ZnOHF nanorods for efficient electrocatalytic reduction of carbon dioxide to carbon monoxide
-
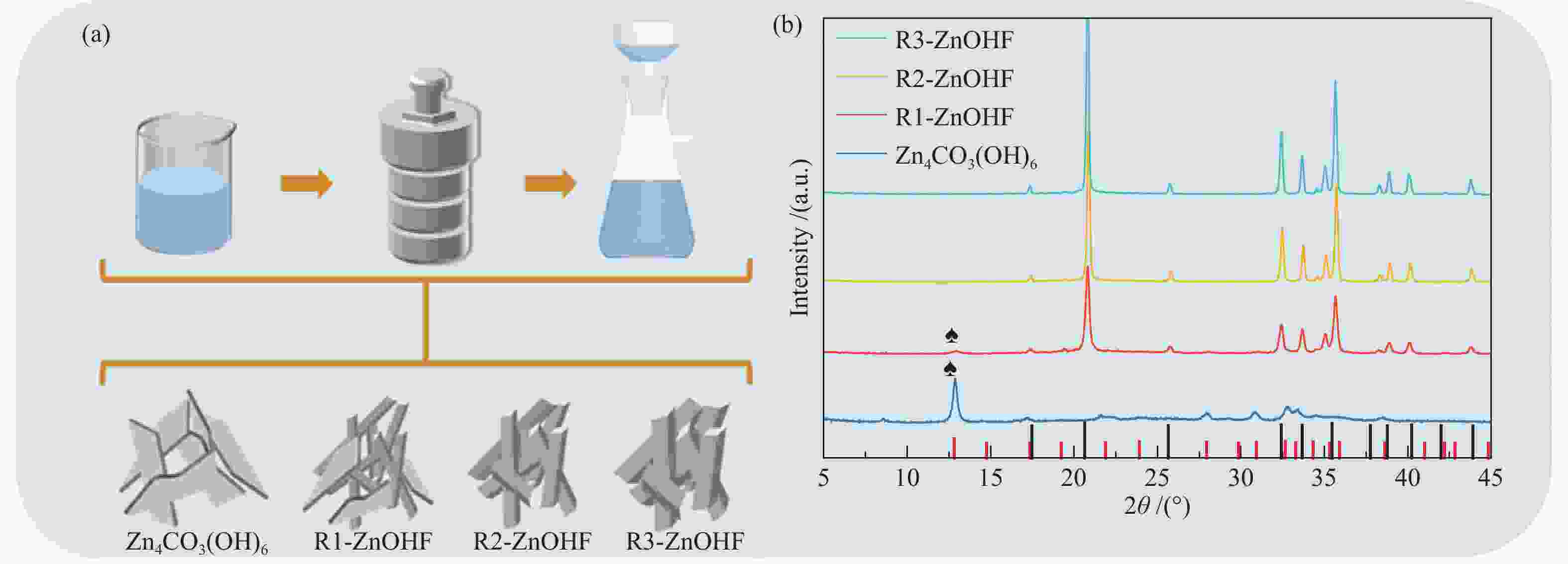
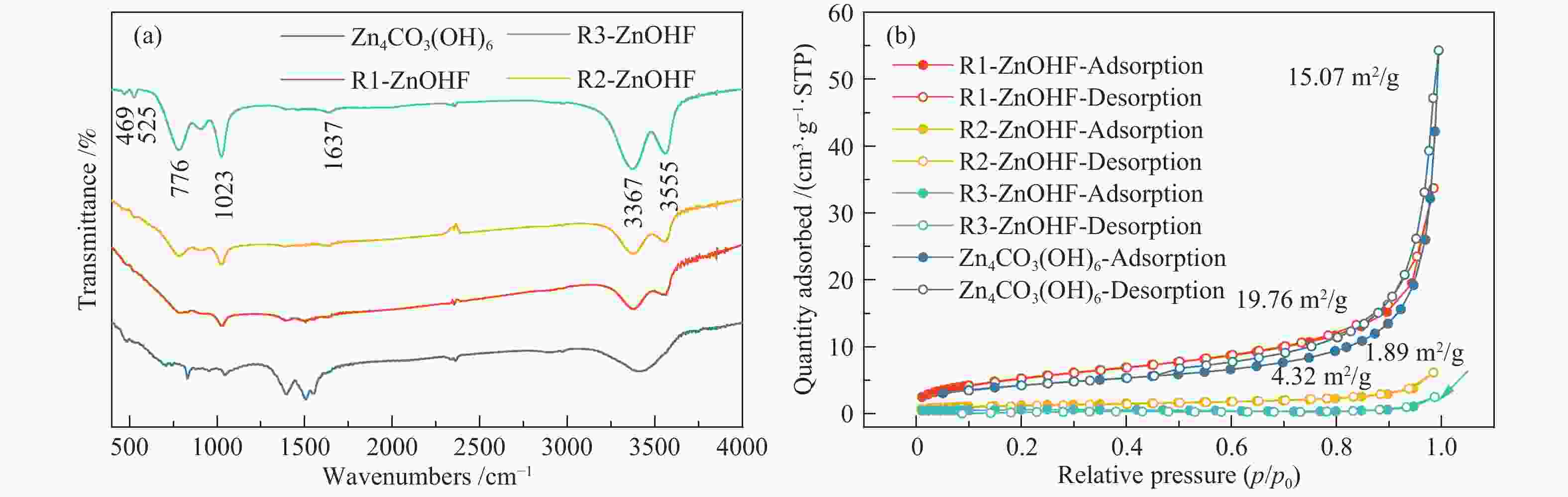
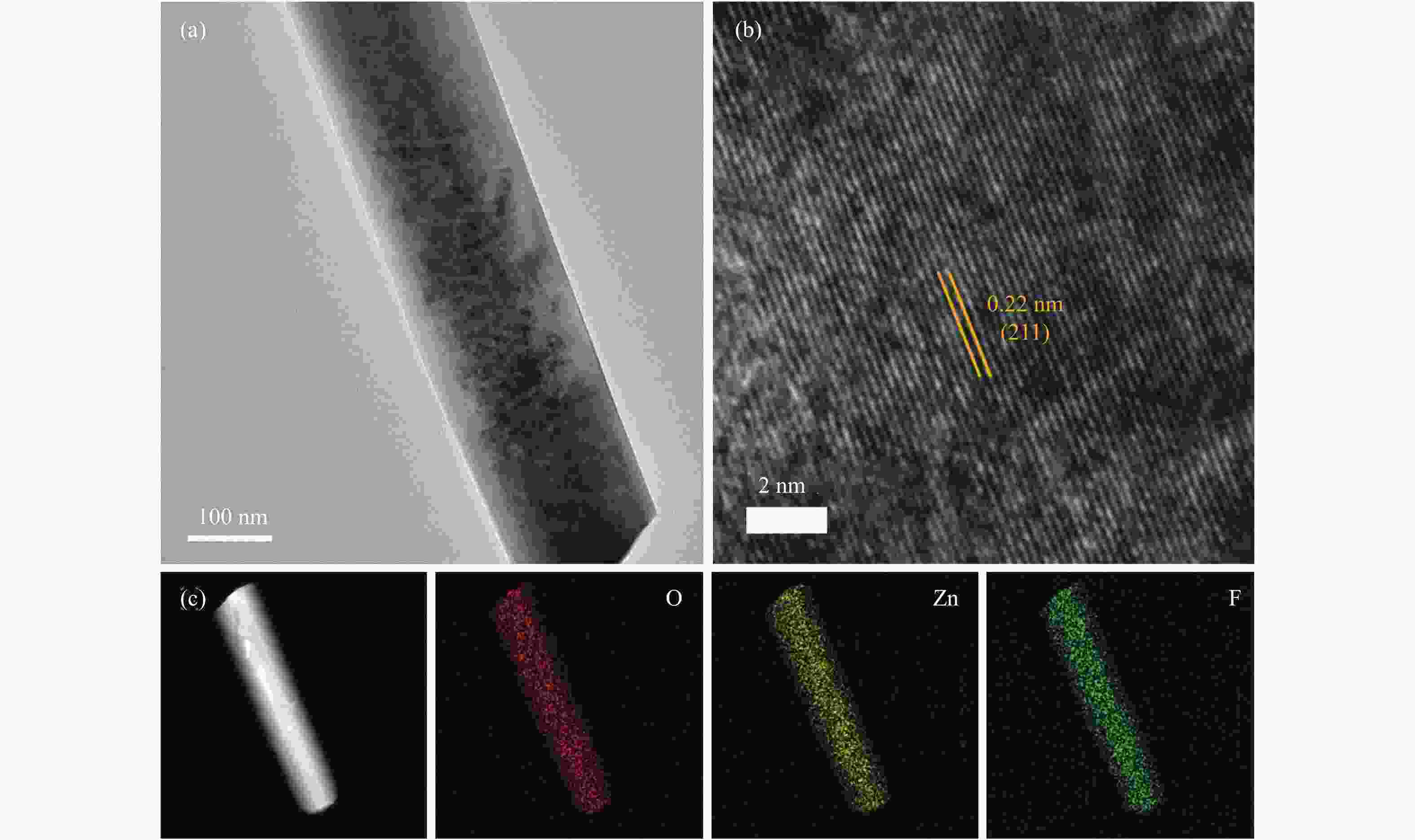
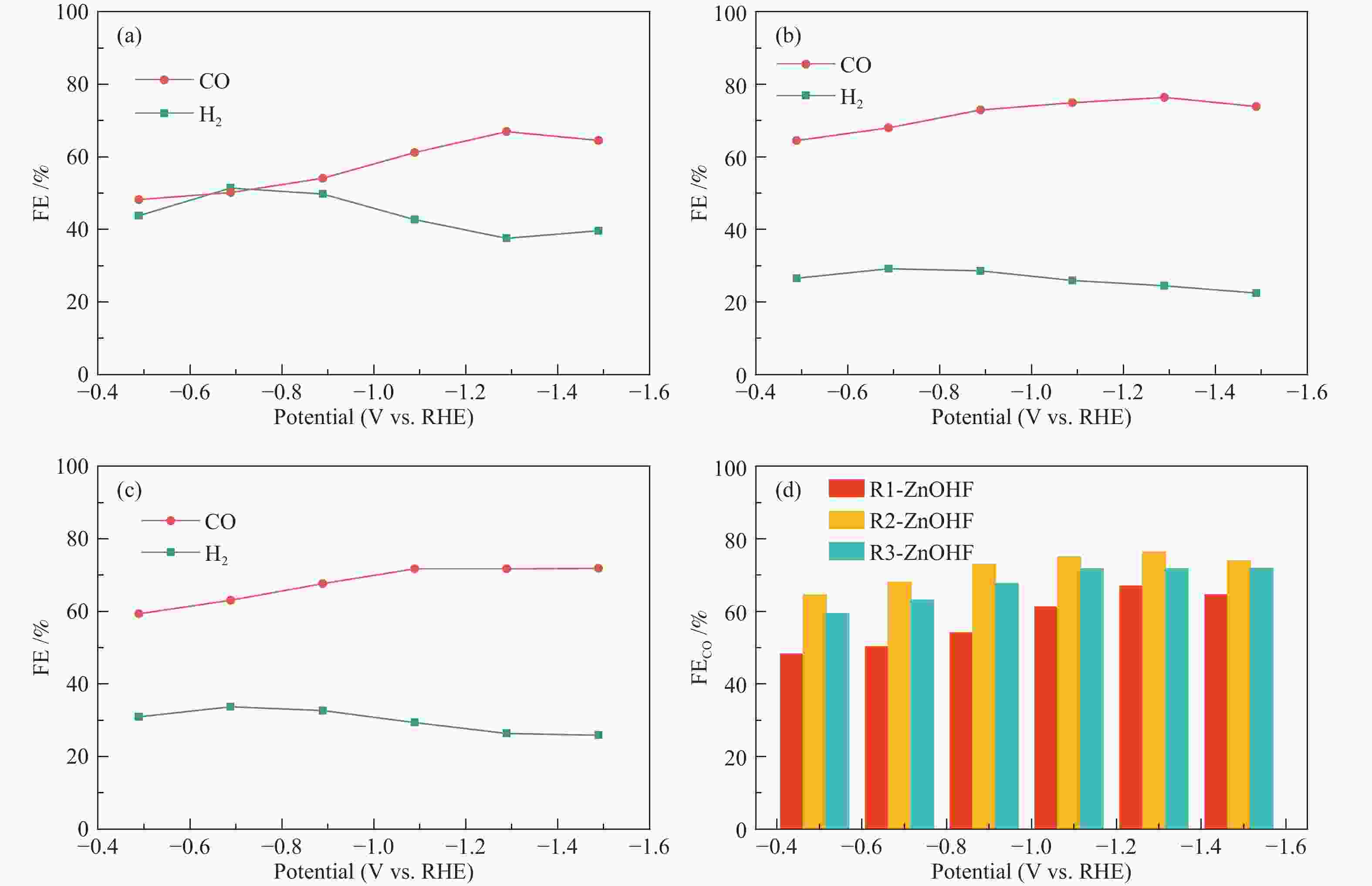
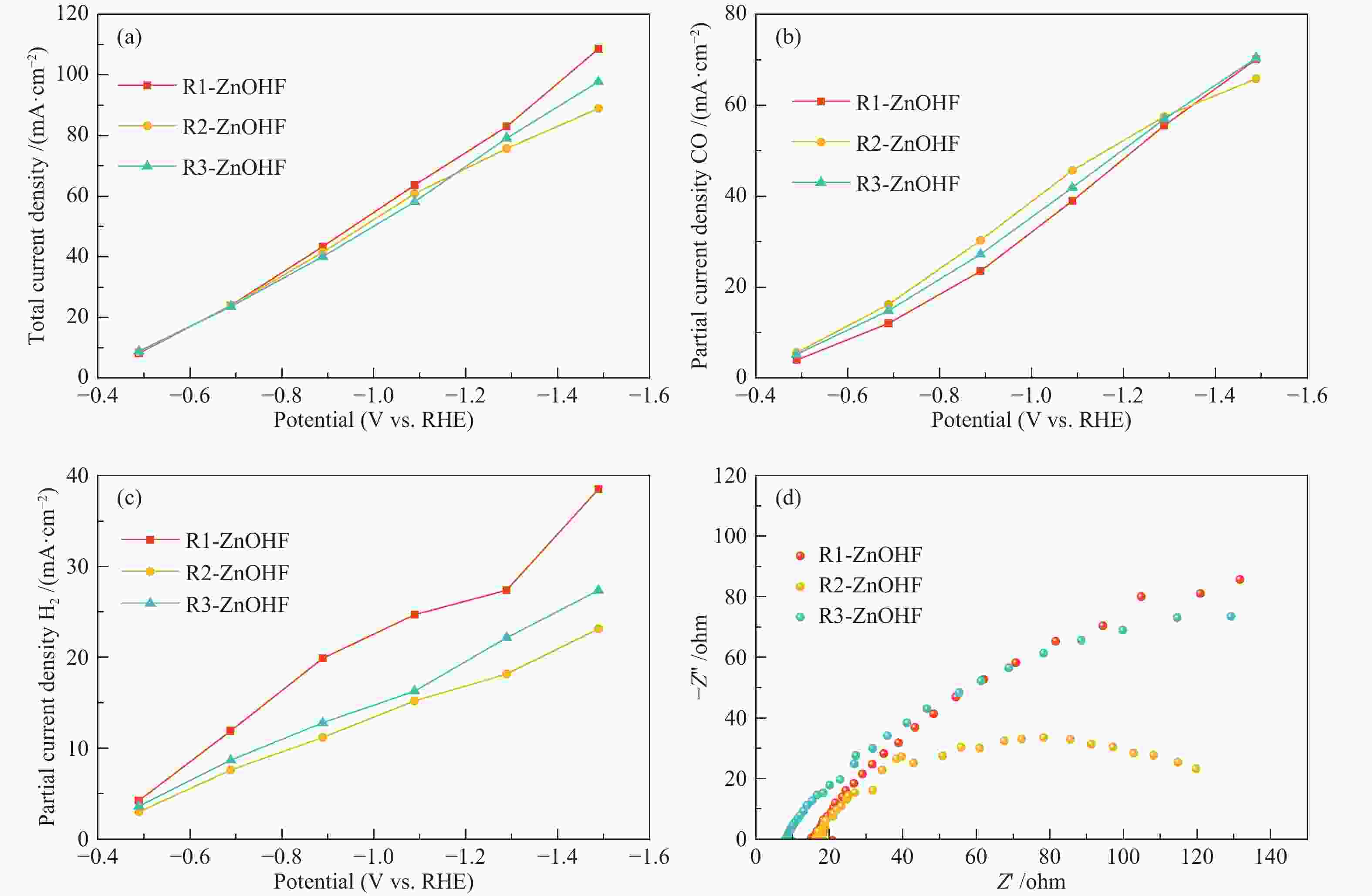
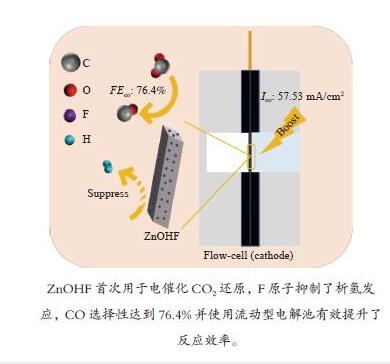
摘要: 通过电催化的方式将二氧化碳资源化利用是缓解或解决目前人类面临的生态危机的理想途径之一。开发廉价高效的催化剂是推动电催化二氧化碳还原走向工业化的关键。一氧化碳是重要的工业原料,利用CO2还原制备CO具有重要的研究意义,但能够将CO2转化为CO的高活性贵金属催化剂难以大规模应用,地球储量丰富的Zn基催化剂是具有潜力的替代者。然而,Zn基催化剂还原活性仍然难以满足现实需求且种类不够丰富。作者将ZnOHF材料引入到电催化CO2还原中,通过简单水热合成法制备了不同尺寸的ZnOHF纳米棒并利用流动型电解池测试其性能。纳米棒较大的比表面积以及材料表面F原子的存在使其具有良好的催化活性,流动型电解池的使用加速了反应传质过程,在−1.28 V (vs. RHE)电势下,R2-ZnOHF纳米棒的CO法拉第效率最高为76.4 %,CO分电流密度为57.53 mA/cm2。Abstract: The utilization of carbon dioxide as a resource through electrocatalysis is one of the ideal ways to alleviate or solve the current ecological crisis mankind facing. The development of inexpensive and efficient catalysts is the key to promoting the industrialization of electrocatalytic carbon dioxide reduction. CO is an important industrial raw material, as a result, CO2 reduction to CO has important research significance. However, high-active noble metal catalysts that can convert CO2 to CO are difficult to apply in large scale. Zn-based catalysts are potential substitutes. However, the reduction activity of Zn-based catalysts still can not meet the actual needs. In this paper, ZnOHF material is employed in the electrocatalytic CO2 reduction for the first time. ZnOHF nanorods of different sizes are prepared through a simple hydrothermal synthesis method and tested in a Flow-Cell. The large specific surface area of the nanorods and the existence of F atoms on the surface of the material lead to good catalytic activity. The Flow-Cell accelerates the reaction mass transfer process. At –1.28 V (vs. RHE), the R2-ZnOHF nanorods have the highest CO Faraday efficiency of 76.4% with the CO current density of 57.53 mA/cm2.
-
Key words:
- ZnOHF /
- carbon dioxide reduction /
- carbon monoxide /
- electrocatalysis /
- flow electrolytic cell
-
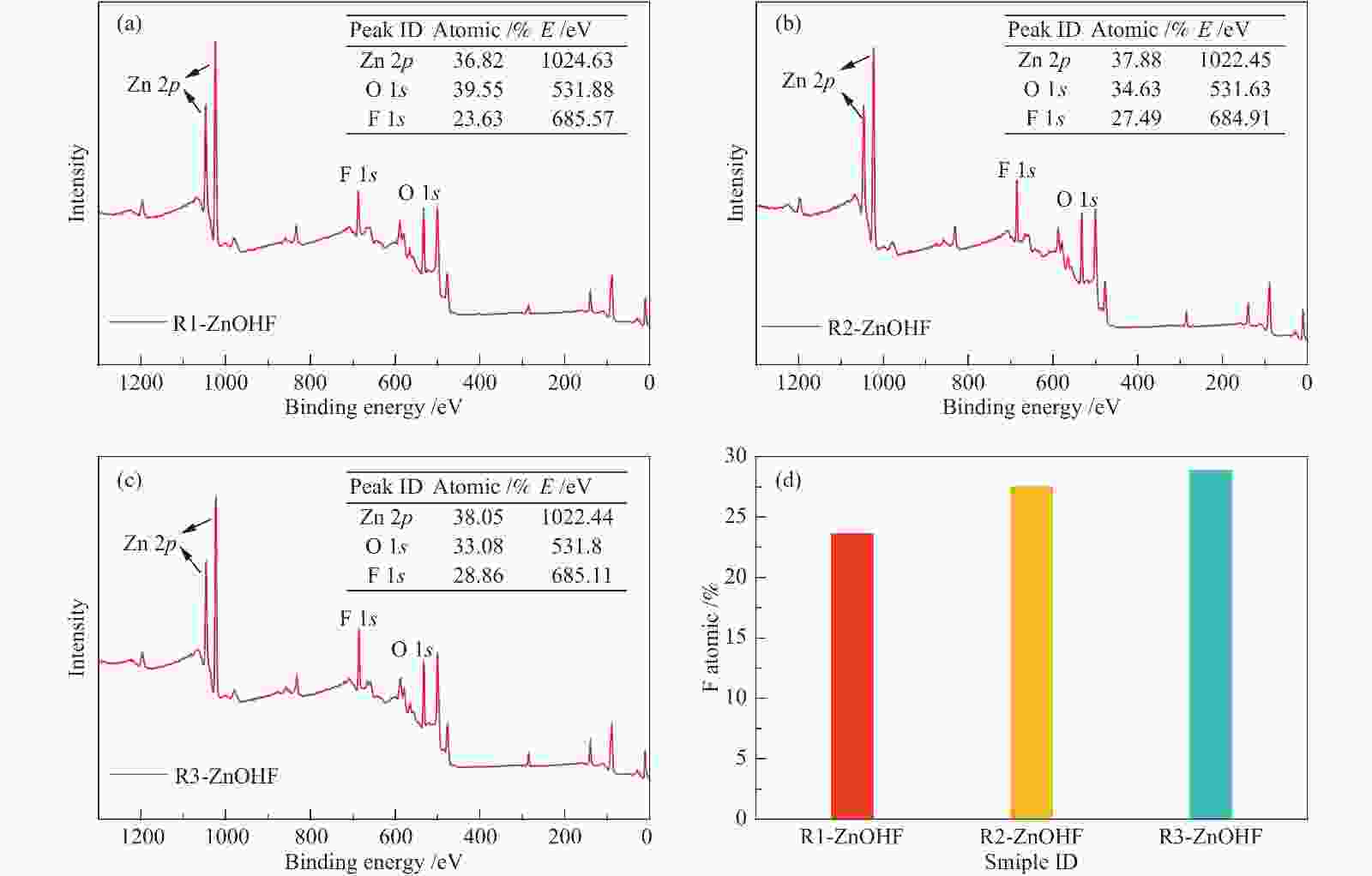
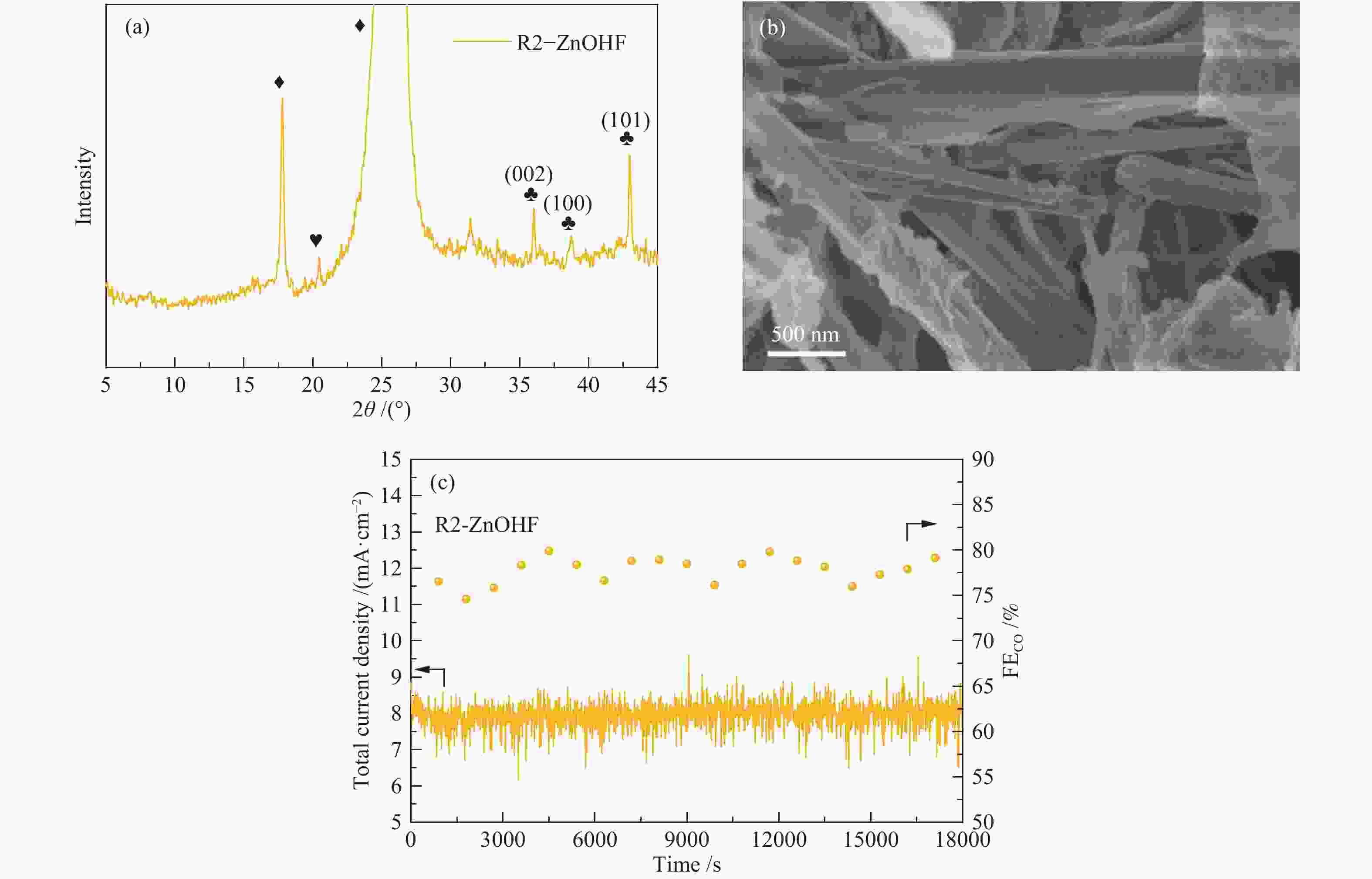
图 4 (a):R1-ZnOHF、(b):R2-ZnOHF、(c):R3-ZnOHF的XPS全谱图(表格为各元素含量及结合能位置);(d):R1-ZnOHF、R2-ZnOHF和R3-ZnOHF的表面F原子含量柱状对比
Figure 4 XPS survey spectra of (a): R1-ZnOHF; (b): R2-ZnOHF; (c): R3-ZnOHF (the table shows the content and binding energy position of each element); (d): surface F atom content for R1-ZnOHF, R2-ZnOHF and R3-ZnOHF
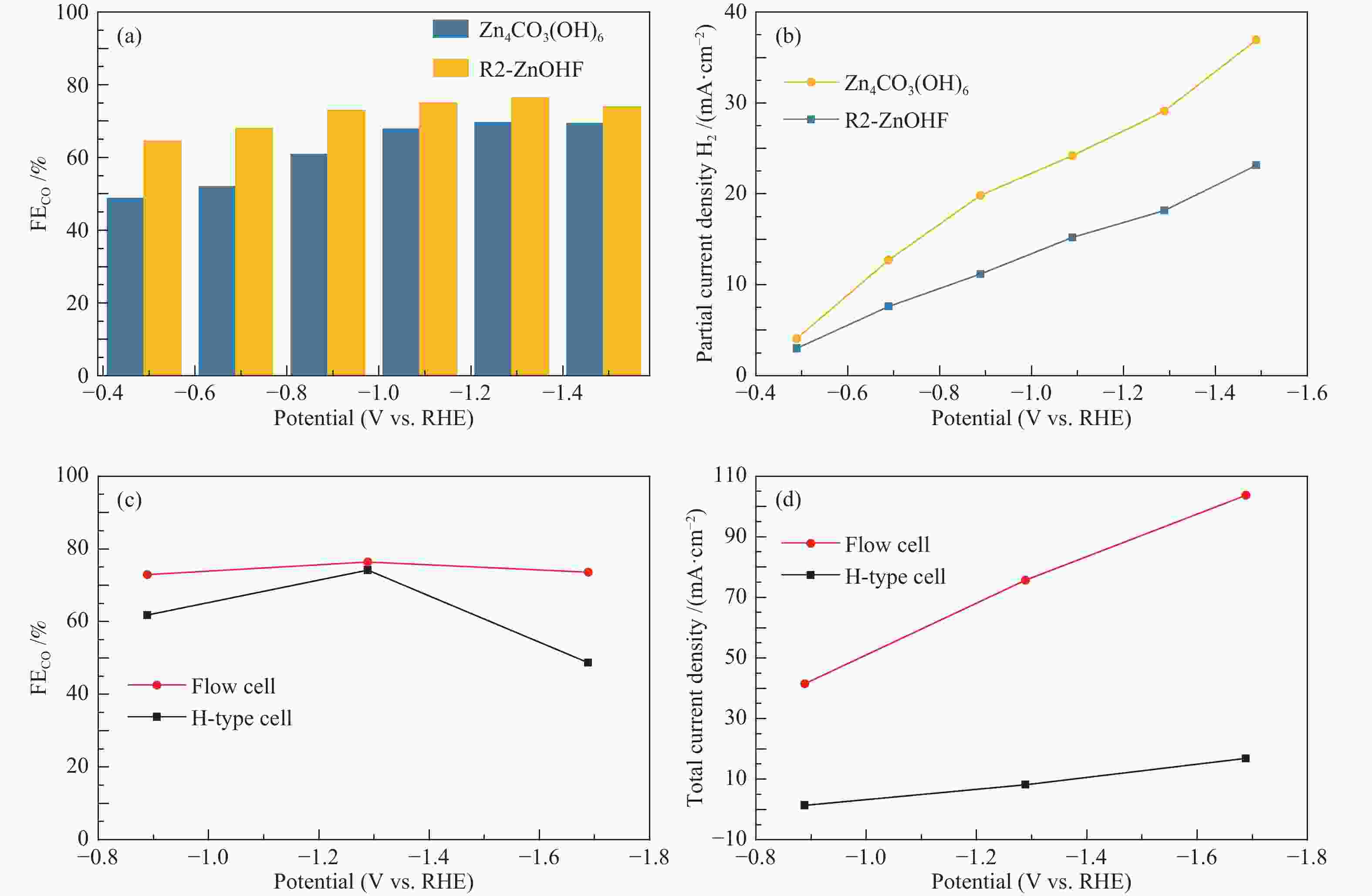
图 8 Zn4CO3(OH)6和R2-ZnOHF的(a)CO法拉第效率对比,(b)H2分电流密度对比;H型电解池和流动型电解池中R2-ZnOHF的(c)CO法拉第效率,(d)总电流密度
Figure 8 (a) CO Faraday efficiency comparison and (b) H2 partial current density comparison of Zn4CO3(OH)6 and R2-ZnOHF; (c) CO Faraday efficiency and (d) Total current density in H-type cell and Flow cell of R2-ZnOHF
-
[1] NEREM R S, BECKLEY B D, FASULLO J T, HAMLINGTON B D, MASTERS D, MITCHUM G T. Climate-change-driven accelerated sea-level rise detected in the altimeter era[J]. Proc Natl Acad Sci,2018,115(9):2022. doi: 10.1073/pnas.1717312115 [2] WIDLANSKY M J, LONG X, SCHLOESSER F. Increase in sea level variability with ocean warming associated with the nonlinear thermal expansion of seawater[J]. Commun Earth Environ,2020,1(1):9. doi: 10.1038/s43247-020-0008-8 [3] ZHENG T, JIANG K, WANG H. Recent advances in electrochemical CO2-to-CO conversion on heterogeneous catalysts[J]. Adv Mater,2018,30(48):1802066. doi: 10.1002/adma.201802066 [4] SUN Z, MA T, TAO H, FAN Q, HAN B. Fundamentals and challenges of electrochemical CO2 reduction using two-dimensional materials[J]. Chem,2017,3(4):560−587. doi: 10.1016/j.chempr.2017.09.009 [5] ZHANG L, ZHAO Z-J, GONG J. Nanostructured materials for heterogeneous electrocatalytic CO2 reduction and their related reaction mechanisms[J]. Angew Chem Int Ed,2017,56(38):11326−11353. doi: 10.1002/anie.201612214 [6] ROSS M B, DE LUNA P, LI Y, DINH C-T, KIM D, YANG P, SARGENT EH. Designing materials for electrochemical carbon dioxide recycling[J]. Nat Catal,2019,2(8):648−658. doi: 10.1038/s41929-019-0306-7 [7] ROSS M B, LI Y, DE LUNA P, KIM D, SARGENT E H, YANG P. Electrocatalytic rate alignment enhances syngas generation[J]. Joule,2019,3(1):257−264. doi: 10.1016/j.joule.2018.09.013 [8] BACK S, YEOM M S, JUNG Y. Active sites of Au and Ag nanoparticle catalysts for CO2 electroreduction to CO[J]. ACS Catal,2015,5(9):5089−5096. doi: 10.1021/acscatal.5b00462 [9] LIU S, TAO H, ZENG L, LIU Q, XU Z, LIU Q, LUO J-L. Shape-dependent electrocatalytic reduction of CO2 to CO on triangular silver nanoplates[J]. J Am Chem Soc,2017,139(6):2160−2163. doi: 10.1021/jacs.6b12103 [10] YANG W, DASTAFKAN K, JIA C, ZHAO C. Design of electrocatalysts and electrochemical cells for carbon dioxide reduction reactions[J]. Adv Mater Technol,2018,3(9):1700377. doi: 10.1002/admt.201700377 [11] GARZA A J, BELL A T, HEAD-GORDON M. Mechanism of CO2 reduction at copper surfaces: Pathways to C2 products[J]. ACS Catal,2018,8(2):1490−1499. doi: 10.1021/acscatal.7b03477 [12] ZHANG Y-J, SETHURAMAN V, MICHALSKY R, PETERSON AA. Competition between CO2 reduction and H2 evolution on transition-metal electrocatalysts[J]. ACS Catal,2014,4(10):3742−3748. doi: 10.1021/cs5012298 [13] CAVE E R, SHI C, KUHL K P, HATSUKADE T, ABRAM D N, HAHN C, CHAN K, JARAMILLO T F. Trends in the catalytic activity of hydrogen evolution during CO2 electroreduction on transition metals[J]. ACS Catal,2018,8(4):3035−3040. doi: 10.1021/acscatal.7b03807 [14] PAN Q, PENG J, SUN T, WANG S, WANG S. Insight into the reaction route of CO2 methanation: Promotion effect of medium basic sites[J]. Catal Commun,2014,45:74−78. doi: 10.1016/j.catcom.2013.10.034 [15] ZHAO S, JIN R, JIN R. Opportunities and challenges in CO2 reduction by gold- and silver-based electrocatalysts: From bulk metals to nanoparticles and atomically precise nanoclusters[J]. ACS Energy Lett,2018,3(2):452−462. doi: 10.1021/acsenergylett.7b01104 [16] CHEN Y, LI CW, KANAN MW. Aqueous CO2 reduction at very low overpotential on oxide-derived Au nanoparticles[J]. J Am Chem Soc,2012,134(49):19969−19972. doi: 10.1021/ja309317u [17] MUN Y, LEE S, CHO A, KIM S, HAN JW, LEE J. Cu-Pd alloy nanoparticles as highly selective catalysts for efficient electrochemical reduction of CO2 to CO[J]. Appl Catal B: Environ,2019,246:82−88. doi: 10.1016/j.apcatb.2019.01.021 [18] LUO W, ZHANG J, LI M, ZÜTTEL A. Boosting CO Production in Electrocatalytic CO2 reduction on highly porous Zn catalysts[J]. ACS Catal,2019,9(5):3783−3791. doi: 10.1021/acscatal.8b05109 [19] DENG W, MIN S, WANG F, ZHANG Z, KONG C. Efficient CO2 electroreduction to CO at low overpotentials using a surface-reconstructed and N-coordinated Zn electrocatalyst[J]. Dalton Trans,2020,49(17):5434−5439. doi: 10.1039/D0DT00800A [20] WON D H, SHIN H, KOH J, CHUNG J, LEE H S, KIM H, WOO S I. Highly efficient, selective, and stable CO2 electroreduction on a hexagonal Zn catalyst[J]. Angew Chem Int Ed,2016,55(32):9297−9300. doi: 10.1002/anie.201602888 [21] NGUYEN D L T, JEE M S, WON D H, OH H-S, MIN B K, HWANG Y J. Effect of halides on nanoporous Zn-based catalysts for highly efficient electroreduction of CO2 to CO[J]. Catal Commun,2018,114:109−113. doi: 10.1016/j.catcom.2018.06.020 [22] HSIEH Y-C, BETANCOURT L E, SENANAYAKE S D, HU E, ZHANG Y, XU W, POLYANSKY D E. Modification of CO2 reduction activity of nanostructured silver electrocatalysts by surface halide anions[J]. ACS Appl Energy Mater,2019,2(1):102−109. doi: 10.1021/acsaem.8b01692 [23] VARELA A S, JU W, REIER T, STRASSER P. Tuning the catalytic activity and selectivity of Cu for CO2 electroreduction in the presence of halides[J]. ACS Catal,2016,6(4):2136−2144. doi: 10.1021/acscatal.5b02550 [24] LIU K, SMITH W A, BURDYNY T. Introductory guide to assembling and operating gas diffusion electrodes for electrochemical CO2 reduction[J]. ACS Energy Lett,2019,4(3):639−643. doi: 10.1021/acsenergylett.9b00137 [25] WEEKES D M, SALVATORE D A, REYES A, HUANG A, BERLINGUETTE C P. Electrolytic CO2 reduction in a flow cell[J]. Acc Chem Res,2018,51(4):910−918. doi: 10.1021/acs.accounts.8b00010 [26] REN S, JOULIÉ D, SALVATORE D, TORBENSEN K, WANG M, ROBERT M, BERLINGUETTE C P. Molecular electrocatalysts can mediate fast, selective CO2 reduction in a flow cell[J]. Science,2019,365(6451):367. doi: 10.1126/science.aax4608 [27] ZHONG D, ZHANG L, ZHAO Q, CHENG D, DENG W, LIU B, ZHANG G, DONG H, YUAN X, ZHAO Z, LI J, GONG J. Concentrating and activating carbon dioxide over AuCu aerogel grain boundaries[J]. J Chem Phys,2020,152(20):204703. doi: 10.1063/5.0007207 [28] AHMAD S, RAWAT P, NAGARAJAN R. Facile green synthesis of Zn(OH)F from the single source precursor KZnF3[J]. Mater Lett,2015,139:86−88. doi: 10.1016/j.matlet.2014.10.037 [29] GONG X, YU L, TIAN G, WANG L, ZHAO Y, MAI W, WANG W. Synthesis and characterization of flower-like ZnO nanostructures via flower-like ZnOHF intermediate[J]. Mater Lett,2014,127:36−39. doi: 10.1016/j.matlet.2014.04.071 [30] MENG F, HOU N, JIN Z, SUN B, GUO Z, KONG L, XIAO X, WU H, LI M, LIU J. Ag-decorated ultra-thin porous single-crystalline ZnO nanosheets prepared by sunlight induced solvent reduction and their highly sensitive detection of ethanol[J]. Sens Actuators, B,2015,209:975−982. doi: 10.1016/j.snb.2014.12.078 [31] ZHONG D, ZHAO Z-J, ZHAO Q, CHENG D, LIU B, ZHANG G, DENG W, DONG H, ZHANG L, LI J, LI J, GONG J. Coupling of Cu(100) and (110) facets promotes carbon dioxide conversion to hydrocarbons and alcohols[J]. Angew Chem Int Ed,2021,60(9):4879−4885. doi: 10.1002/anie.202015159 [32] LI J, CHANG K, ZHANG H, HE M, GODDARD W A, CHEN J G, CHENG M-J, LU Q. Effectively increased efficiency for electroreduction of carbon monoxide using supported polycrystalline copper powder electrocatalysts[J]. ACS Catal,2019,9(6):4709−4718. doi: 10.1021/acscatal.9b00099 [33] QIN B, LI Y, FU H, WANG H, CHEN S, LIU Z, PENG F. Electrochemical reduction of CO2 into tunable syngas production by regulating the crystal facets of earth-abundant Zn catalyst[J]. ACS Appl Mater Interfaces,2018,10(24):20530−20539. doi: 10.1021/acsami.8b04809 -





 下载:
下载: