Effect of Lewis acid sites of FER zeolite on catalytic transformation of isobutene
-
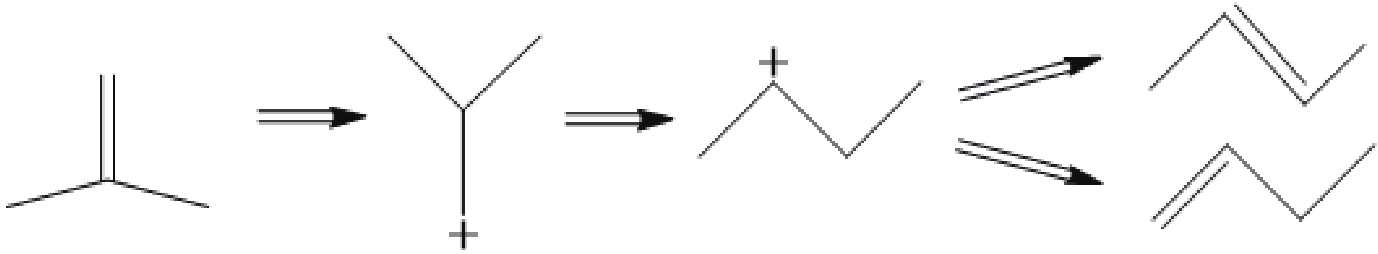
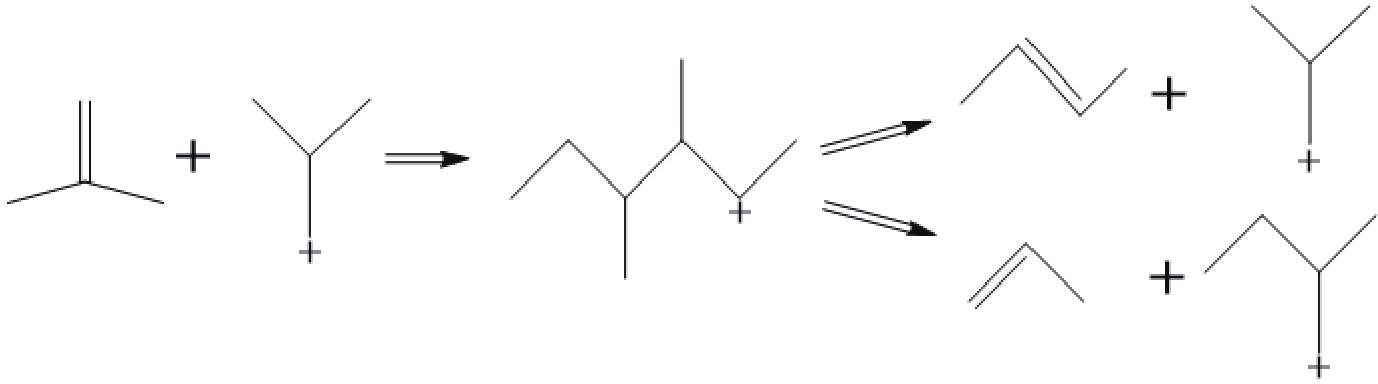
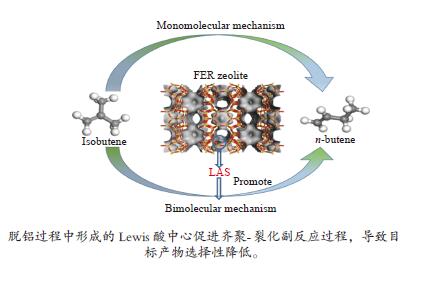
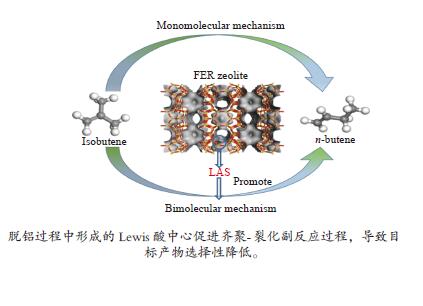
摘要: 采用不同浓度六氟硅酸铵(AHFS)对FER型分子筛进行同晶置换改性,制备了一系列具有不同酸类型、酸密度及可接近性等特征的改性FER分子筛样品,运用XRD、N2吸附-脱附等温线等方法对其织构性质进行表征,通过NH3-TPD和Py-FTIR关联改性FER分子筛的酸性质,并结合反应评价系统探究了改性FER分子筛酸中心与丁烯(1-丁烯、异丁烯)骨架异构化反应间的构效关系。结果表明,根据产物收率确定,以1-丁烯和异丁烯分别作为原料时其骨架异构化的最佳反应温度均为350 ℃,以异丁烯作为原料时副反应更为明显。在脱铝过程中,由于脱铝剂和分子筛中的非骨架铝羟基物种的相互作用,产生了强弱不同的两种新Lewis酸中心,在反应过程中上述两种Lewis酸中心促进了异丁烯齐聚-裂化反应的进行,进而降低了主反应的选择性。Abstract: A series of modified FER zeolite samples with different acid types, acid density and accessibility were prepared by using different concentrations of ammonium hexafluorosilicate (AHFS) for isomorphous substitution. The texture properties were characterized by XRD and N2 adsorption isotherms. And the acid properties were characterized by NH3-TPD and Py-FTIR. The results showed that when 1-butene and isobutene were respectively used as raw materials, the optimal reaction temperature for skeleton isomerization was 350 ℃, and the side reaction was more obvious when isobutene was used as raw material. During the dealumination process, two new Lewis acid sites with different strength were generated, due to the interaction between the dealumination agent and the extra framework aluminum hydroxyl species on the zeolite. During the reaction, the above two Lewis acid sites promoted the oligomerization-cracking of isobutene, thereby, reducing the selectivity of the main reaction.
-
Key words:
- zeolite /
- catalysis /
- FER /
- butene /
- Lewis acid
-
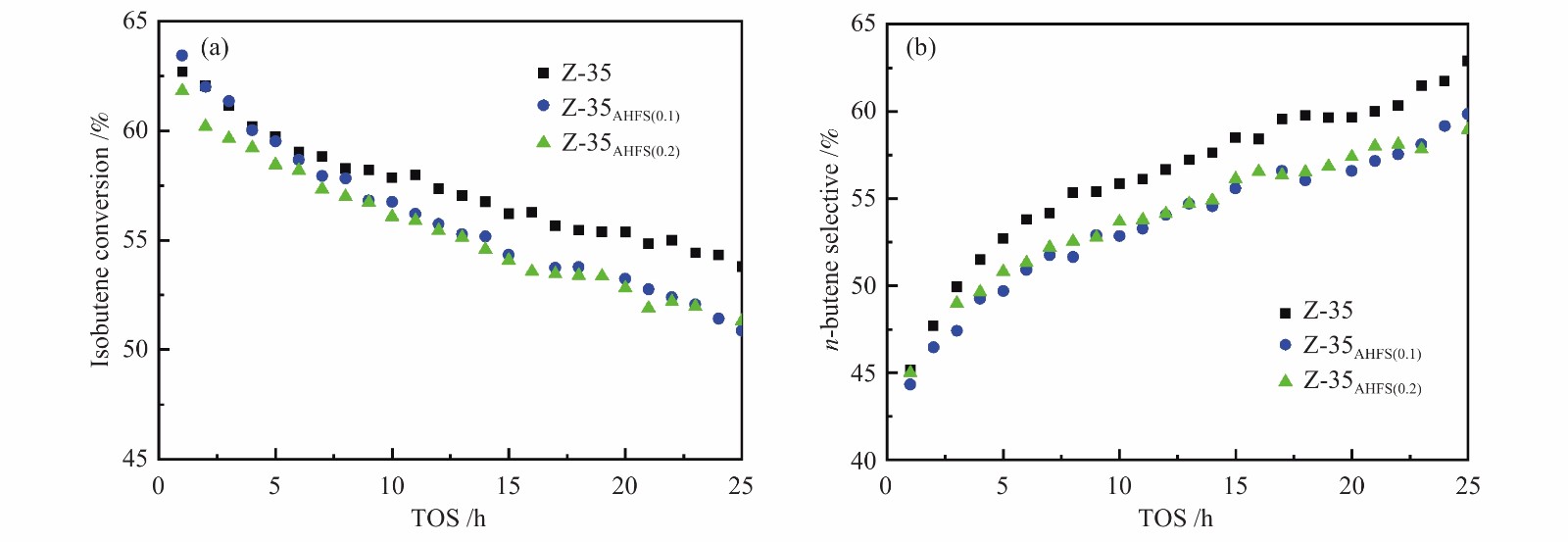
表 1 不同温度下正丁烯骨架异构化和异丁烯骨架异构化反应评价表
Table 1 Evaluation of isomerization reaction of n-butene and isobutene at different temperatures
t/℃ n-butene isomerization isobutene isomerization conv.
x/ %selec.
s/%yield.
y/%conv.
x/%selec.
s/%yield
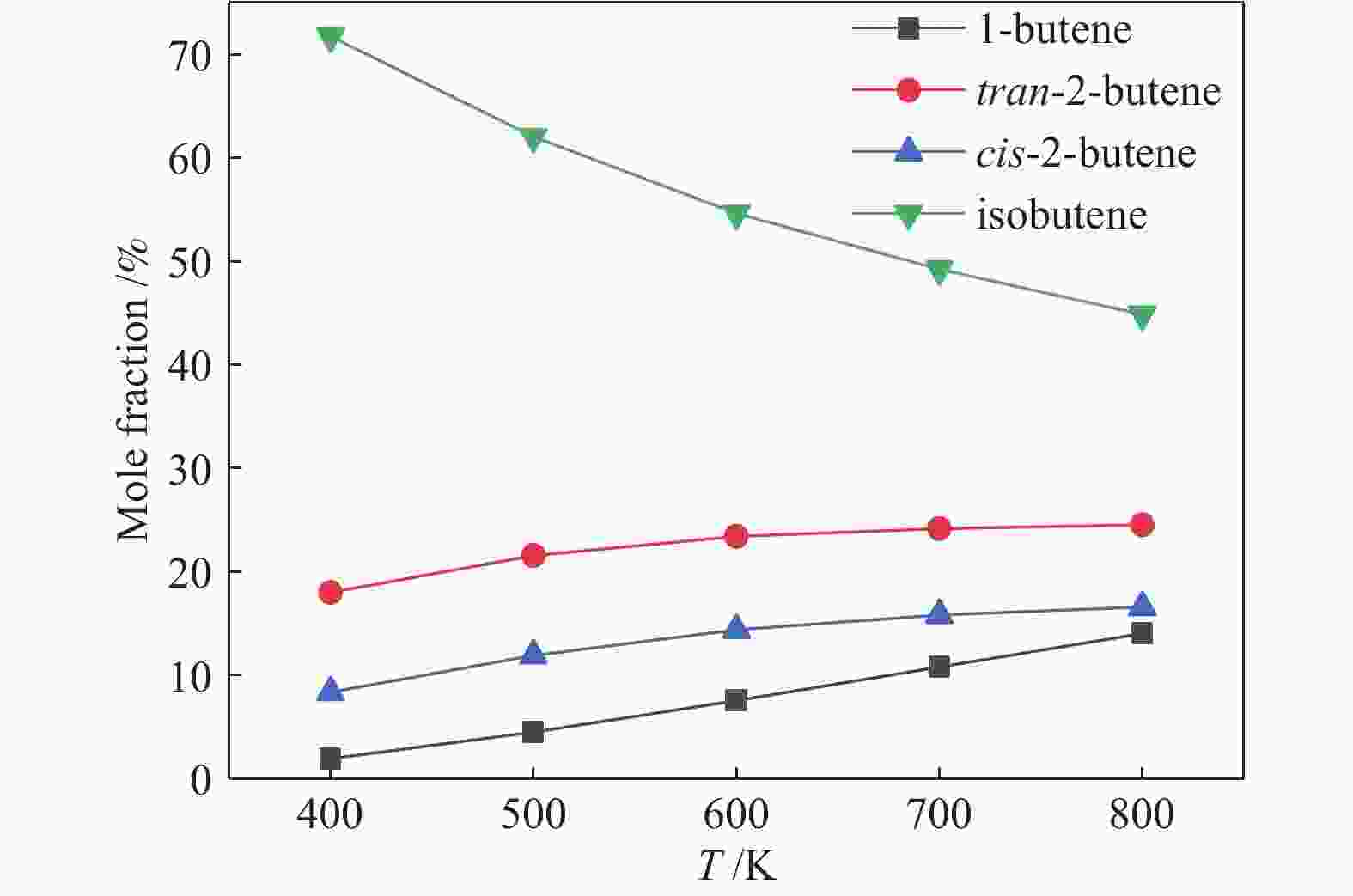
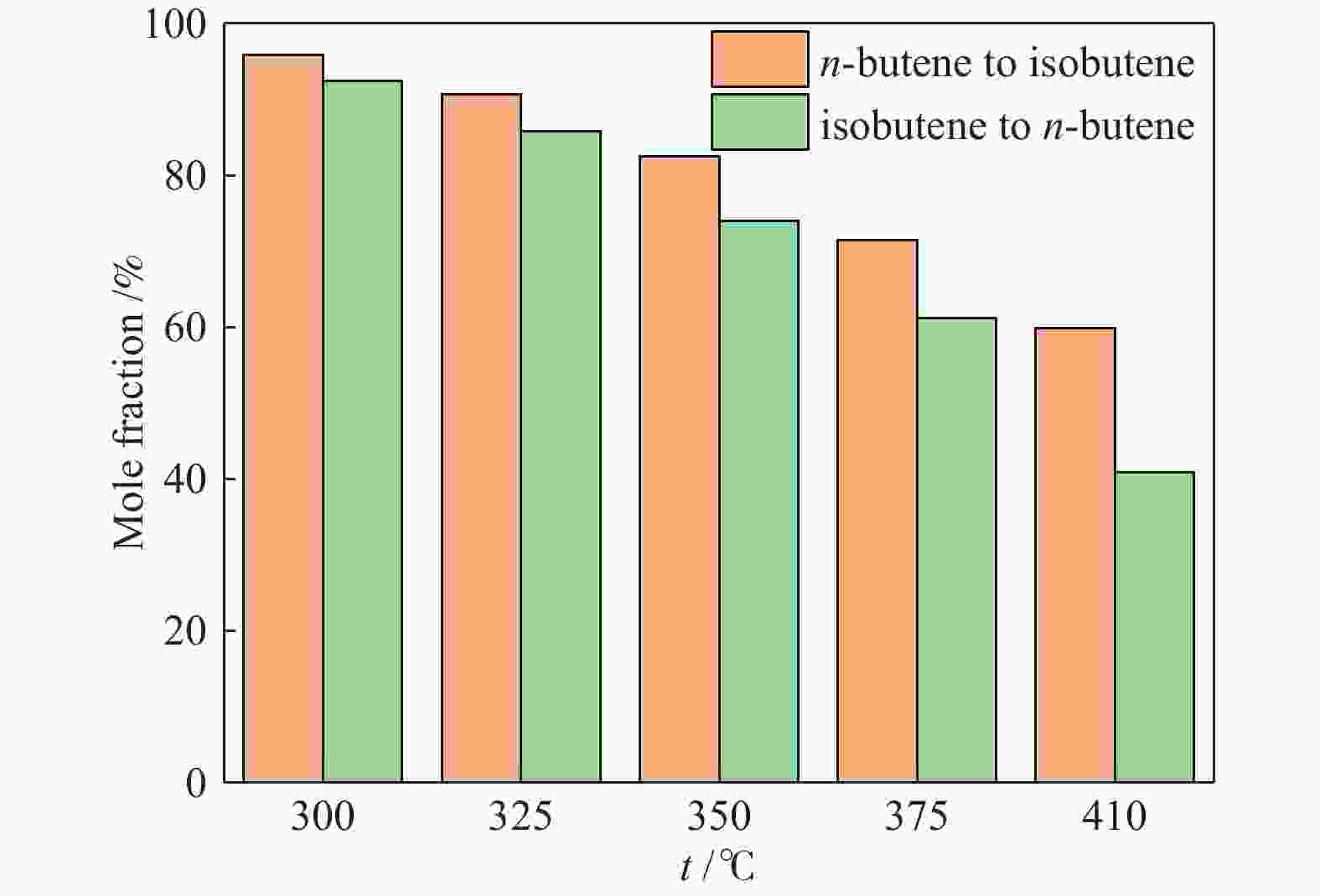
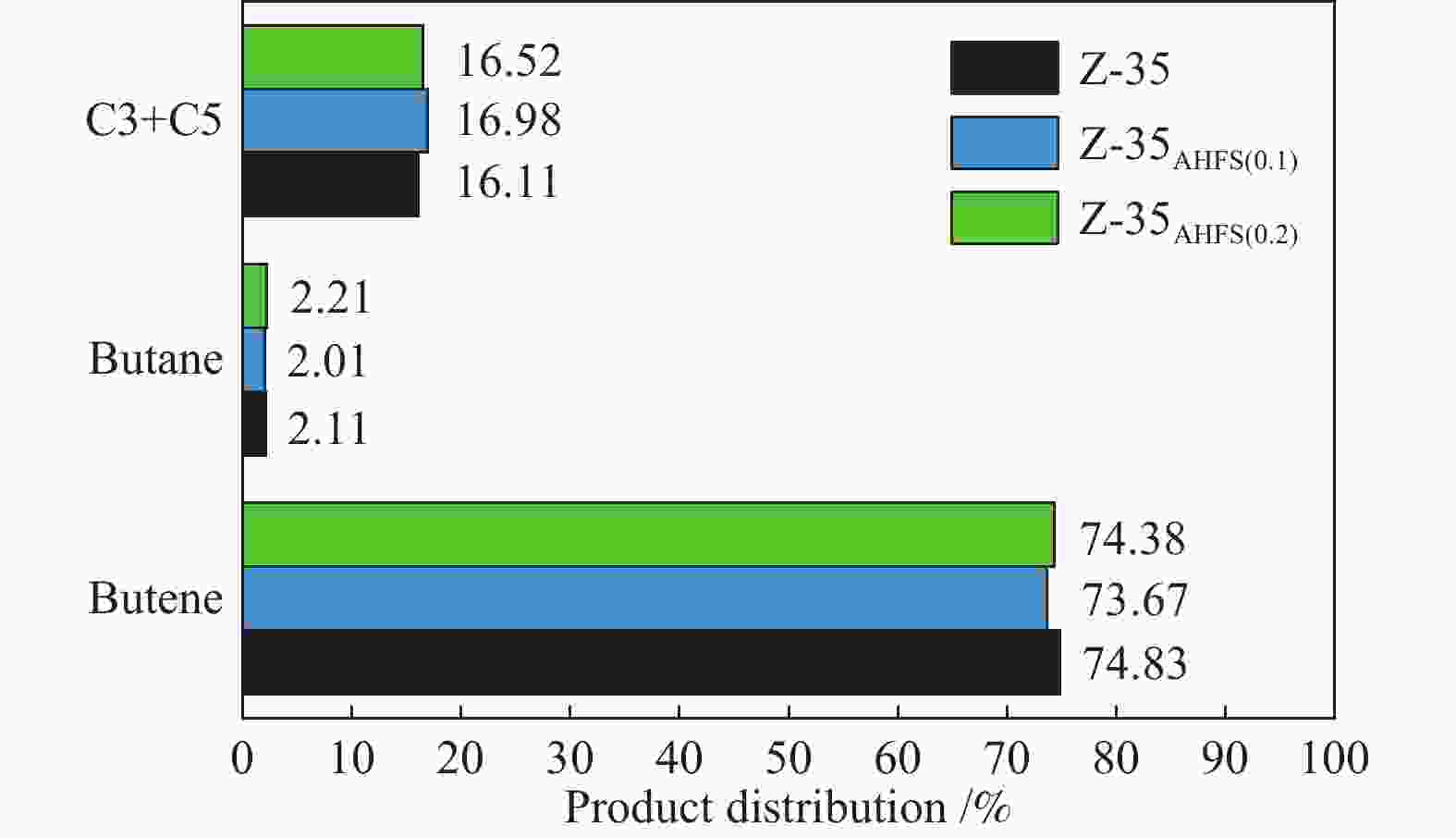
y/%300 15.67 73.71 11.55 29.66 74.58 22.12 325 31.79 70.62 22.45 44.35 68.10 30.34 350 49.99 64.97 32.48 58.28 55.34 32.25 375 60.12 53.06 31.90 68.96 43.53 30.02 410 67.67 40.74 27.57 81.00 27.00 21.87 reaction conditions: 0.1 MPa, WHSV = 6 h−1, reaction time: 8 h 表 2 两个反应的四种丁烯异构体产物分布
Table 2 Distribution of four butene isomer products for two reactions
Sample 1-butene
/%cis-2-
butene
/%tran-2-
butene
/%Isobutene
/%Thermodynamic equilibrium 8.34 14.39 23.64 53.67 Isobutene isomerization 7.77 14.72 21.12 56.40 n-butene isomerization 11.53 19.03 31.71 37.73 reaction conditions: 350 ℃, 0.1 MPa, WHSV = 6 h−1, reaction time: 8 h 表 3 不同浓度AHFS处理HZSM-35分子筛的织构性质
Table 3 Basic physical parameters of the parent and AHFS modified HZSM-35 zeolites
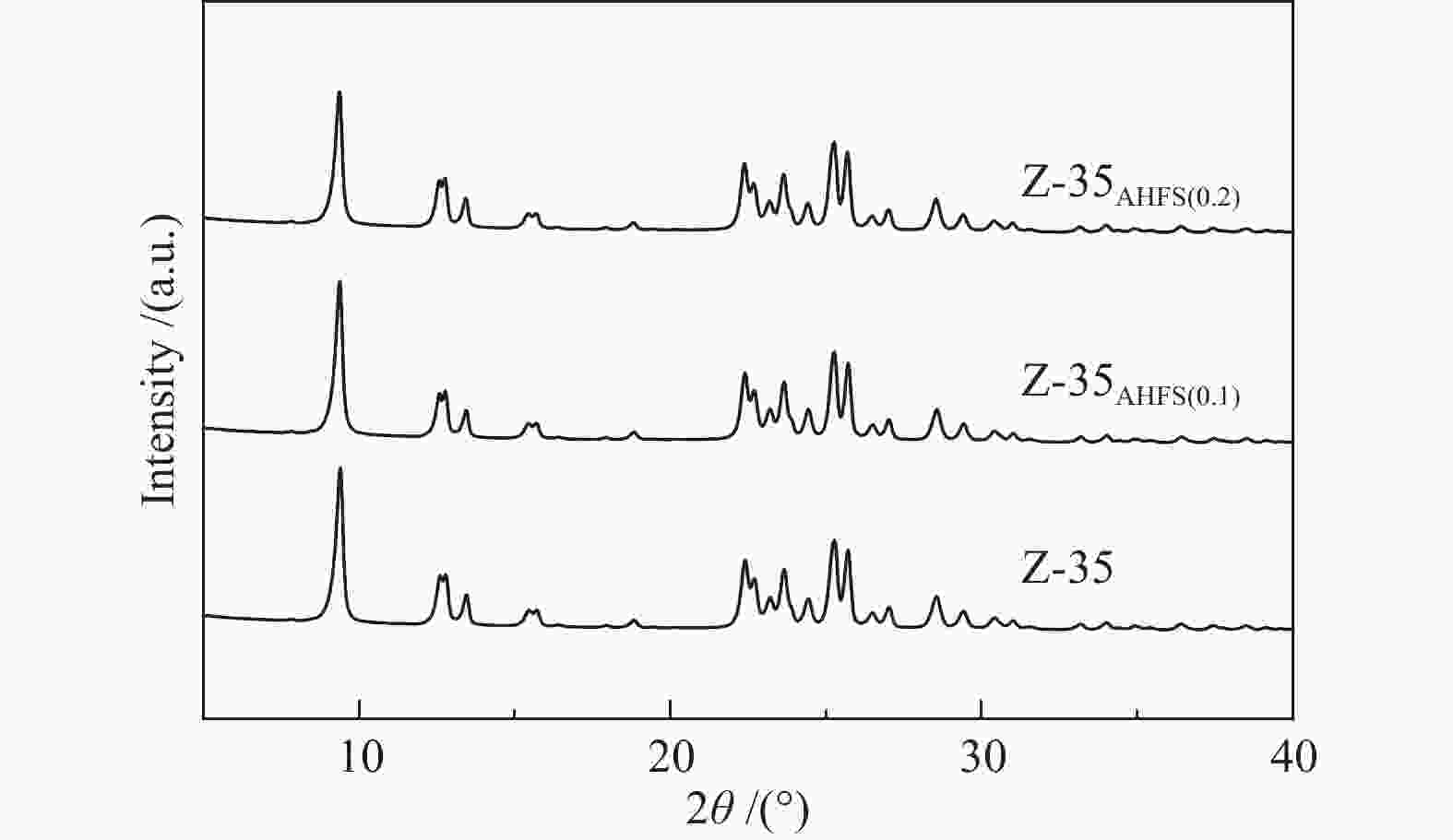
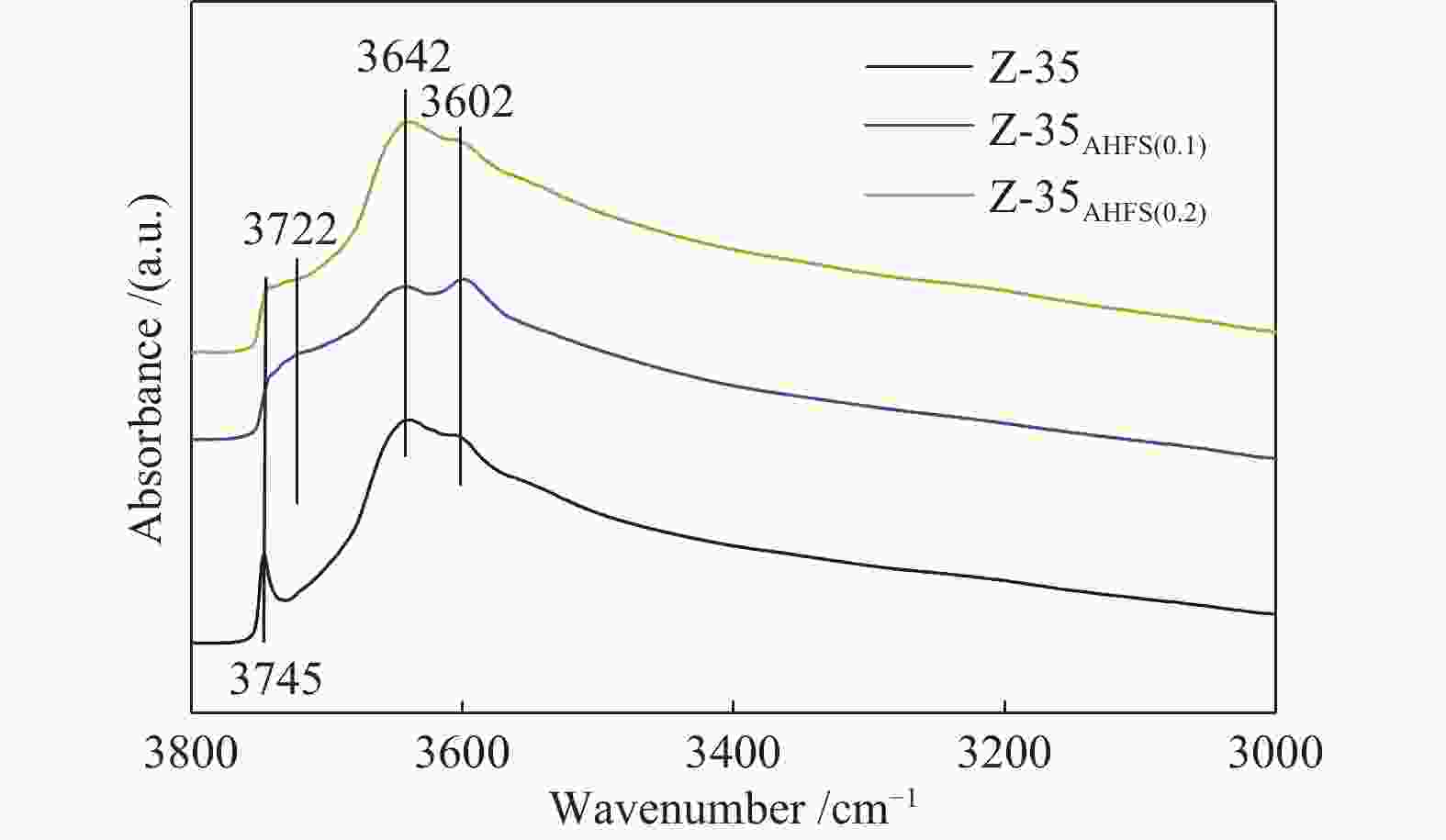
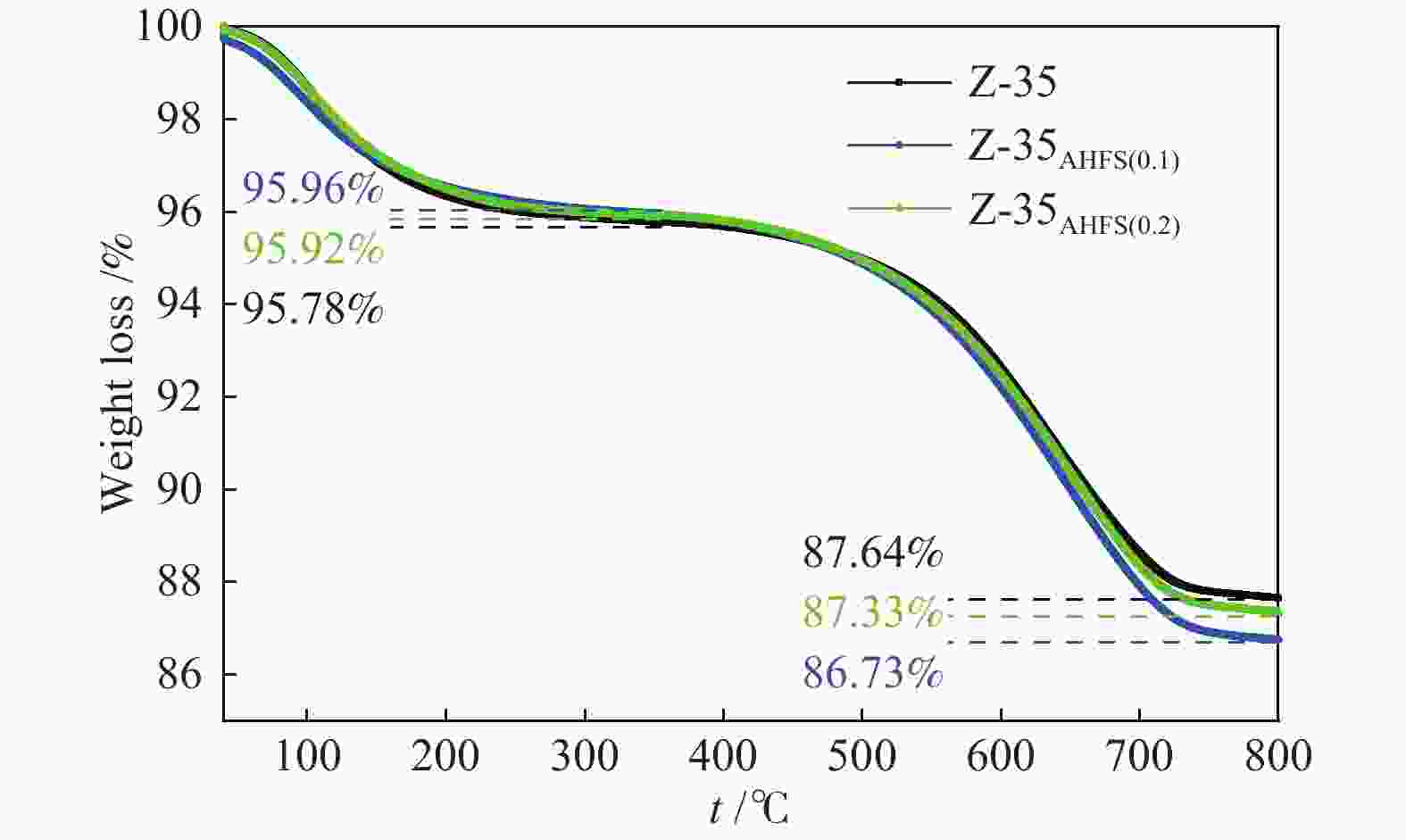
Sample SAR SBET/(m2∙g−1) Smicro/(m2∙g−1) vtotal/(cm3∙g−1) vmicro/(cm3∙g−1) (vtotal− vmicro)/(cm3∙g−1) Pore size/Å Z-35 8.91 298 245 0.318 0.129 0.189 5.216 Z-35AHSF(0.1) 11.80 282 228 0.292 0.120 0.172 5.278 Z-35AHSF(0.2) 12.35 288 223 0.363 0.117 0.246 5.670 -
[1] 温贤昭. 丁烯-1的应用及生产[J]. 石油化工,1982,10:688−692.WEN Xian-zhao. Application and production of 1-butene[J]. Petrochem Technol,1982,10:688−692. [2] FRIEDLANDER R H, WARD D J, OBENAUS F, NIERLICH F. Make plasticizer olefins via n-butene dimerization[J]. Hydrocarbon Process,1986,65(2):31−33. [3] YANG B, JIANG J G, XU H, LIU Y, PENG H, WU P. Selective skeletal isomerization of 1-butene over FER-type zeolites derived from PLS-3 lamellar precursors[J]. Appl Catal A: Gen,2013,455(30):107−113. [4] HOUZVLCKA J, HANDILDAAR S, PONEC V. The Shape Selectivity in the Skeletal Isornenisation of n-Butene to Isobutene[J]. J Catal,1997,167(1):273−278. doi: 10.1006/jcat.1997.1525 [5] DOMOKOS L, LEFFERT L, SESHAN K, LECHER J A. Isomerization of linear butenes to iso-butene over medium pore zeolites[J]. J Catal,2001,197(1):68−80. doi: 10.1006/jcat.2000.3056 [6] WANG Y N, GAO Y, CHU W F, ZHAO D P, CHEN F C, ZHU X X, LI X J, LIU K F, XIE S J, XU L Y. Synthesis and catalytic application of FER zeolite with controllable size[J]. J Mater Chem A,2019,7(13):7573−7580. doi: 10.1039/C8TA09420A [7] XU H, CHEN W, ZHANG G Q, WEI P F, WU Q M, ZHU L F, MENG X J, LI X J, FEI J H, HAN S C, ZHU Q Y, ZHENG A M, MA Y H, XIAO F S. Ultrathin nanosheets of aluminosilicate FER zeolites synthesized in the presence of a sole small organic ammonium[J]. J Mater Chem A,2019,7:16671−16676. doi: 10.1039/C9TA04833B [8] WEI P F, ZHU X X, WANG Y N, CHU W F, XIE S J, YANG Z Q, LIU X B, LI X J, XU L Y. Rapid synthesis of ferrierite zeolite through microwave assisted organic template free route[J]. Microporous Mesoporous Mater,2019,279:220−227. doi: 10.1016/j.micromeso.2018.12.036 [9] HU, Y, LIU, L, ZHANG, H, HU L, ZHANG C, ZHANG H. Effect of crystal size on the skeletal isomerization of n-butene over H-FER zeolite[J]. React Kinet Mech Catal,2014,112:241−248. doi: 10.1007/s11144-014-0684-4 [10] MERIAUDEAU P, BACAUD R, HUNG L N, VU A T. Isomerization of butene in isobutene on ferrierite catalyst: a mono-or bimolecular process[J]. J Mol Catal A: Gen,1996,110:177−179. doi: 10.1016/1381-1169(96)00156-2 [11] ZHENG X C, PONEC V. On the problems of the mechanism of the skeletal isomerization of n-butene[J]. Catal Lett,1994,27(1/2):113−117. doi: 10.1007/BF00806984 [12] GUISNETt M, ANDY P, BOUCHEFF Y, GNEP N S, BENAZZI E. Skeletal isomerization of n-butenes: I. Mechanism of n-butene transformation on a nondeactivated H-ferrierite catalyst[J]. J Catal,1996,158(2):551−560. doi: 10.1006/jcat.1996.0053 [13] HOUZVICKA J, PONEC V. Skeletal isomerization of n-butene[J]. Catal Rev,1997,39(4):319−344. doi: 10.1080/01614949708007099 [14] GON S, SOON H P. The reversible skeletal isomerization between n-butenes and iso-butene over solid acid catalysts[J]. Catal Today,1998,44(1):215−222. [15] JO D, HONG S B. Mechanisms of the reverse skeletal isomerization of n-butenes to isobutene over zeolite catalysts[J]. ChemCatChem,2016,9(1):114−116. [16] JO D, HONG S B, CAMBLOR M A. Monomolecular skeletal isomerization of 1-butene over selective zeolite catalysts[J]. ACS Catal,2015,5(4):2270−2274. doi: 10.1021/acscatal.5b00195 [17] GAO X, QIN Z, WANG B, ZHAO X, LI J, ZHAO H, LIU H, SHEN B. High silica REHY zeolite with low rare earth loading as high-performance catalyst for heavy oil conversion[J]. Appl Catal A: Gen,2012,413:254−260. [18] ALBEETY R A, GEHRIG C A. Standard chemical thermodynamic properties of alkane isomer groups[J]. J Phys Chem Ref Data,1985,14(3):803−820. doi: 10.1063/1.555737 [19] 汪哲明, 阎子峰. 丁烯异构化催化剂进展[J]. 石油化工,2002,31(4):311−315. doi: 10.3321/j.issn:1000-8144.2002.04.016WANG Zhe-ming, YAN Zi-feng. Progress of butene isomerization catalyst[J]. Pet Technol,2002,31(4):311−315. doi: 10.3321/j.issn:1000-8144.2002.04.016 [20] 柯明, 汪燮卿, 张凤美. 分子筛孔结构和硅铝比对催化裂化产品中乙烯选择性的影响[J]. 石油炼制与化工,2003,9:53−58. doi: 10.3969/j.issn.1005-2399.2003.09.013KE Ming, WANG Xie-qing, ZHANG Feng-mei. Effect of zeolite pore structure and silicon-aluminum ratio on the selectivity of ethylene in catalytic cracking products[J]. Pet Process Petrochem,2003,9:53−58. doi: 10.3969/j.issn.1005-2399.2003.09.013 [21] 赵明明. 正丁烯骨架异构催化剂ZSM-35成型及改性研究[D]. 北京: 北京化工大学, 2018.ZHAO Ming-ming. Research on forming and modification of the catalyst ZSM-35 for n-butene skeletal isomerization[D]. Beijing: Beijing University of Chemical Technology, 2018. [22] QIU Y, ZHAO G, LIU G, WANG L, ZHANG X. Catalytic cracking of supercritiel n-dodecane over wall-coated nano-Ag/HZSM-5 zeolites[J]. Ind Eng Chem Res,2014,53(47):18104−18111. doi: 10.1021/ie503335h [23] 周华东. 改性ZSM-35分子筛催化正丁烯骨架异构化的研究[D]. 北京: 中国石油大学(北京), 2016.ZHOU Hua-dong. Study on the skeletal isomerization of n-butene catalyzed by modified ZSM-35 zeolite[D]. Beijing: China University of Petroleum(Beijing), 2016. [24] 段欣瑞, 李孝国, 张永坤, 边凯, 张安峰, 侯章贵, 郭新闻. 纳米HZSM-5的酸性调控及其催化苯和稀乙烯制乙苯[J]. 石油学报(石油加工),2021,37(01):181−189.DUAN Xin-rui, LI Xiao-guo, ZHANG Yong-kun, BIAN kai, ZHANG An-feng, HOU Zhang-gui, GUO Xin-wen. Acid modification of nanosized HZSM-5 for alkylation of benzene with dilute ethylene[J]. Acta Pet Sin (Pet Process Sect),2021,37(01):181−189. [25] 王闻年, 袁德林, 李浩, 任申勇, 郭巧霞, 申宝剑. β沸石结构及其催化性能的调控[J]. 化工学报,2016,67(8):3429−3435.WANG Wen-nian, YUAN De-lin, LI Hao, REN Shen-yong, GUO Qiao-xia, SHEN Bao-jian. Regulation of structure and catalytic performance of β-zeolite by post treatments[J]. J Chem Ind Eng,2016,67(8):3429−3435. [26] LIU W, HU H Q, YANG L, ZHANG L, XIA C J, WANG Q, KE M. Distribution of effective ferrierite active sites for skeletal isomerization of n-butene to isobutene[J]. Chem Select,2019,4(27):7851−7857. [27] REALE E, LEYVA A, CORMA A, MARTINEZ C, GARCIA H, REY F. A fluoride-catalyzed sol-gel route to catalytically active non-ordered mesoporous silica materials in the absence of surfactants[J]. J Mater Chem,2005,15(17):1742−1754. doi: 10.1039/b415066j [28] KAO H M, CHEN Y C, TING C C, CHEN P T, JIANG J C. Characterization of extraframework aluminum in H-mordenite dealuminated with ammonium hexafluorosilicate[J]. Catal Today,2004,97(1):13−23. doi: 10.1016/j.cattod.2004.01.007 [29] ZUNULDA R F, CARLOS A Q, NORA S F, RAUL A C. Tungsten promoted ammonium and potassium ferrierite: deactivation during the skeletal isomerization of linear butenes[J]. Appl Catal A: Gen,2001,216(1-2):91−101. doi: 10.1016/S0926-860X(01)00545-2 [30] WANG Q L, GIANNETTO G, GUISNET M. Dealumination of zeolites III. Effect of extra-framework aluminum species on the activity, selectivity, and stability of Y zeolites in n-heptane cracking[J]. J Catal,1991,130(2):471−482. doi: 10.1016/0021-9517(91)90129-R [31] YOON J W, LEE J H, CHANG J S, CHOO D H, LEE S J, JHUNG S H. Trimerization of isobutene over zeolite catalysts: Remarkable performance over a ferrierite zeolite[J]. Catal Commun,2007,8(6):967−970. doi: 10.1016/j.catcom.2006.10.006 [32] GYRGY O. Comparison of butene skelctal isomerization selectivity over a pair of commercial H-ferrierites[J]. Micropor Mesopor Mat,2007,104(1/3):192−198. doi: 10.1016/j.micromeso.2007.02.004 [33] MUKHAMBETOV I N, EGOROVA S R, MUKHAMED A N, LAMBEROV A A. Hydrothermal modification of the alumina catalyst for the skeletal isomerization of n-butenes[J]. Appl Catal A: Gen,2018,554:64−70. doi: 10.1016/j.apcata.2018.01.024 [34] 郭忠森, 祖运, 惠宇, 秦玉才, 王焕, 张晓彤, 宋丽娟. 烯烃对噻吩在介孔分子筛Al-MCM-41活性位物种上吸附脱硫机制的影响[J]. 燃料化学学报,2019,47(4):474−483.GUO Zhong-sen, ZU Yun, HUI Yu, QIN Yu-cai, WANG Huan, ZHANG Xiao-tong, SONG Li-juan. Influence of olefin on the mechanism of thiophene adsorption on the active species of Al-MCM-41 mesoporous zeolites[J]. J Fuel Chem Technol,2019,47(4):474−483. [35] 祖运, 秦玉才, 高雄厚, 莫周胜, 张磊, 张晓彤, 宋丽娟. 催化裂化条件下噻吩与改性Y分子筛的作用机制[J]. 燃料化学学报,2015,43(7):862−869. doi: 10.3969/j.issn.0253-2409.2015.07.012ZU Yun, QIN Yu-cai, GAO Xiong-hou, MO Zhou-sheng, ZHANG Lei, ZHANG Xiao-tong, SONG Li-juan. Mechanisms of thiophene conversion over the modified Y zeolites under catalytic cracking conditions[J]. J Fuel Chem Technol,2015,43(7):862−869. doi: 10.3969/j.issn.0253-2409.2015.07.012 [36] ZHENG J, QIN Y C, LI Q, ZHANG L, GAO X H, SONG L J. A periodic DFT study of the synergistic mechanisms between extra-framework aluminum species and Brønsted acid sites in HY zeolites[J]. Ind Eng Chem Res,2020,59(7):2736−2744. doi: 10.1021/acs.iecr.9b05277 -




 下载:
下载: