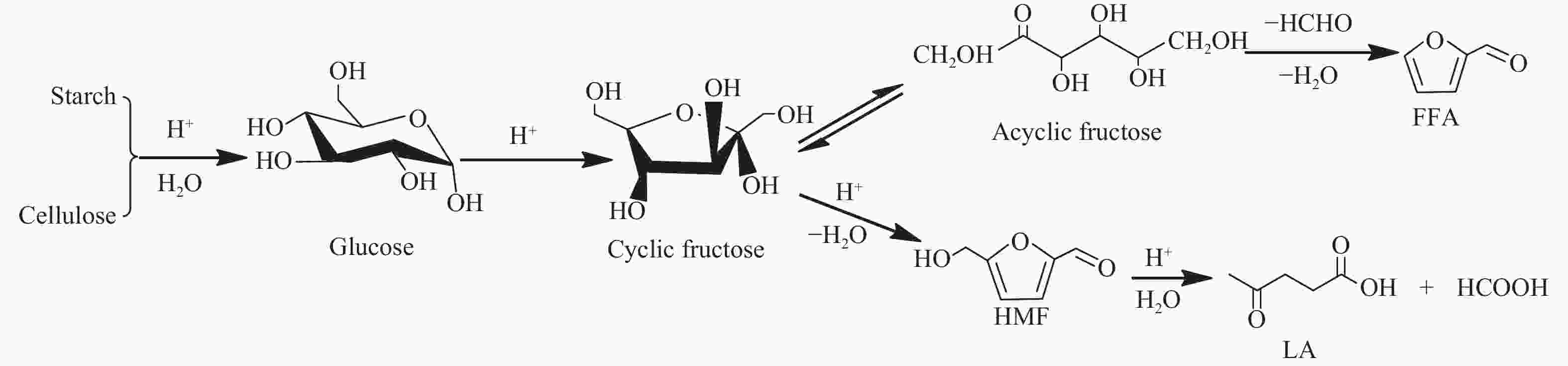
Catalytic conversion of cellulose and starch to furfural over zeolites
-
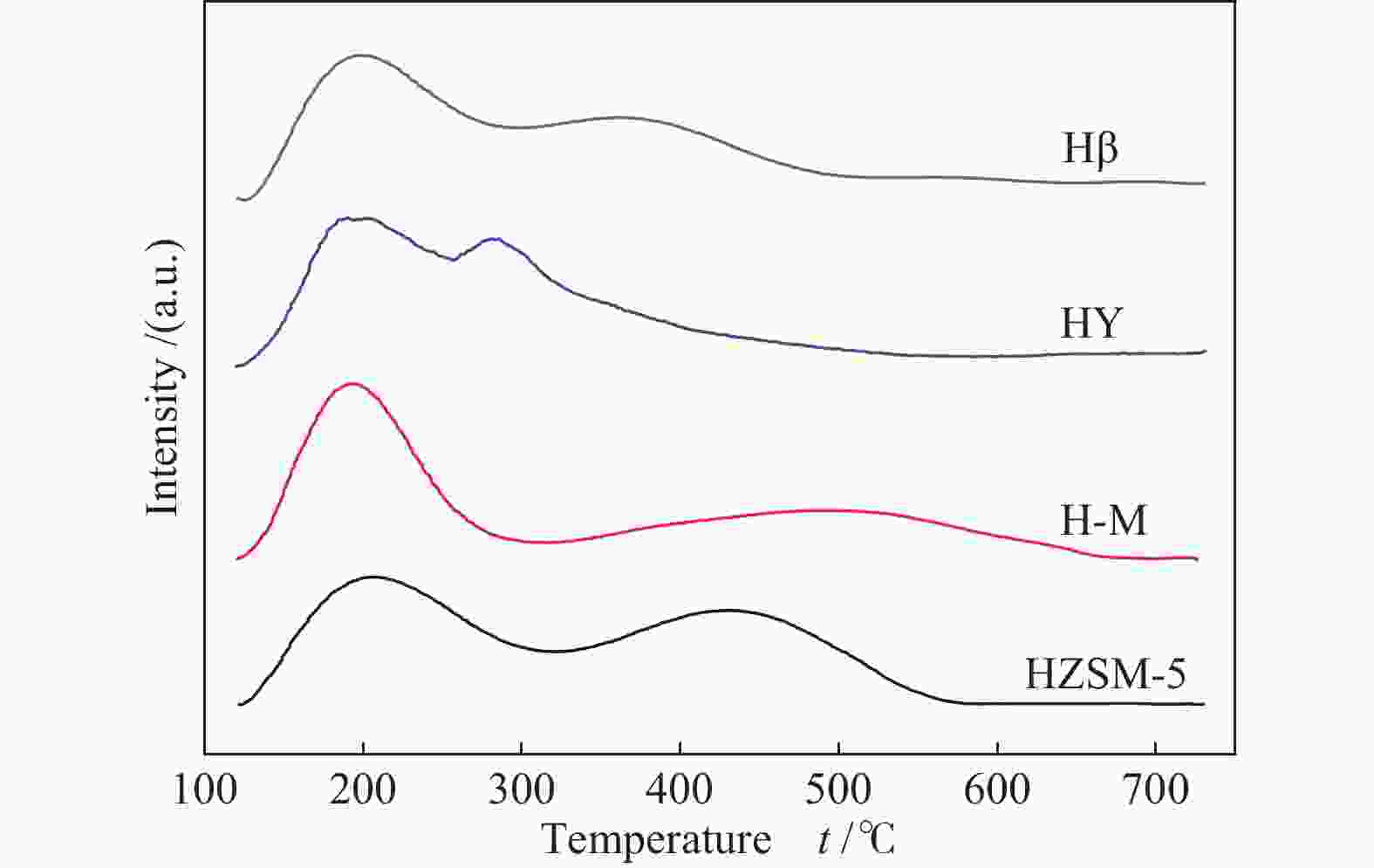
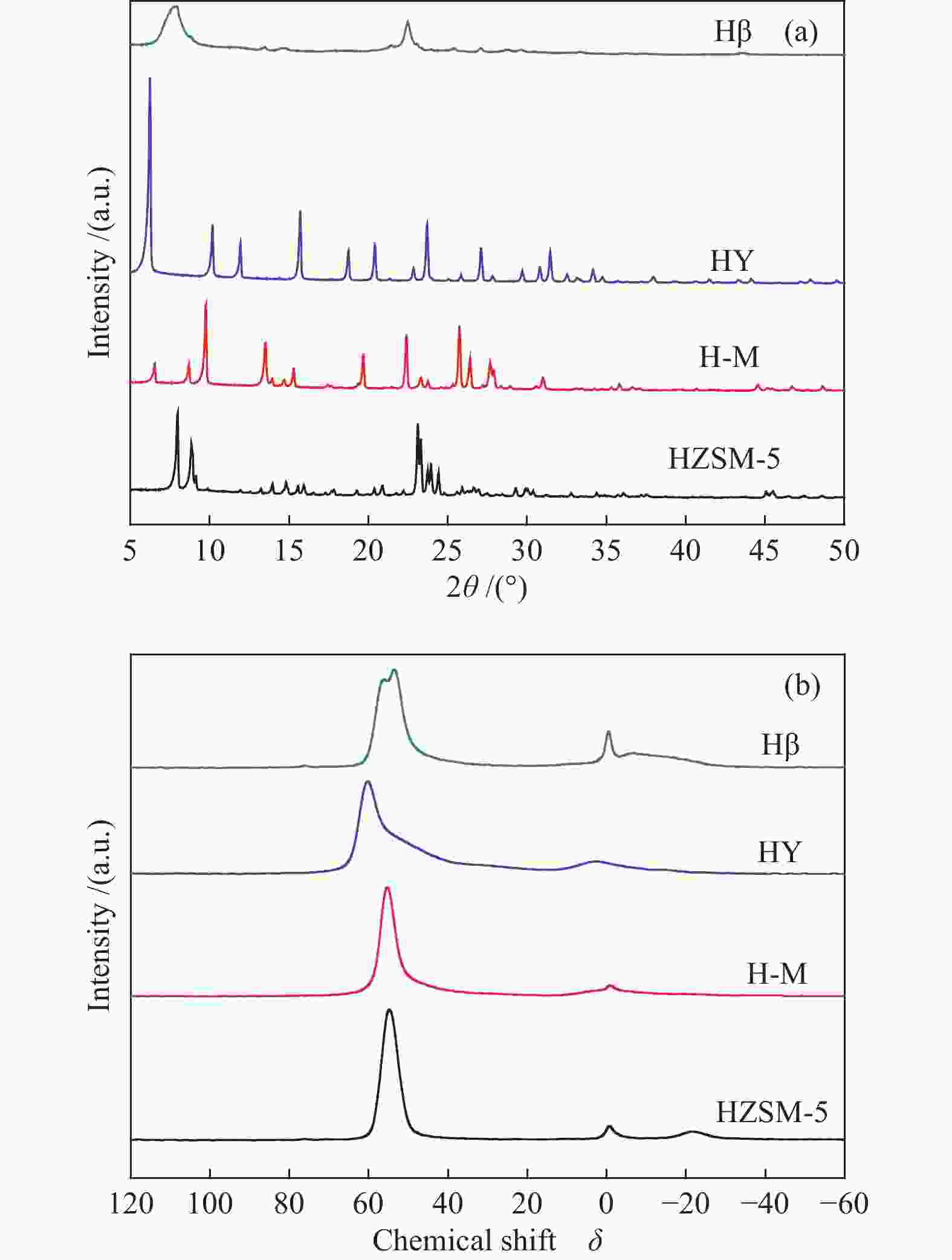
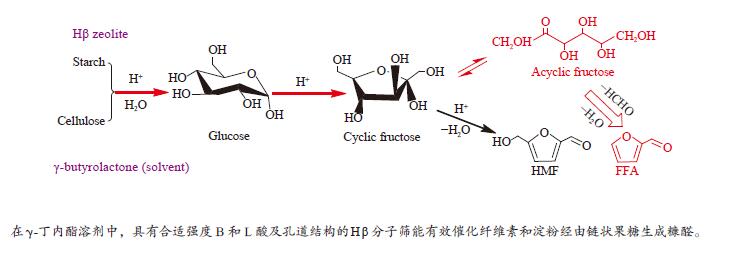
摘要: 采用四种分子筛进行纤维素和淀粉催化转化反应,并借助X射线衍射、铝核磁、吡啶吸附红外光谱和NH3-TPD等手段,对分子筛酸性质及孔道结构在催化纤维素和淀粉转化过程中的作用规律进行了研究。结果表明,Hβ分子筛上合适的B酸和L酸强度、数量及孔道结构使纤维素和淀粉主要反应生成糠醛。HY分子筛因酸性相对较弱,无法有效催化纤维素转化,但能有效催化淀粉生成5-羟甲基糠醛。H-mordenite和HZSM-5分子筛没有足够的L酸位,使纤维素和淀粉反应生成的葡萄糖无法异构为果糖,抑制了果糖进一步转化为糠醛或5-羟甲基糠醛的反应。5-羟甲基糠醛的生成取决于分子筛的酸性质,糠醛的生成除了由分子筛的酸性质决定,还需要合适的孔道结构。Abstract: Conversion of cellulose and starch to furfural was investigated over four zeolites. The zeolites were characterized by X-ray diffraction, 27Al MAS NMR, IR spectra of pyridine adsorption and NH3 temperature-programmed desorption. The roles of acidity and pore structure of zeolites in conversion of cellulose and starch were discussed in detail. The results showed that Hβ zeolite with appropriate Brønsted acid sites, Lewis acid sites and pore structure was effective to produce furfural from cellulose and starch. HY zeolite could not catalyze cellulose reaction with high conversion because of its weak acidity. However, HY zeolite was effective to produce 5-hydroxymethylfurfural (HMF) from starch. H-mordenite and HZSM-5 zeolites with fewer Lewis acid sites could not cause the isomerization reaction from glucose to fructose. So, the further conversion of fructose to furfural or HMF was inhibited. The formation of HMF only depended on the acid properties of zeolites. The formation of furfural was not only determined by the acidity of zeolites, but also by their appropriate pore structure.
-
Key words:
- zeolite /
- cellulose /
- starch /
- furfural /
- 5-hydroxymethylfurfural
-
表 1 分子筛的化学组成、织构性质及酸性质
Table 1 Chemical composition, physical properties and acidity of zeolites
Catalyst Si/Ala SBETb/(m2·g−1) Smicrob/(m2·g−1) Py-FTIR/(μmol·g−1) Pore diameter/Å[19] Bc Lc B/Lc Bd Ld Hβ 13 485 332 312 178 1.8 221 159 6.6 × 6.7 5.6 × 5.6 HY 3 520 443 301 125 2.4 133 89 7.4 × 7.4 H-Me 9 406 349 297 42 7.1 261 13 7.0 × 6.7 3.4 × 4.8 HZSM-5 19 318 190 326 31 10.5 299 17 5.1 × 5.5 5.3 × 5.6 a: determined by ICP analysis; b: SBET: BET surface area, Smicro: micropore area; c: the acid density was determined after desorption at 200 ℃; d: the acid density was determined after desorption at 350 ℃; e: H-M: H-mordenite 表 2 纤维素在不同分子筛上的转化a
Table 2 Conversion of cellulose over various zeolites a
Entry Catalyst Conversion x/% Yield w/% glu FFA HMF 1 Hβ 96.9 0.2 34.2 1.0 2 HY 22.3 0.1 2.0 2.7 3 H-M 42.5 14.9 1.2 2.4 4 HZSM-5 62.0 24.6 3.0 3.7 a: 0.3 g catalyst, 10.5 g cellulose/γ-butyrolactone-water, cellulose concentration 4.8%, water concentration 20%, 170 ℃, 180 min, 2 MPa N2; glu: glucose, HMF: 5-hydroxymethylfurfural, FFA: furfural 表 3 淀粉在不同分子筛上的转化a
Table 3 Conversion of starch over various zeolites a
Entry Catalyst Conversion x/% Yield w/% glu FFA HMF LA 1 Hβ 99.9 0.6 41.9 8.8 0.4 2 HY 99.9 1.4 7.6 47.4 3.7 3 H-M 74.4 39.7 0.7 1.2 1.5 4 HZSM-5 83.6 44.5 1.8 2.6 8.4 a: 0.2 g catalyst, 10.5 g starch/γ-butyrolactone-water, starch concentration 4.8%, water concentration 4.8%, 170 ℃, 40 min, 2 MPa N2; LA: levulic acid 表 4 葡萄糖在不同分子筛上的转化a
Table 4 Conversion of glucose over various zeolites a
Entry Catalyst Conversion x/% Yield w/% fru FFA HMF LA 1 Hβ 99.9 2.1 42.8 14.0 13.3 2 HY 99.8 3.2 6.2 48.2 8.1 3 H-M 64.3 2.0 1.1 1.8 4.5 4 HZSM-5 69.8 0.7 3.5 1.9 14.7 a: 0.2 g catalyst, 10.5 g glucose/γ-butyrolactone-water, glucose concentration 4.8%, water concentration 4.8%, 170 ℃, 40 min, 2 MPa N2; fru: fructose 表 5 果糖在不同分子筛上的转化a
Table 5 Conversion of fructose over various zeolites a
Entry Catalyst Conversion x/% Yield w/% FFA HMF LA 1 Hβ 99.9 49.5 17.5 12.4 2 HY 99.9 3.4 56.0 3.4 3 H-M 87.8 25.2 39.7 3.0 4 HZSM-5 99.7 23.0 48.7 11.9 a: 0.2 g catalyst, 10.5 g fructose/γ-butyrolactone-water, fructose concentration 4.8%, water concentration 4.8%, 170 ℃, 40 min, 2 MPa N2 表 6 淀粉在Hβ分子筛上的转化a
Table 6 Conversion of starch over Hβ zeolite a
Entry t/ ℃ Water
concentration
w/%Conversion
x/%Yield w/% glu fru FFA HMF 1 140 4.8 95.0 9.8 2.0 29.4 13.8 2 150 4.8 96.8 4.2 0.1 37.8 11.1 3 160 4.8 97.5 1.2 0.1 40.8 10.3 4 170 4.8 99.9 0.6 0.1 41.9 8.8 5 180 4.8 99.9 0.1 0.1 37.4 7.9 6 170 0 99.0 0.6 0.1 37.5 10.7 7 170 9.5 99.9 0.2 0.1 42.7 9.4 8 170 15 99.9 0.2 0.1 38.4 13.3 9 170 20 99.9 0.4 0.1 36.5 18.3 10 170 30 99.0 3.7 1.2 28.8 28.4 11 170 40 99.0 16.7 4.8 20.8 27.2 12c 170 9.5 99.9 0.3 0.1 39.5 9.8 13d 170 9.5 99.9 0.1 0.1 38.1 9.6 a: 0.2 g catalyst, 10.5 g starch/γ-butyrolactone-water, starch concentration 4.8%, 40 min, 2 MPa N2; c: 90 min; d: 180 min 表 7 纤维素在Hβ分子筛上的转化a
Table 7 Conversion of cellulose over Hβ zeolite a
Entry t/ ℃ Water concentration w/% Conversion x/% Yield w/% glu fru FFA HMF LA 1 160 20 47.5 0.5 0.1 21.0 2.0 26.7 2 170 20 96.9 0.2 0.1 34.2 1.0 28.0 3 180 20 98.3 0.1 0.1 30.2 0.6 20.6 4 170 0 85.3 0.1 0.1 22.7 2.1 5.5 5 170 4.8 89.9 0.1 0.1 25.5 4.3 7.5 6 170 9.5 90.1 0.2 0.1 27.5 1.2 13.4 7 170 15 96.0 0.2 0.1 30.9 0.3 22.9 8 170 30 65.3 1.0 0.1 26.9 2.3 35.0 9 170 40 42.5 1.3 0.3 13.3 1.9 22.2 10b 170 20 62.4 0.6 0.3 21.8 2.6 18.4 11c 170 20 83.7 0.4 0.2 29.4 2.0 22.2 12d 170 20 98.0 0.1 0.1 28.6 0.6 20.8 a: Hβ 0.3 g, 10.5 g cellulose/γ-butyrolactone-water, cellulose concentration 4.8%; 180 min, 2 MPa N2; b: 40 min; c: 90 min; d: 300 min 表 8 Hβ分子筛的重复利用a
Table 8 Reusability of Hβ zeolite for converting starch and cellulose a
Entry Recycling runs Substrate Reaction time/min Water concentration w/% FFA yield w/% 1 0 starch 40 9.5 42.7 2 1 starch 40 9.5 37.6 3 2 starch 40 9.5 35.8 4 3 starch 40 9.5 33.3 5 4 starch 40 9.5 32.9 6b 5 starch 40 9.5 39.6 7 0 cellulose 180 20 34.2 8 1 cellulose 180 20 28.3 9 2 cellulose 180 20 24.5 10 3 cellulose 180 20 20.3 11b 4 cellulose 180 20 29.7 a: starch, Hβ 0.2 g; cellulose, Hβ 0.3 g; 10.5 g starch or cellulose/γ-butyrolactone-water, starch or cellulose concentration 4.8%; 170 ℃,2 MPa N2; b: the Hβ zeolite after recycling runs was calcined at 500 ℃ for 5 h -
[1] KARINEN R, VILONEN K, NIEMELA M. Biorefining: Heterogeneously catalyzed reactions of carbohydrates for the production of furfural and hydroxymethylfurfural[J]. ChemSusChem,2011,4(8):1002−1016. doi: 10.1002/cssc.201000375 [2] 袁正求, 龙金星, 张兴华, 夏 莹, 王铁军, 马隆龙. 木质纤维素催化转化制备能源平台化合物[J]. 化学进展,2016,28(1):103−110. doi: 10.7536/PC150744YUAN Zheng-qiu, LONG Jin-xing, ZHANG Xing-hua, XIA Ying, WANG Tie-jun, MA Long-long. Catalytic conversion of lignocellulose into energy platform chemicals[J]. Prog Chem,2016,28(1):103−110. doi: 10.7536/PC150744 [3] WANG Y, DING G, YANG X, ZHENG H, ZHU Y, LI Y. Selectively convert fructose to furfural or hydroxymethylfurfural on Beta zeolite: The manipulation of solvent effects[J]. Appl Catal B: Environ,2018,235:150−157. doi: 10.1016/j.apcatb.2018.04.043 [4] BOZELL J J, PETERSEN G R. Technology development for the production of biobased products from biorefinery carbohydrates–the US Department of Energy’s “Top 10” revisited[J]. Green Chem,2010,12(4):539−554. doi: 10.1039/b922014c [5] 郑洪岩, 王月清, 常西亮, 牛宇岚, 杨红, 宋永波, 姚英, 丁国强, 朱玉雷. Hβ分子筛催化甜高粱秆汁转化制呋喃类化合物[J]. 燃料化学学报,2019,47(5):605−610. doi: 10.3969/j.issn.0253-2409.2019.05.012ZHENG Hong-yan, WANG Yue-qing, CHANG Xi-liang, NIU Yu-lan, YANG Hong, SONG Yong-bo, YAO Ying, DING Guo-qiang, ZHU Yu-lei. Catalytic conversion of sweet sorghum stalk juice to furan compounds over Hβ zeolite[J]. J Fuel Chem Technol,2019,47(5):605−610. doi: 10.3969/j.issn.0253-2409.2019.05.012 [6] MOREAUA C, BELGACEM M N, GANDINI A. Recent Catalytic advances in the chemistry of substituted furans from carbohydrates and in theensuing polymers[J]. Top Catal,2004,27(1/4):11−30. [7] DUTTA S, DE S, SAHA B, ALAM MD I. Advances in conversion of hemicellulosic biomass to furfural and upgrading to biofuels[J]. Catal Sci Technol,2012,2(10):2025−2036. doi: 10.1039/c2cy20235b [8] LANGE J P, VAN DER HEIDE E, VAN BUIJTENEN J, PRICE R. Furfural-a promising platform for lignocellulosic biofuels[J]. ChemSusChem,2012,5(1):150−166. doi: 10.1002/cssc.201100648 [9] 黄仲涛, 耿建铭. 工业催化[M]. 3版. 北京: 化学工业出版社, 2014, 70−71.HUANG Zhong-tao, GENG Jian-ming. Industrial Catalysis[M]. 3nd ed. Beijing: Chemical Industry Press, 2014, 70−71. [10] GÜRBÜZ E I, GALLO J R, ALONSO D M, WETTSTEIN S G, LIM W Y, DUMESIC J A. Conversion of hemicellulose into furfural using solid acid catalysts in γ-valerolactone[J]. Angew Chem Int Ed,2013,52(4):1270−1274. doi: 10.1002/anie.201207334 [11] WANG Y, YANG X, ZHENG H, LI X, ZHU Y, LI Y. Mechanistic insights on catalytic conversion fructose to furfural on beta zeolite via selective carbon-carbon bond cleavage[J]. Mol Catal,2019,463:130−139. [12] CUI J, TAN J, DENG T, CUI X, ZHU Y, LI Y. Conversion of carbohydrates to furfural via selective cleavage of the carbon-carbon bond: the cooperative effects of zeolite and solvent[J]. Green Chem,2016,18(6):1619−1624. doi: 10.1039/C5GC01948F [13] KIM B, JEONG J, LEE D, KIM S, YOON H J, LEE Y S, CHO J K. Direct transformation of cellulose into 5-hydroxymethyl-2-furfural using a combination of metal chlorides in imidazolium ionic liquid[J]. Green Chem,2011,13(6):1503−1506. doi: 10.1039/c1gc15152e [14] 王小艳, 秦磊, 刘辉, 李凡, 李春, 佟毅. 淀粉质燃料乙醇发酵胁迫及菌株耐受性改造[J]. 精细化工,2019,36(4):568−574.WANG Xiao-yan, QIN Lei, LIU Hui, LI Fan, LI Chun, TONG Yi. Research progress of starchy fuel ethanol fermentation and the tolerance of Saccharomyces cerevisiae[J]. Fine Chem,2019,36(4):568−574. [15] 罗虎, 孙振江, 李永恒, 梁坤国, 许旺发. 玉米淀粉生产酒精的研究[J]. 酿酒科技,2018,(2):30−33.LUO Hu, SUN Zhen-jiang, LI Yong-heng, LIANG Kun-guo, XU Wang-fa. Alcohol production by corn starch[J]. Liquor-making Sci Technol,2018,(2):30−33. [16] WANG J, XI J, WANG Y. Recent advances in the catalytic production of glucose from lignocellulosic biomass[J]. Green Chem,2015,17(2):737−751. [17] YANG Y, XIANG X, TON D G, HU C, ABU-OMAR M M. One-pot synthesis of 5-hydroxymethylfurfural directly from starch over SO42−/ZrO2-Al2O3 solid catalyst[J]. Bioresource Technol,2012,116:302−306. doi: 10.1016/j.biortech.2012.03.081 [18] 徐如人, 庞文琴, 于吉红, 霍启升, 陈接胜. 分子筛与多孔材料化学[M]. 北京: 科学出版社, 2004, 247−252.XU Ru-ren, PANG Wen-qin, YU Ji-hong, HUO Qi-sheng, CHEN Jie-sheng. Chemistry-Zeolites and Porous Materials[M]. Beijing: Science Press, 2004, 247−252. [19] 栗同林, 刘希尧, 朴玉玲, 蔡春飞, 王祥生. 萘与不同烷基化试剂在沸石上的烷基化反应[J]. 催化学报,1998,19(2):181−183. doi: 10.3321/j.issn:0253-9837.1998.02.019LI Tong-lin, LIU Xi-yao, PIAO Yu-ling, CAI Chun-fei, WANG Xing-sheng. Alkylation of naphthalene with various alkylating agents over some zeolites[J]. Chin J Catal,1998,19(2):181−183. doi: 10.3321/j.issn:0253-9837.1998.02.019 [20] 刘萌, 吴志杰, 潘 涛. 沸石分子筛酸性质表征方法研究进展[J]. 应用化学,2020,37(1):1−15. doi: 10.11944/j.issn.1000-0518.2020.01.190199LIU Meng, WU Zhi-jie, PAN Tao. Recent advance in the characterization of acidic properties of zeolites[J]. Chin J Appl Chem,2020,37(1):1−15. doi: 10.11944/j.issn.1000-0518.2020.01.190199 [21] YUE C, LI G, PIDKO E A, WIESFELD J J, RIGUTTO M, HENSEN E J M. Dehydration of glucose to 5-hydroxyme thylfurfural using Nb-doped tungstite[J]. ChemSusChem,2016,9(17):2421−2429. -




 下载:
下载: