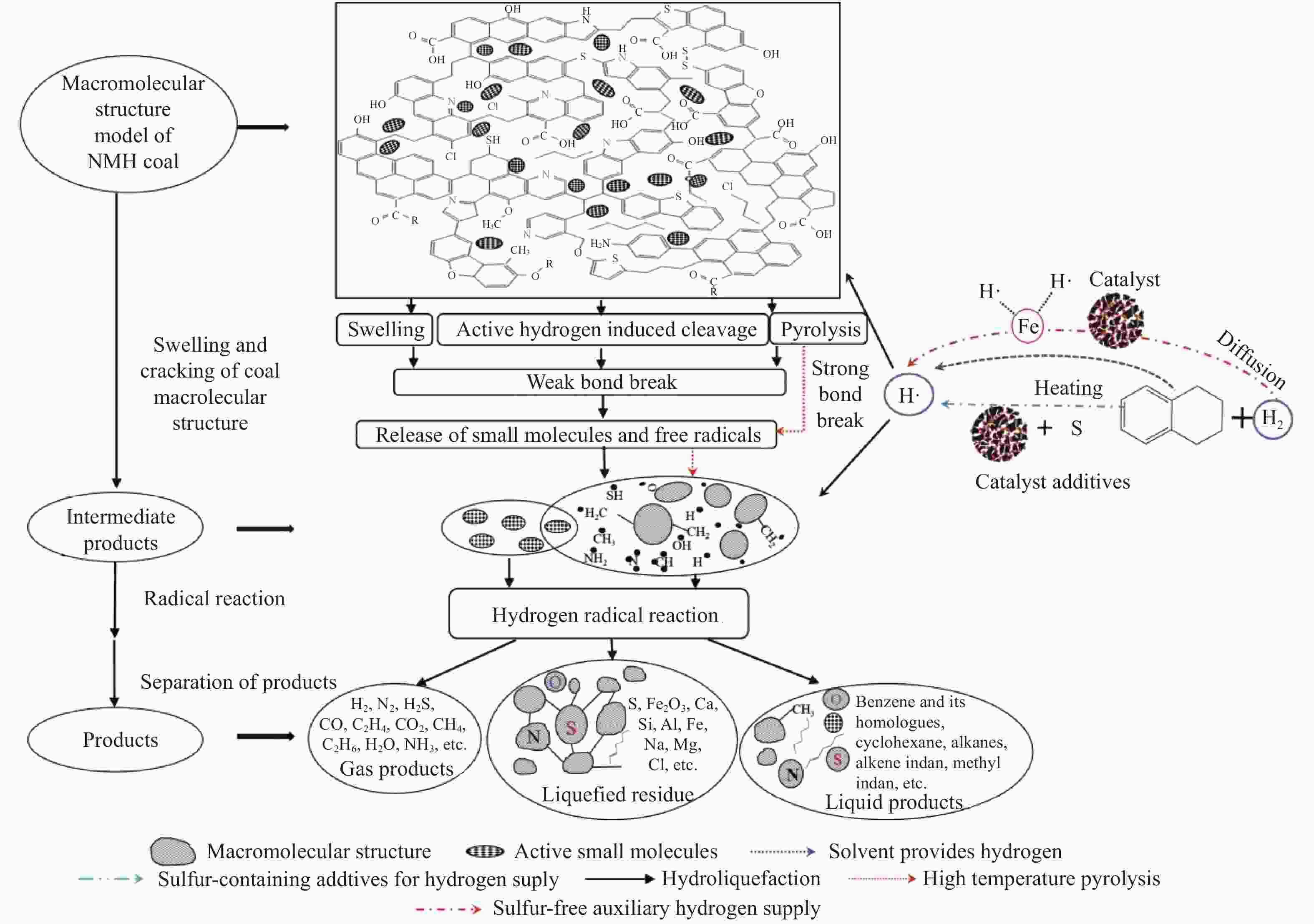
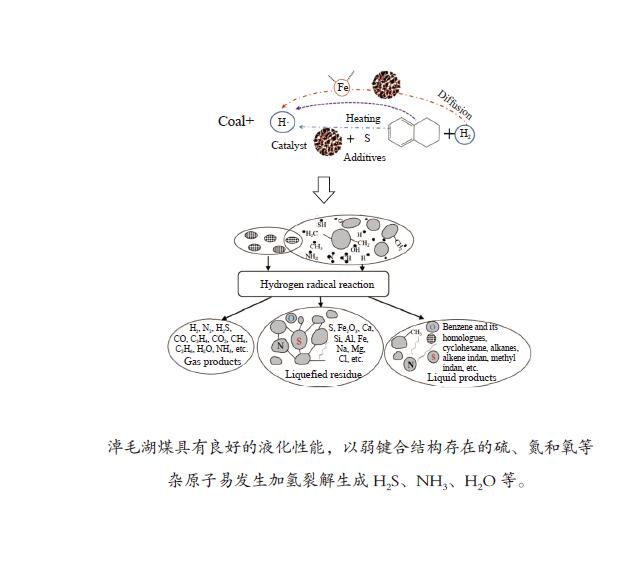
摘要: 以淖毛湖煤为原料,进行加氢直接液化,考察了加氢温度与转化率和油收率的关系,并解析了加氢条件下煤中硫、氮和氧的迁移转化特性。结果表明,淖毛湖煤具有良好的液化性能,400 ℃和2 MPa氢初压条件下即可达到69.6%的转化率和55.3%的油产率。结合气相色谱质谱联用(GC-MS)和气相色谱-原子发射光谱 (GC-AED)等方法对产物分析发现,以弱键合结构存在的硫、氮和氧等杂原子易发生加氢裂解生成H2S、NH3、H2O等。液化油品中含硫化合物主要以噻吩及噻吩同系物为主;含氮化合物含量极低,主要由含氮杂环化合物构成;含氧化合物在液化油中主要以酚及酚的同系物为主。存在于芳香结构中的杂原子会随着自由基缩合反应,生成更稳定的含杂原子稠环化合物富集在液化残渣中。Abstract: In this work, Naomaohu coal was used for direct liquefaction. The conversion and oil yield as a function of hydrogenating temperature were investigated, and the transformation characteristics of heteroatoms in raw coal and liquefaction products were clarified. The results showed that Naomaohu coal presented high liquefaction performance even under lower initial pressure of 2 MPa. The conversion and oil yield could reach 69.6% and 55.3%, respectively, at a pressure of 2 MPa and 400 ℃. Gas chromatography-mass spectrometry and gas chromatography-atomic emission spectrometry were combined to analyze the products. It was found that the weak bond structure containing heteroatoms was easier hydrogenated to fracture and removed in the form of H2S, NH3, H2O, etc. The sulfur-containing compounds in liquefied products were mainly thiophene and thiophene homologues. The content of nitrogen-containing compounds was trace and mainly consisted of nitrogen heterocyclic compounds. The oxygen-containing compounds in liquefied oil were dominantly phenols and their homologues. The heteroatoms in the aromatic structure would generate more stable heteroatom-containing compounds in the liquefaction residue via the free radical polycondensation reaction.
-
Key words:
- Naomaohu coal /
- direct liquefaction /
- heteroatoms /
- transformation characteristics
-
表 1 淖毛湖煤的煤质分析
Table 1 Proximate and ultimate analyses of Naomaohu coal
Proximate analysis w/% Ultimate analysis wdaf/% Mad Vdaf Ad FCdaf C H N St O* 7.02 52.41 5.41 47.59 77.03 6.01 1.01 0.39 15.56 note:ad: air dried base;d: dry base;daf: dry and ash-free base;*: by different 表 2 液化产物的组成
Table 2 Component distribution of liquefied oil
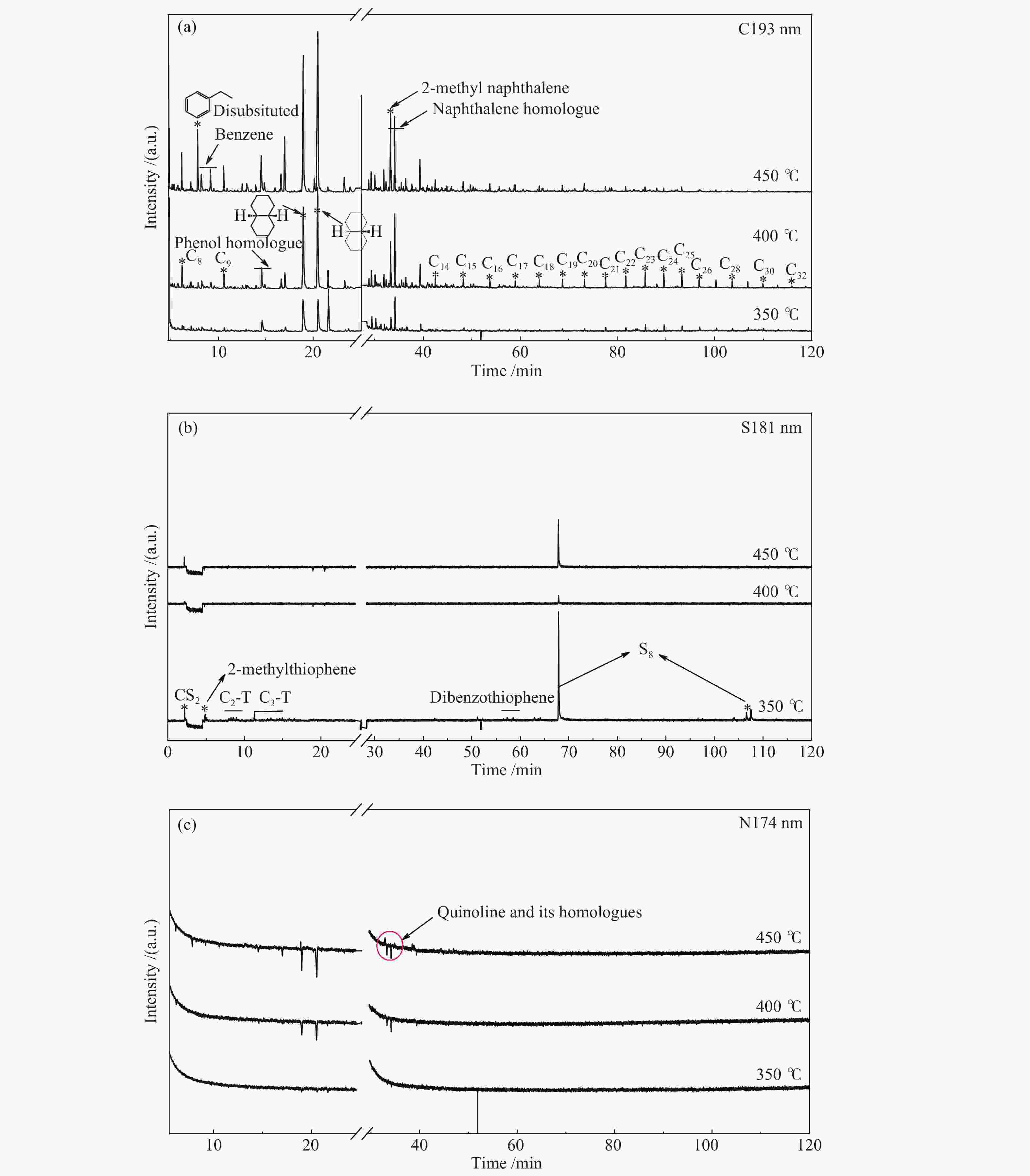
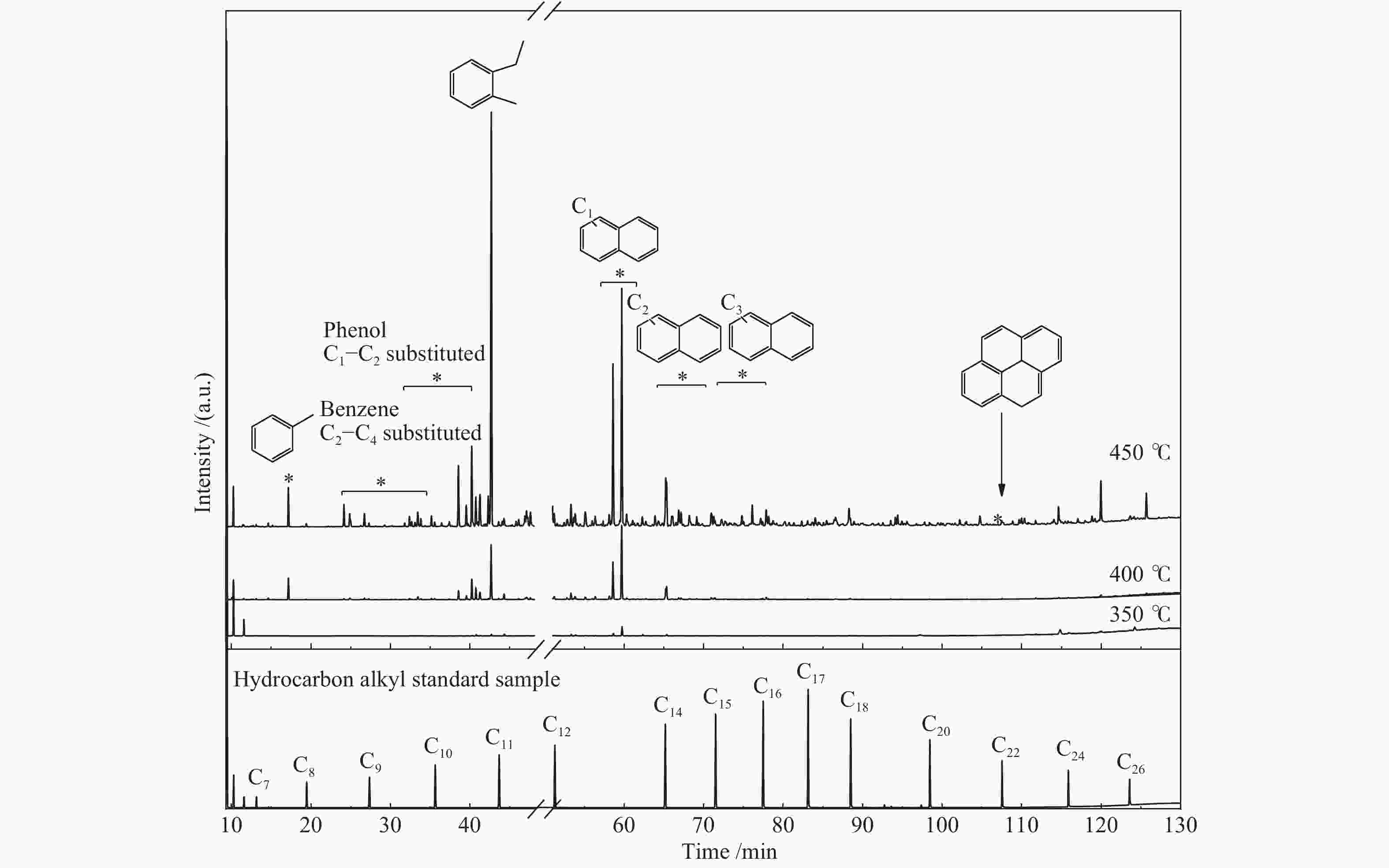
Items Compounds Content /% 350 ℃ 400 ℃ 450 ℃ Chain hydrocarbon alkane(C7−C32 alkane, olefins) 1.91 1.29 2.69 tetrahydronaphthalene 27.31 29.09 18.32 Aromatic hydrocarbons and their homologues benzene and its homologues 0.15 0.85 2.68 naphthalene 16.15 46.41 45.95 C1−C3 substituted naphthalene 0.60 4.64 7.74 indene and its homologues 0.10 1.85 5.67 diphenyl and its homologues 0.00 0.20 0.42 anthracene and phenanthrene homologues 0.01 0.07 0.81 pyrene 0.00 0.22 2.24 anthracene and pyrene homologues 0.01 2.05 1.98 total 17.02 59.29 67.49 S-containing compound quinoline and others 0.10 0.12 0.20 N-containing compound thiophene and its homologues 0.21 0.12 0.23 benzothiophene and its homologues 0.24 0.23 0.15 total 0.45 0.35 0.38 O-containing compound phenol 0.00 0.10 0.16 C1−C4 substituted phenol 0.05 0.65 1.57 naphthol 0.00 0.01 0.04 others 0.75 0.49 0.20 total 0.80 1.25 1.97 -
[1] 方正美, 吕海燕, 张媛媛, 宁奕飞, 潘铁英, 张德祥. 溶剂特性对淖毛湖煤加氢液化中间产物反应行为的影响[J]. 燃料化学学报,2019,47(8):907−914. doi: 10.3969/j.issn.0253-2409.2019.08.002FANG Zheng-mei, LÜ Hai-yan, ZHANG Yuan-yuan, NING Yi-fei, PAN Tie-ying, ZHANG De-xiang. Effect of solvent characteristics on reaction behavior of hydroliquefaction intermediate products from Naomaohu coal[J]. J Fuel Chem Technol,2019,47(8):907−914. doi: 10.3969/j.issn.0253-2409.2019.08.002 [2] MOCHIDA I, OSAMO O, YOON S H. Chemicals from direct coal liquefaction[J]. Chem Rev,2013,114(3):1637−1672. [3] HOU R R, BAI Z Q, ZHENG H Y, FENG Z H, YE D H, GUO Z X, KONG L X, BAI J, LI W. Behaviors of hydrogen bonds formed by lignite and aromatic solvents in direct coal liquefaction: Combination analysis of density functional theory and experimental methods[J]. Fuel,2020,265:117011. doi: 10.1016/j.fuel.2020.117011 [4] HAO P, BAI Z Q, HOU R R, XU J L, BAI J, GUO Z X, KONG L X, LI W. Effect of solvent and atmosphere on product distribution, hydrogen consumption and coal structural change during preheating stage in direct coal liquefaction[J]. Fuel,2018,211:783−788. doi: 10.1016/j.fuel.2017.09.122 [5] ALI A, ZHAO C. Direct liquefaction techniques on lignite coal: A review[J]. Chin J Catal,2020,41(3):375−389. doi: 10.1016/S1872-2067(19)63492-3 [6] LIN X C, YANG S S, CHEN X J, ZHENG P P, WANG Y G, ZHANG S. Effects of calcium on the evolution of nitrogen during pyrolysis of a typical low rank coal[J]. Int J Coal Sci Technol,2020,7:397−404. doi: 10.1007/s40789-019-00290-3 [7] KULAKOVA V, BUTUZOVA L, ANDRADE J M, SHEVKOPLYAS V, TURCHANINA O. Characterization of sulfur coal-derived liquids as a source of hydrocarbons to produce chemicals and synthetic fuels[J]. Fuel,2016,184:314−324. doi: 10.1016/j.fuel.2016.07.005 [8] WANG Z C, GE Y, SHUI H F, REN S B, PAN C X, KANG S G, LEI Z P, ZHAO Z J, HU J C. Molecular structure and size of asphaltene and preasphaltene from direct coal liquefaction[J]. Fuel Process Technol,2015,137:305−311. doi: 10.1016/j.fuproc.2015.03.015 [9] FENG J, XUE X Y, LI X H, LI W Y, GUO X F, LIU K. Products analysis of Shendong long-flame coal hydropyrolysis with iron-based catalysts[J]. Fuel Process Technol,2015,130:96−100. doi: 10.1016/j.fuproc.2014.09.035 [10] XU B, LU W Y, SUN Z, HE T, GORONCY A, ZHANG Y L, FAN M H. High-quality oil and gas from pyrolysis of Powder River Basin coal catalyzed by an environmentally-friendly, inexpensive composite iron-sodium catalysts[J]. Fuel Process Technol,2017,167:334−344. doi: 10.1016/j.fuproc.2017.05.028 [11] LI X, HU S X, JIN L J, HU H Q. Role of iron-based catalyst and hydrogen transfer in direct coal liquefaction[J]. Energy Fuels,2008,22(2):1126−1129. doi: 10.1021/ef7006062 [12] MARKO R D, NENAD D R, NATALIA O, GUY B M, KEVIN M V G. Quantitative on-line analysis of sulfur compounds in complex hydrocarbon matrices[J]. J Chromatogr A,2017,1509:102−113. doi: 10.1016/j.chroma.2017.06.006 [13] WANG Z C, XUE W T, ZHU J, CHEN E S, PAN C X, KANG S G, LEI Z P, REN S B, SHUI H F. Study on the stability of hydro-liquefaction residue of Shenfu sub-bituminous coal[J]. Fuel,2016,181:711−717. doi: 10.1016/j.fuel.2016.05.042 [14] CHEN Z Z, XIE J, LIU Q Y, WANG H X, GAO S S, SHI L, LIU Z Y. Characterization of direct coal liquefaction catalysts by their sulfidation behavior and tetralin dehydrogenation activity[J]. J Energy Inst,2019,92:1213−1222. doi: 10.1016/j.joei.2018.05.009 [15] KHAN M, AHMAD I, ISHAQ M, SHAKIRULLAH M, JAN M T, REHMAN E U, BAHADER A. Spectral characterization of liquefied products of Pakistani coal[J]. Fuel Process Technol,2003,85(1):63−74. [16] 黄澎, 张晓静, 毛学锋, 李伟林. 神府煤液化油加氢精制过程中硫氮化合物分布的变化[J]. 燃料化学学报,2016,44(1):37−43. doi: 10.3969/j.issn.0253-2409.2016.01.006HUANG Peng, ZHANG Xiao-jing, MAO Xue-feng, LI Wei-lin. Change of sulfur and nitrogen compounds in the direct liquefaction oil from Shenfu coal upon the hydrofining process[J]. J Fuel Chem Technol,2016,44(1):37−43. doi: 10.3969/j.issn.0253-2409.2016.01.006 -




 下载:
下载: