Influence of Ni on the active phase and hydrodenitrogenation and hydrodesulfurization activities of MoS2 catalysts
-
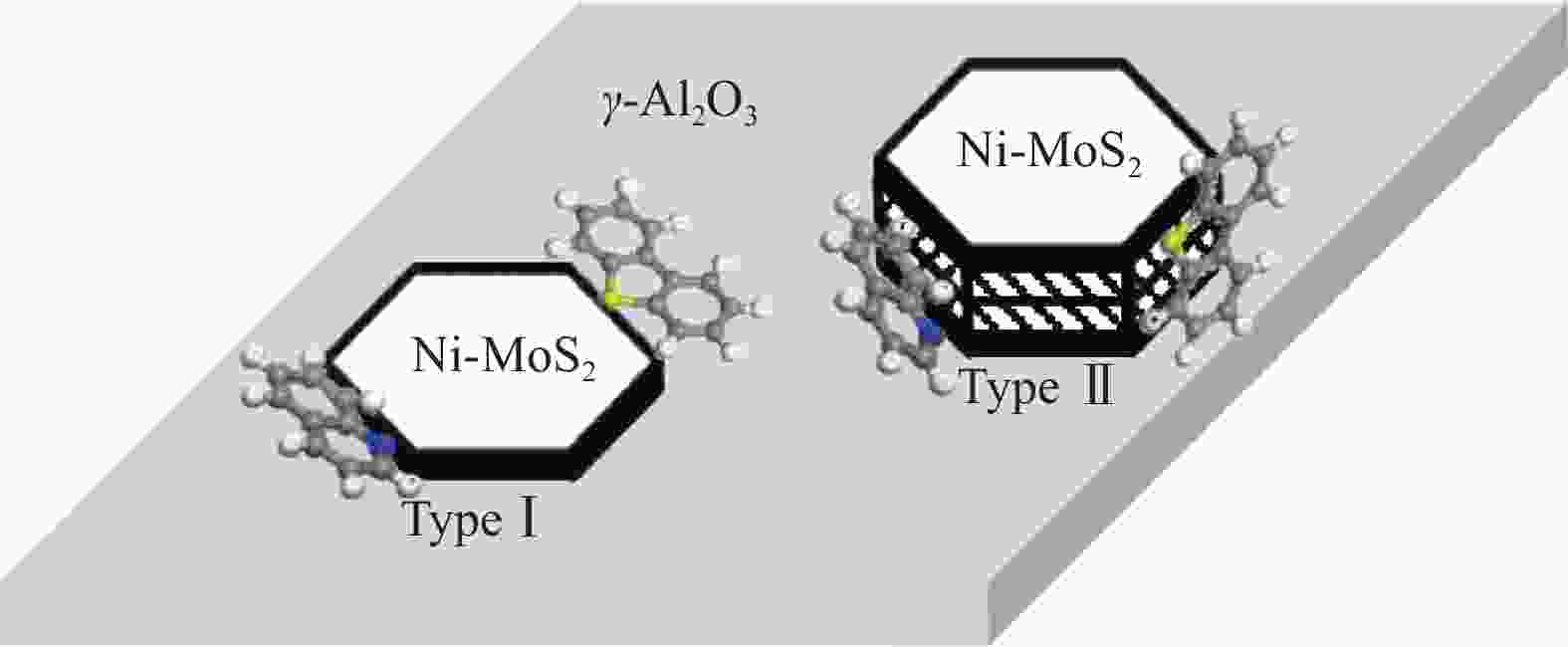
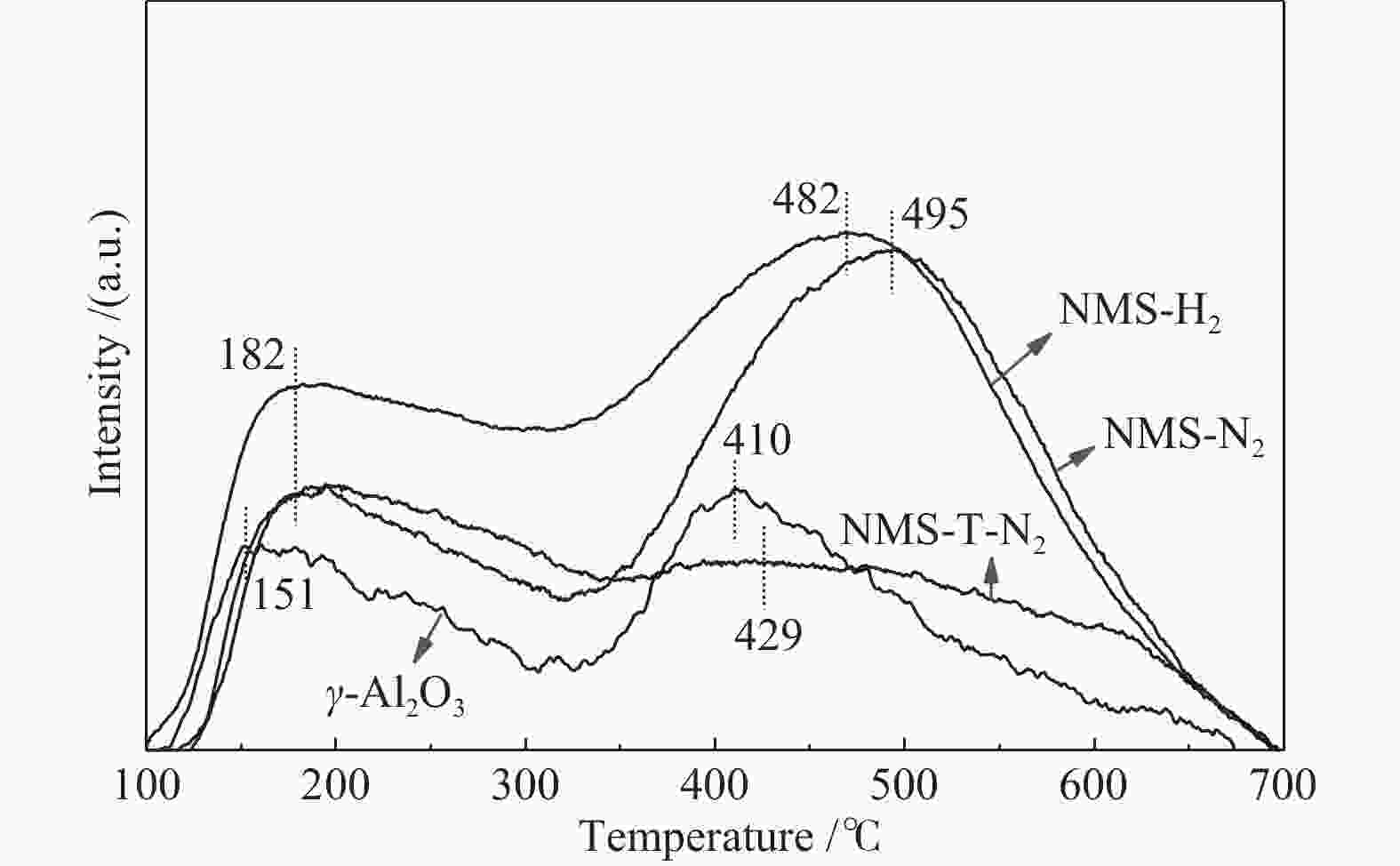
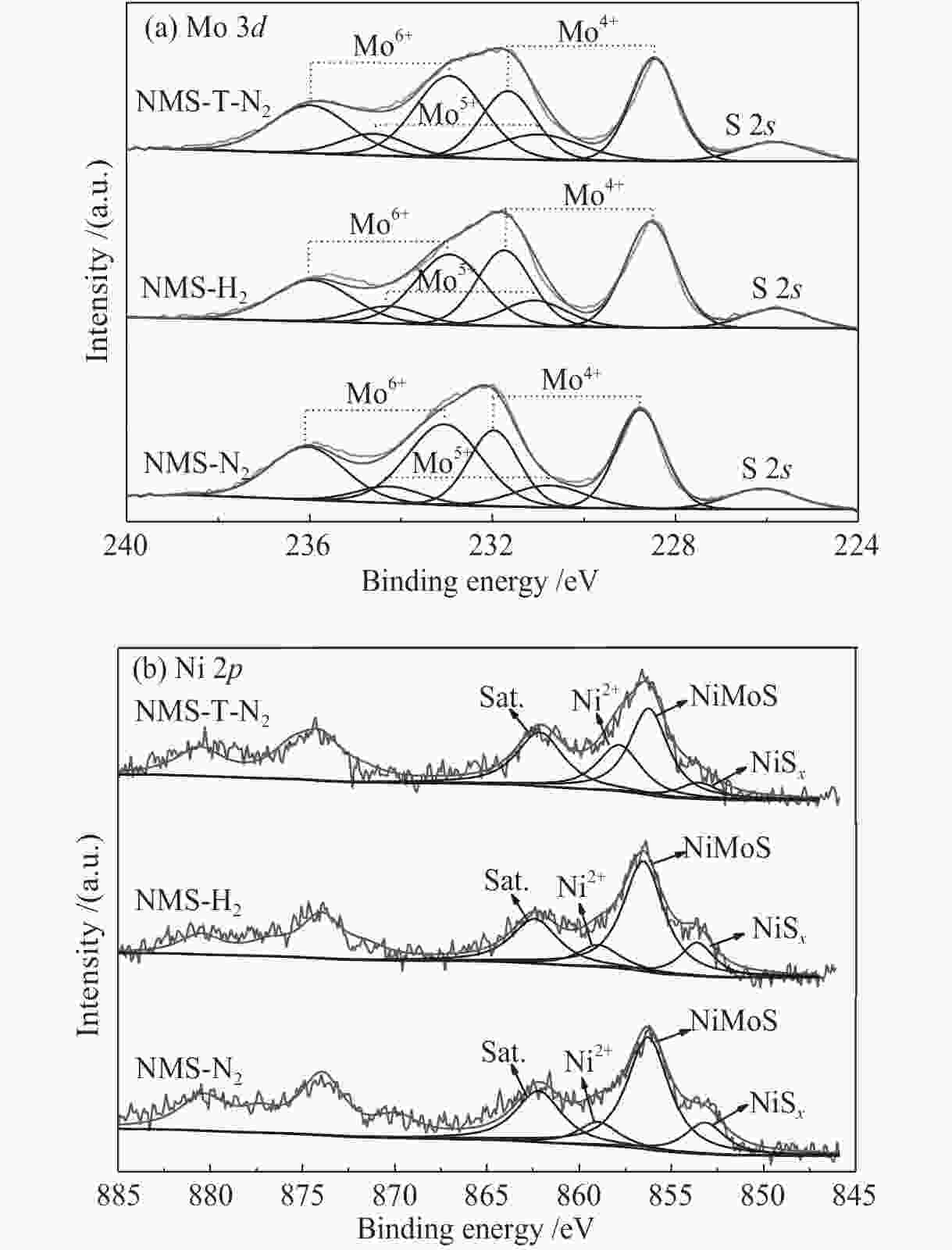
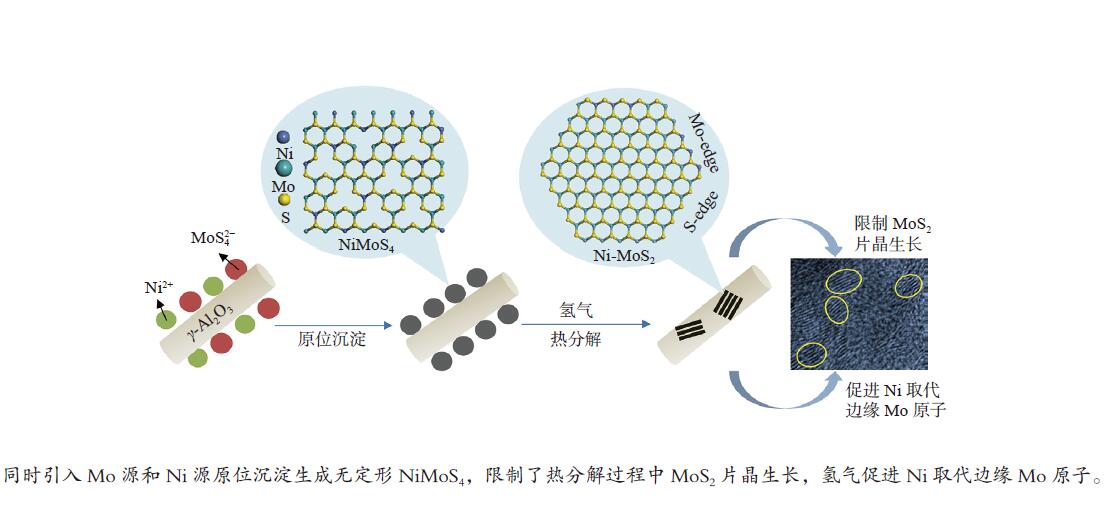
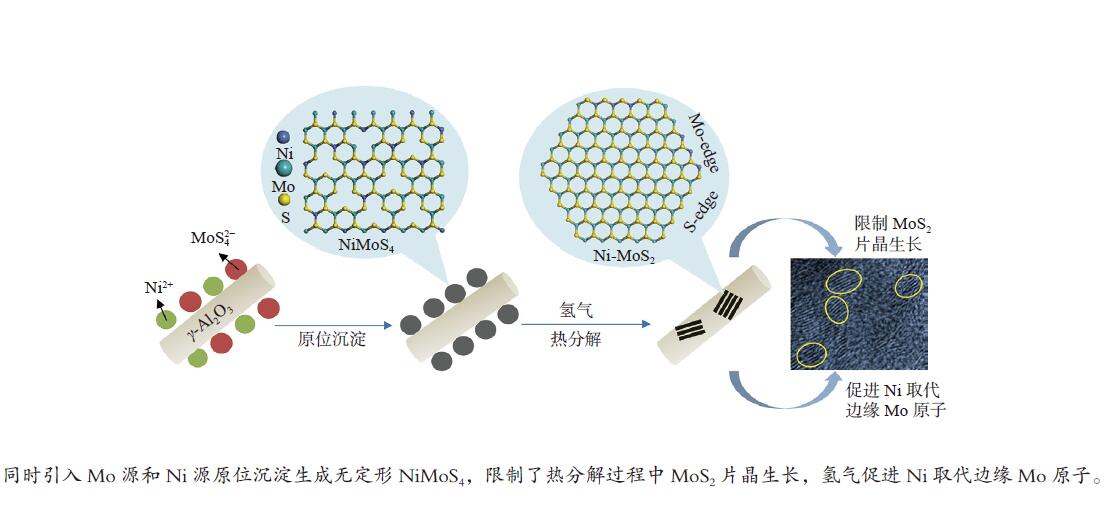
摘要: 为了获得较多高活性II型MoS2活性相,采用四硫代钼酸铵原位热分解法制备了MoS2基催化剂,对比分析了Ni源引入方式和热分解气氛对MoS2活性相微观结构、表面元素化学状态和加氢脱氮脱硫性能等的影响。结果表明,同时引入Mo源和Ni源原位沉淀生成无定形NiMoS4后,再热分解有利于Ni取代MoS2片晶边缘的Mo原子,被修饰后的MoS2片晶保持较高的分散度、适宜的长度(3−5 nm)和堆叠层数(2−4层),从而在边缘暴露较多具有加氢和氢解活性的rim和corner活性位点。热分解气氛H2比N2更有利于Ni在热分解过程中取代MoS2边缘的Mo原子,形成更多II型Ni-Mo-S活性结构,有利于喹啉和二苯并噻吩的吸附活化和加氢反应。当加氢反应温度340 ℃、氢压3 MPa、重时空速23.4 h−1、氢油比为600和使用0.1 g NMS-H2催化剂时,喹啉加氢脱氮转化率达23.8%,二苯并噻吩加氢脱硫转化率达93.3%。Abstract: To obtain type II active phase with higher activity, MoS2-based catalysts were prepared by thermal decomposition of ammonium tetrathiomolybdate. The influence of Ni adding way and decomposition atmosphere on the microstructures of MoS2 slabs, chemical state of surface elements, as well as hydrodesulfurization and hydrodenitrogenation activities were investigated. Results indicated that simultaneous impregnation of Mo and Ni precursors caused in situ deposition of amorphous NiMoS4 over the support surface, which subsequently facilitated the substitution of Mo atoms by Ni atoms at MoS2 edges. Accordingly, these decorated catalysts exhibited higher dispersion of MoS2 slabs with more suitable slab length (3–5 nm) and stacking number (2–4), which attributed to larger numbers of rim and corner active sites exposed at the edges. These active sites were essential in hydrogenation and hydrogenolysis reactions. In comparison with N2 atmosphere, thermal decomposition in H2 atmosphere was more conducive to the substitution of Mo atoms by Ni atoms at MoS2 edges, which provided more active Ni-Mo-S structures for the adsorption, activation and hydrogenolysis of quinoline and dibenzothiophene molecules. The catalyst prepared by thermal decomposition of NiMoS4 in H2 atmosphere showed superior activities in the quinoline hydrodenitrogenation with 23.8% conversion and in the dibenzothiophene hydrodesulfurization with 93.3% conversion, under the conditions of 340 °C, 3 MPa, a weight hourly space velocity of 23.4 h–1, H2/oil volume ratio of 600 and 0.1 g of NMS-H2 catalysts.
-
Key words:
- NiMo sulfide /
- quinoline /
- dibenzothiophene /
- hydrogenation reaction
-
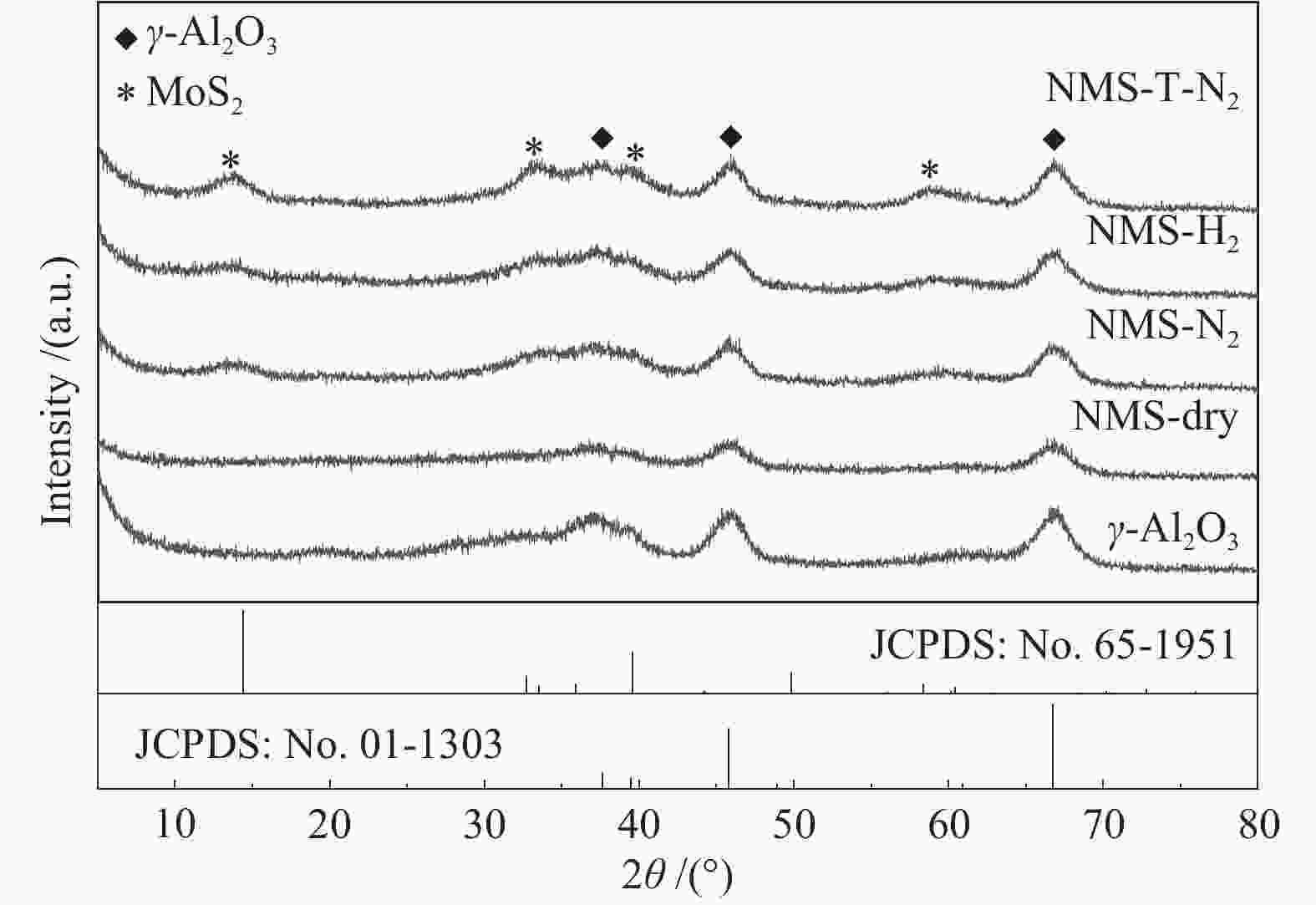
表 1 样品的物理结构性质
Table 1 Physical structure properties of sample
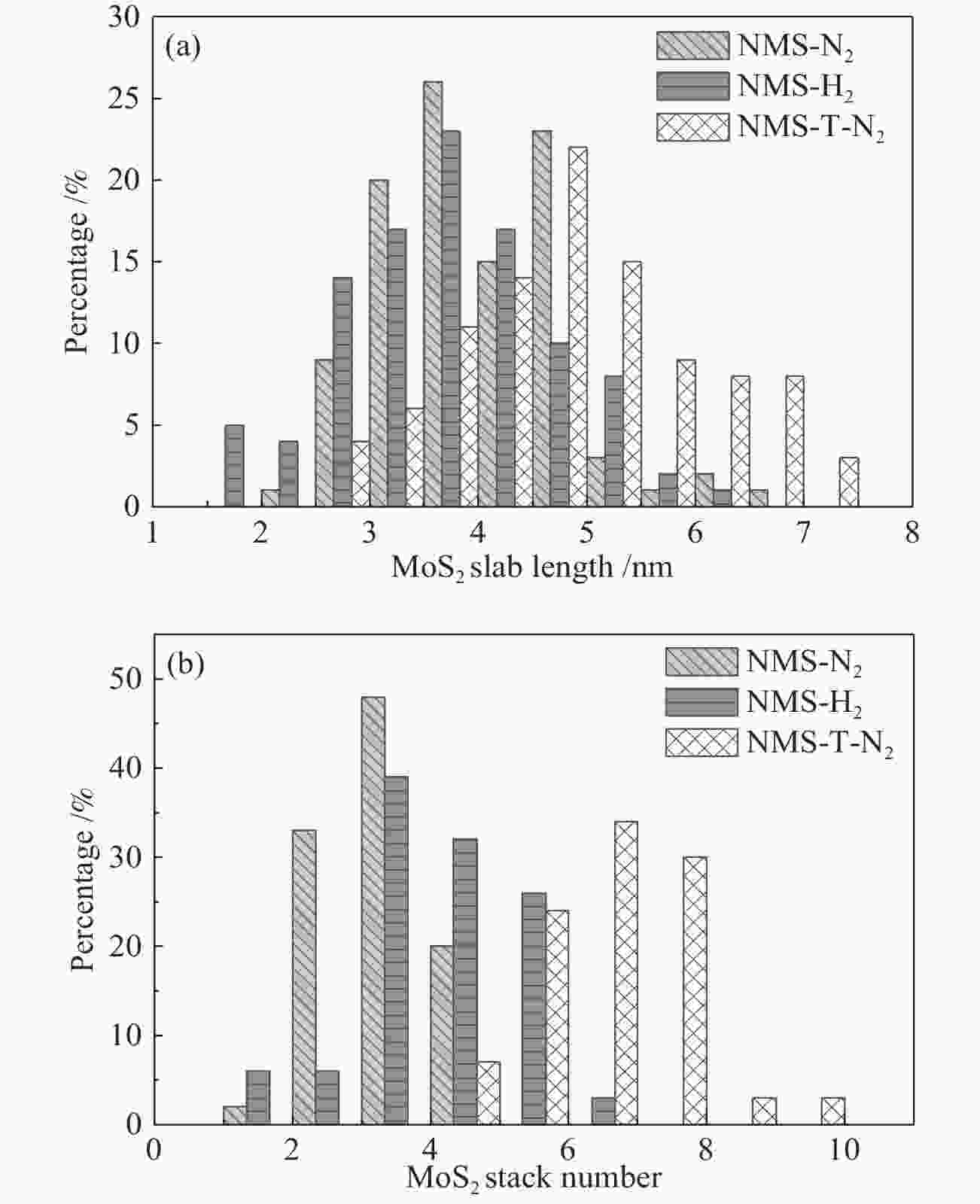
Sample Specific surface area/(m2·g−1) Total pore volume/(cm3·g−1) Most probable pore size/nm γ-Al2O3 288 1.03 14.3 NMS-N2 204 0.57 11.3 NMS-H2 206 0.60 11.7 NMS-T-N2 198 0.62 12.6 表 2 MoS2片晶平均长度LA、平均堆叠层数NA、分散度fMo和不同活性位点的比例
Table 2 Average length LA, stack number NA, Mo atoms dispersion and fraction of different active sites
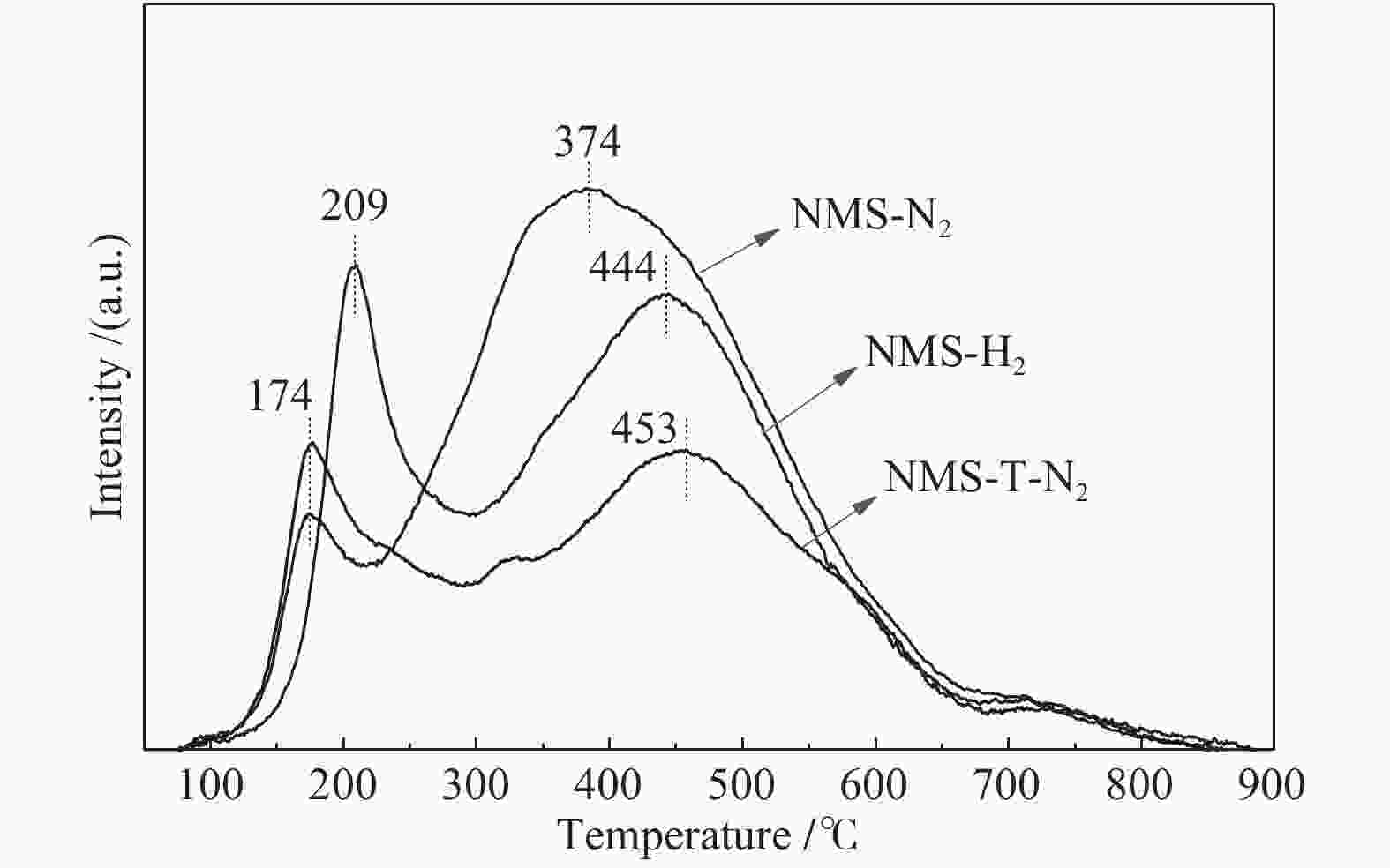
Catalyst LA/nm NA fMo R fe fr fc NMS-N2 3.9 4.2 0.28 1.09 0.248 0.228 0.059 NMS-H2 3.7 3.4 0.29 0.58 0.257 0.443 0.073 NMS-T-N2 4.9 6.6 0.23 1.33 0.213 0.160 0.038 表 3 Mo 3d和Ni 2p信号的分峰拟合
Table 3 Deconvolution results of Mo 3d and Ni 2p signals
Sample Mosulf/
%Mo distribution/
%Nisulf/
%Ni distribution/
%Mo4+ Mo5+ Mo6+ NiSx NiMoS Ni2+ NMS-N2 40 40 13 47 71 15 56 29 NMS-H2 47 47 14 39 87 16 71 13 NMS-T-N2 39 39 17 44 66 11 55 34 表 4 不同催化剂的喹啉HDN性能和选择性
Table 4 Quinoline HDN activity and selectivity over different catalysts
Catalyst xHDN/% Product selectivity/% Route ratio PCH PCHE PB DHQ 58THQ OPA 14THQ (PCH + PCHE)/PB NMS-N2 12.8 9.7 1.4 3.8 9.6 25.7 3.5 46.2 2.92 NMS-H2 23.8 17.8 2.2 6.5 10.3 24.9 4.7 33.4 3.07 NMS-T-N2 8.6 6.3 1.3 2.9 8.2 20.4 3.3 57.5 2.62 表 5 不同催化剂的二苯并噻吩HDS性能和选择性
Table 5 Dibenzothiophene HDS activity and selectivity over different catalyst
Catalyst xHDS/% Product selectivity/% Route ratio BP CHB BCH BP/(CHB + BCH) NMS-N2 91.6 74.8 24.0 1.2 2.96 NMS-H2 93.3 65.3 30.8 3.9 1.88 NMS-T-N2 81.9 80.5 19.1 0.4 4.12 -
[1] 刘敏, 陈贵锋, 王永刚, 赵鹏, 曲思建. 白石湖煤液化粗油加氢精制过程硫、氮化合物转化规律[J]. 燃料化学学报,2019,47(7):870−875. doi: 10.3969/j.issn.0253-2409.2019.07.012LIU Min, CHEN Gui-feng, WANG Yong-gang, ZHAO Peng, QU Si-jian. Conversion of sulphur and nitrogen compounds in hydrofining process of Baishihu coal liquefaction oil[J]. J Fuel Chem Technol,2019,47(7):870−875. doi: 10.3969/j.issn.0253-2409.2019.07.012 [2] VÁZQUEZ-GARRIDO I, LÓPEZ-BENÍTEZ A, BERHAULT G, GUEVARA-LARA A. Effect of support on the acidity of NiMo/Al2O3-MgO and NiMo/TiO2-Al2O3 catalysts and on the resulting competitive hydrodesulfurization/hydrodenitrogenation reactions[J]. Fuel,2019,236:55−64. doi: 10.1016/j.fuel.2018.08.053 [3] LIU Z, HAN W, HU D, SUN S, HU A, WANG Z, JIA Y, ZHAO X, YANG Q. Effects of Ni-Al2O3 interaction on NiMo/Al2O3 hydrodesulfurization catalysts[J]. J Catal,2020,387:62−72. doi: 10.1016/j.jcat.2020.04.008 [4] EIJSBOUTS S, LI X, JUAN-ALCANIZ J, VAN DEN OETELAAR L C A, BERGWERFF J A, LOOS J, CARLSSON A, VOGT E T C. Electron tomography reveals the active phase-support interaction in sulfidic hydroprocessing catalysts[J]. ACS Catal,2017,7(7):4817−4821. doi: 10.1021/acscatal.7b01201 [5] GUTIÉRREZ O Y, KLIMOVA T. Effect of the support on the high activity of the (Ni)Mo/ZrO2-SBA-15 catalyst in the simultaneous hydrodesulfurization of DBT and 4, 6-DMDBT[J]. J Catal,2011,281(1):50−62. doi: 10.1016/j.jcat.2011.04.001 [6] GE H, WEN X-D, RAMOS M A, CHIANELLI R R, WANG S, WANG J, QIN Z, LYU Z, LI X. Carbonization of ethylenediamine coimpregnated CoMo/Al2O3 catalysts sulfided by organic sulfiding agent[J]. ACS Catal,2014,4(8):2556−2565. doi: 10.1021/cs500477x [7] BARA C, LAMIC-HUMBLOT A-F, FONDA E, GAY A-S, TALEB A-L, DEVERS E, DIGNE M, PIRNGRUBER G D, CARRIER X. Surface-dependent sulfidation and orientation of MoS2 slabs on alumina-supported model hydrodesulfurization catalysts[J]. J Catal,2016,344:591−605. doi: 10.1016/j.jcat.2016.10.001 [8] BOUWENS S M A M, VANZON F B M, VANDIJK M P, VANDERKRAAN A M, DEBEER V H J, VANVEEN J A R, KONINGSBERGER D C. On the structural differences between alumina-supported CoMoS type I and alumina-, silica-, and carbon-supported CoMoS type II phases studied by XAFS, MES, and XPS[J]. J Catal,1994,146(2):375−393. doi: 10.1006/jcat.1994.1076 [9] OKAMOTO Y, KATO A, USMAN, RINALDI N, FUJIKAWA T, KOSHIKA H, HIROMITSU I, KUBOTA T. Effect of sulfidation temperature on the intrinsic activity of Co-MoS2 and Co-WS2 hydrodesulfurization catalysts[J]. J Catal,2009,265(2):216−228. doi: 10.1016/j.jcat.2009.05.003 [10] SÁNCHEZ J, MORENO A, MONDRAGÓN F, SMITH K J. Morphological and structural properties of MoS2 and MoS2-amorphous silica-alumina dispersed catalysts for slurry-phase hydroconversion[J]. Energy Fuels,2018,32(6):7066−7077. doi: 10.1021/acs.energyfuels.8b01081 [11] MELLO M D D, BRAGGIO F D A, MAGALHÃES B D C, ZOTIN J L, SILVA M A P D. Effects of phosphorus content on simultaneous ultradeep HDS and HDN reactions over NiMoP/alumina catalysts[J]. Ind Eng Chem Res,2017,56(37):10287−10299. doi: 10.1021/acs.iecr.7b02718 [12] BADOGA S, DALAI A K, ADJAYE J, HU Y. Insights into individual and combined effects of phosphorus and EDTA on performance of NiMo/MesoAl2O3 catalyst for hydrotreating of heavy gas oil[J]. Fuel Process Technol,2017,159:232−246. doi: 10.1016/j.fuproc.2017.01.034 [13] 王广建, 赵强, 陈国良, 王建爽, 石林. 柠檬酸引入方式对CoMo/TiO2-Al2O3催化剂加氢脱硫性能的影响[J]. 工业催化,2019,27(7):54−60. doi: 10.3969/j.issn.1008-1143.2019.07.010WANG Guang-jian, ZHAO Qiang, CHEN Guo-liang, WANG Jian-shuang, SHI Lin. Effect of citric acid introduction methods on hydrodesulfurization performance of CoMo/TiO2-Al2O3 catalysts[J]. Ind Catal,2019,27(7):54−60. doi: 10.3969/j.issn.1008-1143.2019.07.010 [14] SOLNICKOVA L. Nano-sized carbon-supported molybdenum disulphide particles for hydrodesulphurization[D]. Vancouver: The University of British Columbia, 2016. [15] GANIYU S A, ALHOOSHANI K, ALI S A. Single-pot synthesis of Ti-SBA-15-NiMo hydrodesulfurization catalysts: Role of calcination temperature on dispersion and activity[J]. Appl Catal B: Environ,2017,203:428−441. doi: 10.1016/j.apcatb.2016.10.052 [16] VARAKIN A N, MOZHAEV A V, PIMERZIN A A, NIKULSHIN P A. Comparable investigation of unsupported MoS2 hydrodesulfurization catalysts prepared by different techniques: Advantages of support leaching method[J]. Appl Catal B: Environ,2018,238:498−508. doi: 10.1016/j.apcatb.2018.04.003 [17] NIEFIND F, BENSCH W, DENG M, KIENLE L, CRUZ-REYES J, GRANADOS J M D V. Co-promoted MoS2 for hydrodesulfurization: New preparation method of MoS2 at room temperature and observation of massive differences of the selectivity depending on the activation atmosphere[J]. Appl Catal A: Gen,2015,497:72−84. doi: 10.1016/j.apcata.2015.03.003 [18] SUN K, GUO H, JIAO F, CHAI Y, LI Y, LIU B, MINTOVA S, LIU C. Design of an intercalated Nano-MoS2 hydrophobic catalyst with high rim sites to improve the hydrogenation selectivity in hydrodesulfurization reaction[J]. Appl Catal B: Environ,2021,286:119907. doi: 10.1016/j.apcatb.2021.119907 [19] LIU B, LIU L, CHAI Y, ZHAO J, LI Y, LIU D, LIU Y, LIU C. Effect of sulfiding conditions on the hydrodesulfurization performance of the ex-situ presulfided CoMoS/γ-Al2O3 catalysts[J]. Fuel,2018,234:1144−1153. doi: 10.1016/j.fuel.2018.08.001 [20] LAI W, CHEN Z, ZHU J, YANG L, ZHENG J, YI X, FANG W. NiMoS flower-like structure with self-assembled nanosheets as high-performance hydrodesulfurization catalysts[J]. Nanoscale,2016,8(6):3823−3833. doi: 10.1039/C5NR08841K [21] PELARDY F, SANTOS A S, DAUDIN A, DEVERS E, BELIN T, BRUNET S. Sensitivity of supported MoS2-based catalysts to carbon monoxide for selective HDS of FCC gasoline: Effect of nickel or cobalt as promoter[J]. Appl Catal B: Environ,2017,206:24−34. doi: 10.1016/j.apcatb.2016.12.057 [22] BREMMER G M, HAANDEL L, HENSEN E J M, FRENKEN J W M, KOOYMAN P J. The effect of oxidation and resulfidation on (Ni/Co)MoS2 hydrodesulfurisation catalysts[J]. Appl Catal B: Environ,2019,243:145−150. doi: 10.1016/j.apcatb.2018.10.014 [23] RANGARAJAN S, MAVRIKAKIS M. On the preferred active sites of promoted MoS2 for hydrodesulfurization with minimal organonitrogen inhibition[J]. ACS Catal,2017,7(1):501−509. doi: 10.1021/acscatal.6b02735 [24] BERHAULT G, MEHTA A, PAVEL A C, YANG J, RENDON L, YÁCAMAN M J, ARAIZA L C, MOLLER A D, CHIANELLI R R. The role of structural carbon in transition metal sulfides hydrotreating catalysts[J]. J Catal,2001,498:9−19. [25] LIU J, LI W-Y, FENG J, GAO X, LUO Z-Y. Promotional effect of TiO2 on quinoline hydrodenitrogenation activity over Pt/γ-Al2O3 catalysts[J]. Chem Eng Sci,2019,207:1085−1095. doi: 10.1016/j.ces.2019.07.040 [26] LIU J, LI W-Y, FENG J, GAO X. Effects of Fe species on promoting the dibenzothiophene hydrodesulfurization over the Pt/γ-Al2O3 catalysts[J]. Catal Today,2020,. doi: 10.1016/j.cattod.2020.07.035 [27] DAAGE M, CHIANELLI R R. Structure-function relations in molybdenum sulfide catalysts: The "Rim-Edge" model[J]. J Catal,1994,149(2):414−427. doi: 10.1006/jcat.1994.1308 [28] KASZTELAN S, TOULHOAT H, GRIMBLOT J, BONNELLE J P. A geometrical model of the active phase of hydrotreating catalysts[J]. Appl Catal,1984,13(1):127−159. doi: 10.1016/S0166-9834(00)83333-3 [29] XU J, GUO Y, HUANG T, FAN Y. Hexamethonium bromide-assisted synthesis of CoMo/graphene catalysts for selective hydrodesulfurization[J]. Appl Catal B: Environ,2019,244:385−395. doi: 10.1016/j.apcatb.2018.11.065 [30] ZHOU W, ZHANG Q, ZHOU Y, WEI Q, DU L, DING S, JIANG S, ZHANG Y. Effects of Ga- and P-modified USY-based NiMoS catalysts on ultra-deep hydrodesulfurization for FCC diesels[J]. Catal Today,2018,305:171−181. doi: 10.1016/j.cattod.2017.07.006 [31] JIAO J, FU J, WEI Y, ZHAO Z, DUAN A, XU C, LI J, SONG H, ZHENG P, WANG X, YANG Y, LIU Y. Al-modified dendritic mesoporous silica nanospheres-supported NiMo catalysts for the hydrodesulfurization of dibenzothiophene: Efficient accessibility of active sites and suitable metal-support interaction[J]. J Catal,2017,356:269−282. doi: 10.1016/j.jcat.2017.10.003 [32] HU C, CREASER D, FOULADVAND S, GRÖNBECK H, SKOGLUNDH M. Methyl crotonate hydrogenation over Pt: Effects of support and metal dispersion[J]. Appl Catal A: Gen,2016,511:106−116. doi: 10.1016/j.apcata.2015.12.003 [33] ALBERSBERGER S, SHI H, WAGENHOFER M, HAN J, GUTIÉRREZ O Y, LERCHER J A. On the enhanced catalytic activity of acid-treated, trimetallic Ni-Mo-W sulfides for quinoline hydrodenitrogenation[J]. J Catal,2019,380:332−342. doi: 10.1016/j.jcat.2019.09.034 [34] NGUYEN M-T, TAYAKOUT-FAYOLLE M, CHAINET F, PIRNGRUBER G D, GEANTET C. Use of kinetic modeling for investigating support acidity effects of NiMo sulfide catalysts on quinoline hydrodenitrogenation[J]. Appl Catal A: Gen,2017,530:132−144. doi: 10.1016/j.apcata.2016.11.015 [35] WANG H, LIU S, SMITH K J. Understanding selectivity changes during hydrodesulfurization of dibenzothiophene on Mo2C/carbon catalysts[J]. J Catal,2019,369:427−439. doi: 10.1016/j.jcat.2018.11.035 [36] RYDBERG H, DION M, JACOBSON N, SCHRÖDER E, HYLDGAARD P, SIMAK S, LANGRETH D, LUNDQVIST B. Van der waals density functional for layered structures[J]. Phys Rev Lett,2003,91(12):126402. doi: 10.1103/PhysRevLett.91.126402 [37] BYSKOV L, NØRSKOV J, CLAUSEN B, TOPSØE H. DFT calculations of unpromoted and promoted MoS2-based hydrodesulfurization catalysts[J]. J Catal,1999,187(1):109−122. doi: 10.1006/jcat.1999.2598 [38] ZHENG P, LI T, CHI K, XIAO C, FAN J, WANG X, DUAN A. DFT insights into the formation of sulfur vacancies over corner/edge site of Co/Ni-promoted MoS2 and WS2 under the hydrodesulfurization conditions[J]. Appl Catal B: Environ,2019,257:117937. doi: 10.1016/j.apcatb.2019.117937 [39] GUTIÉRREZ O, HRABAR A, HEIN J, YU Y, HAN J, LERCHER J. Ring opening of 1, 2, 3, 4-tetrahydroquinoline and decahydroquinoline on MoS2/γ-Al2O3 and Ni-MoS2/γ-Al2O3[J]. J Catal,2012,295:155−168. doi: 10.1016/j.jcat.2012.08.003 -




 下载:
下载: