Effect of Zr-TiO2 catalyst on NO emission from coal-burning and its catalytic mechanism
-
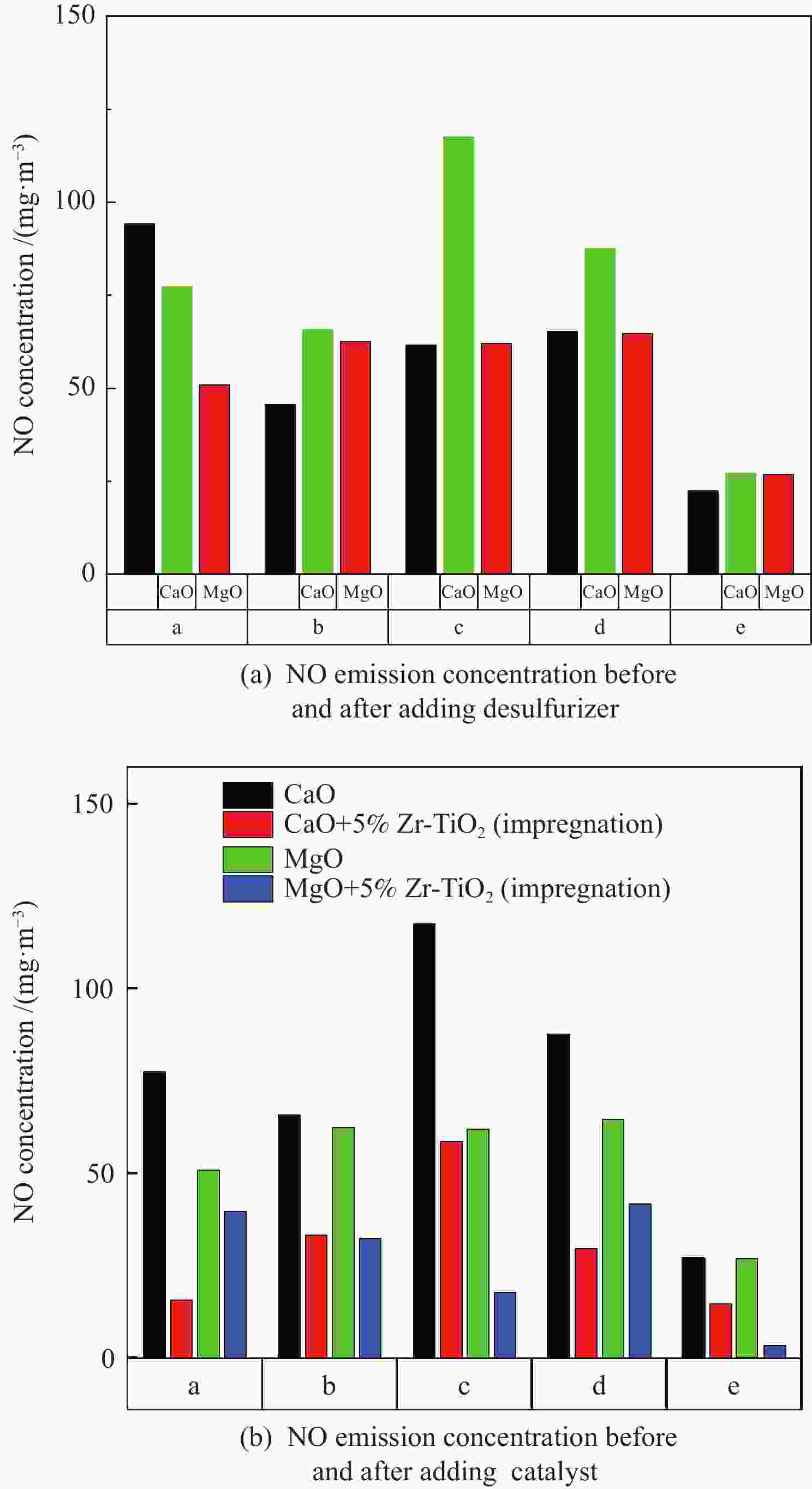
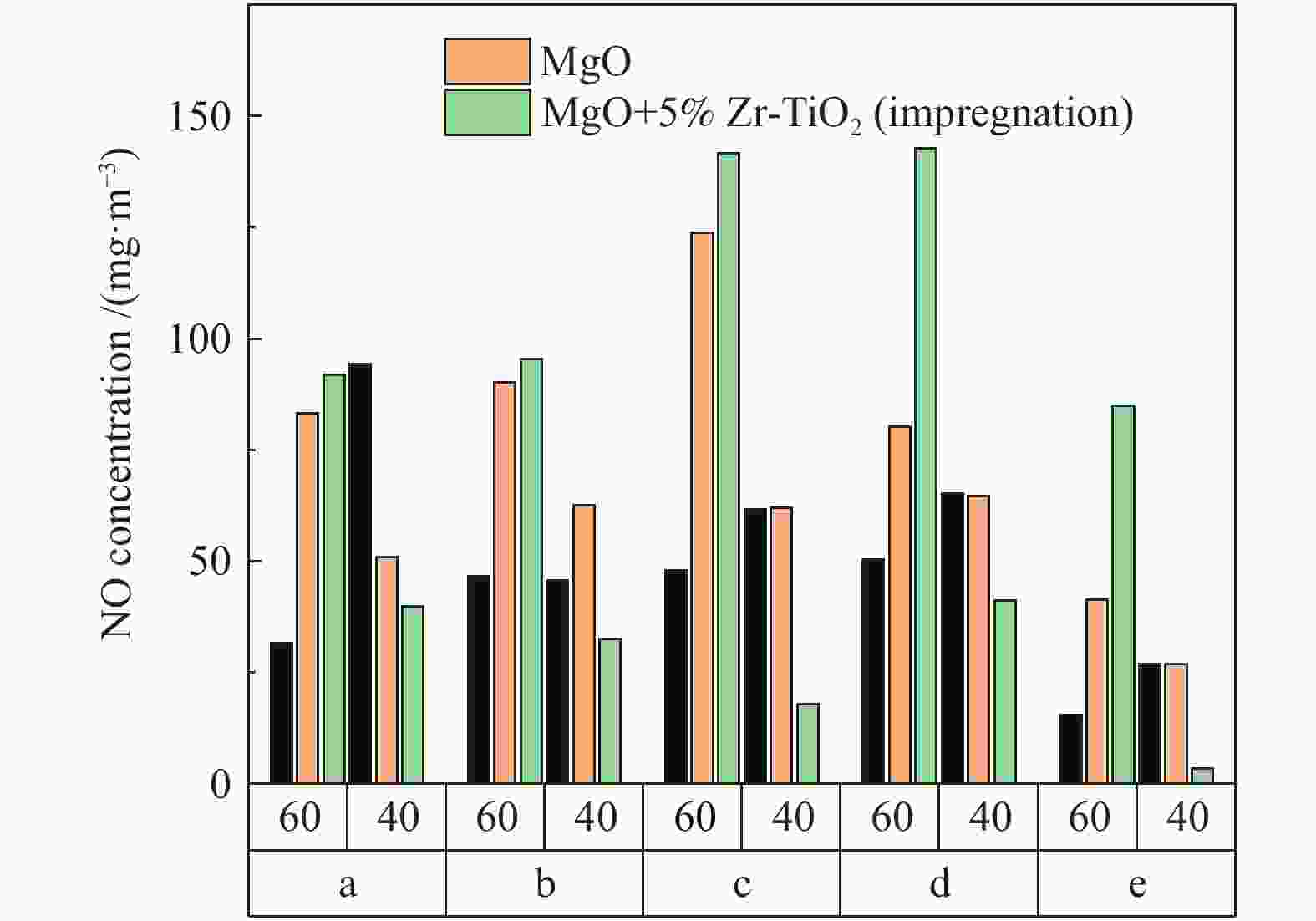
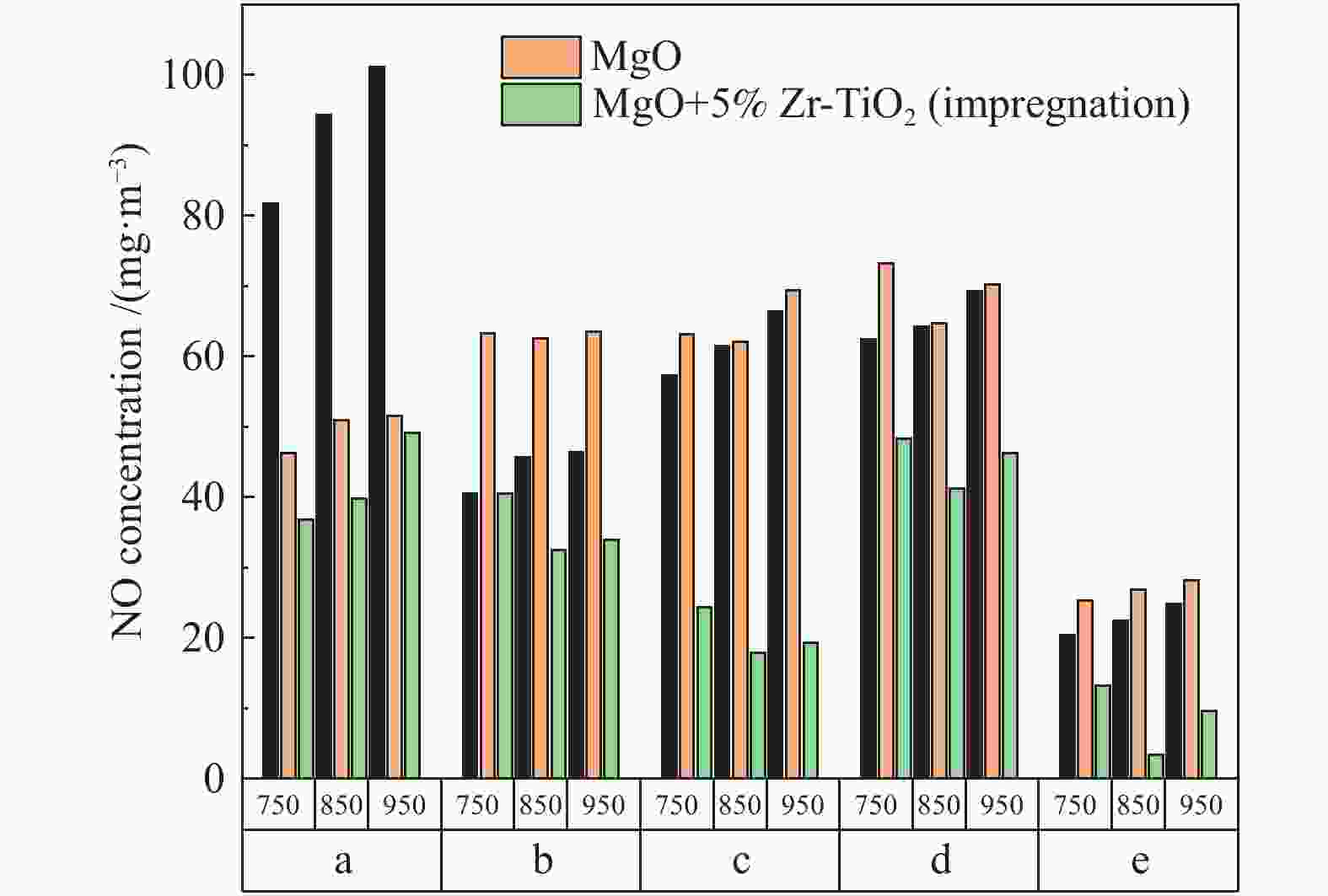
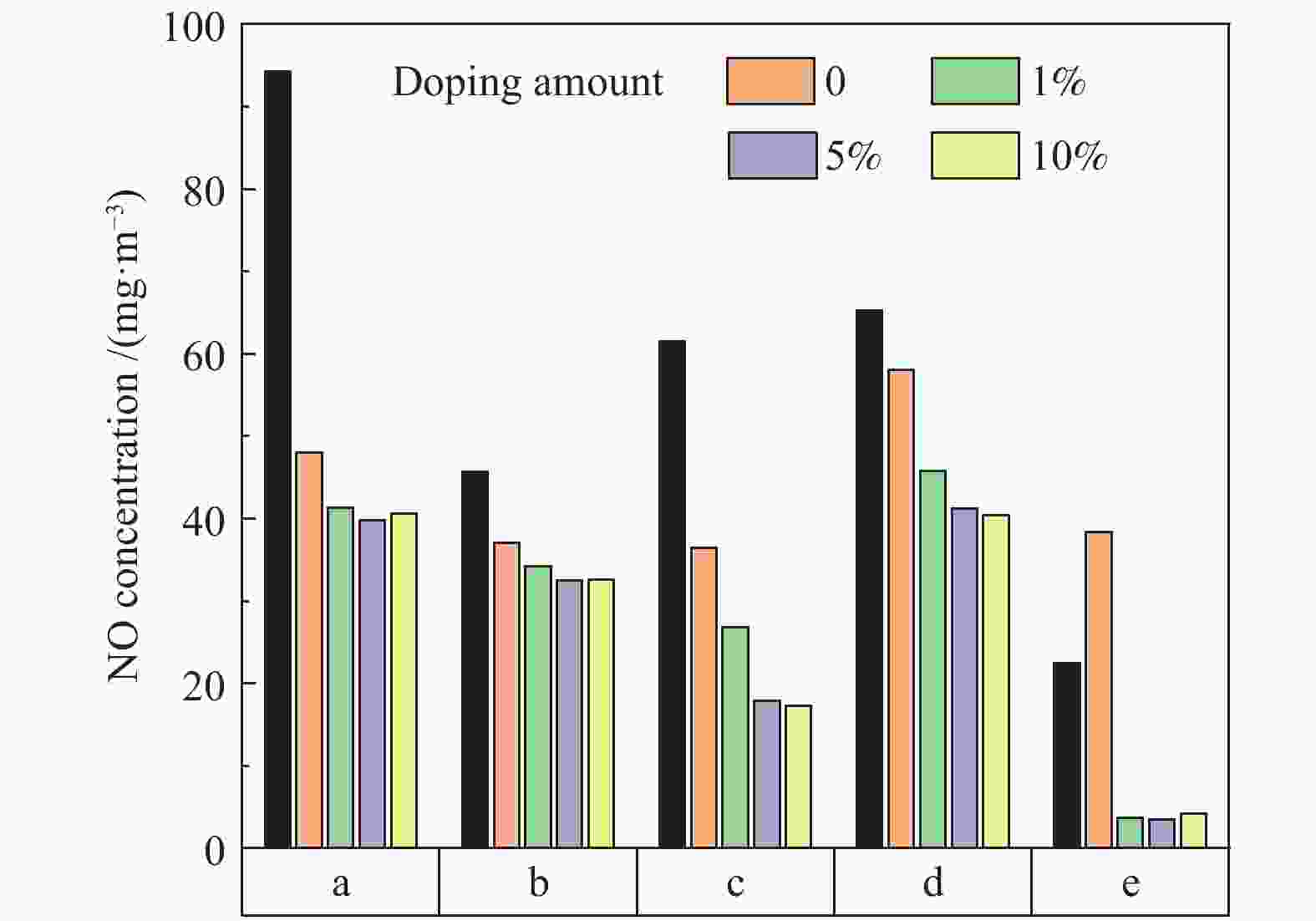
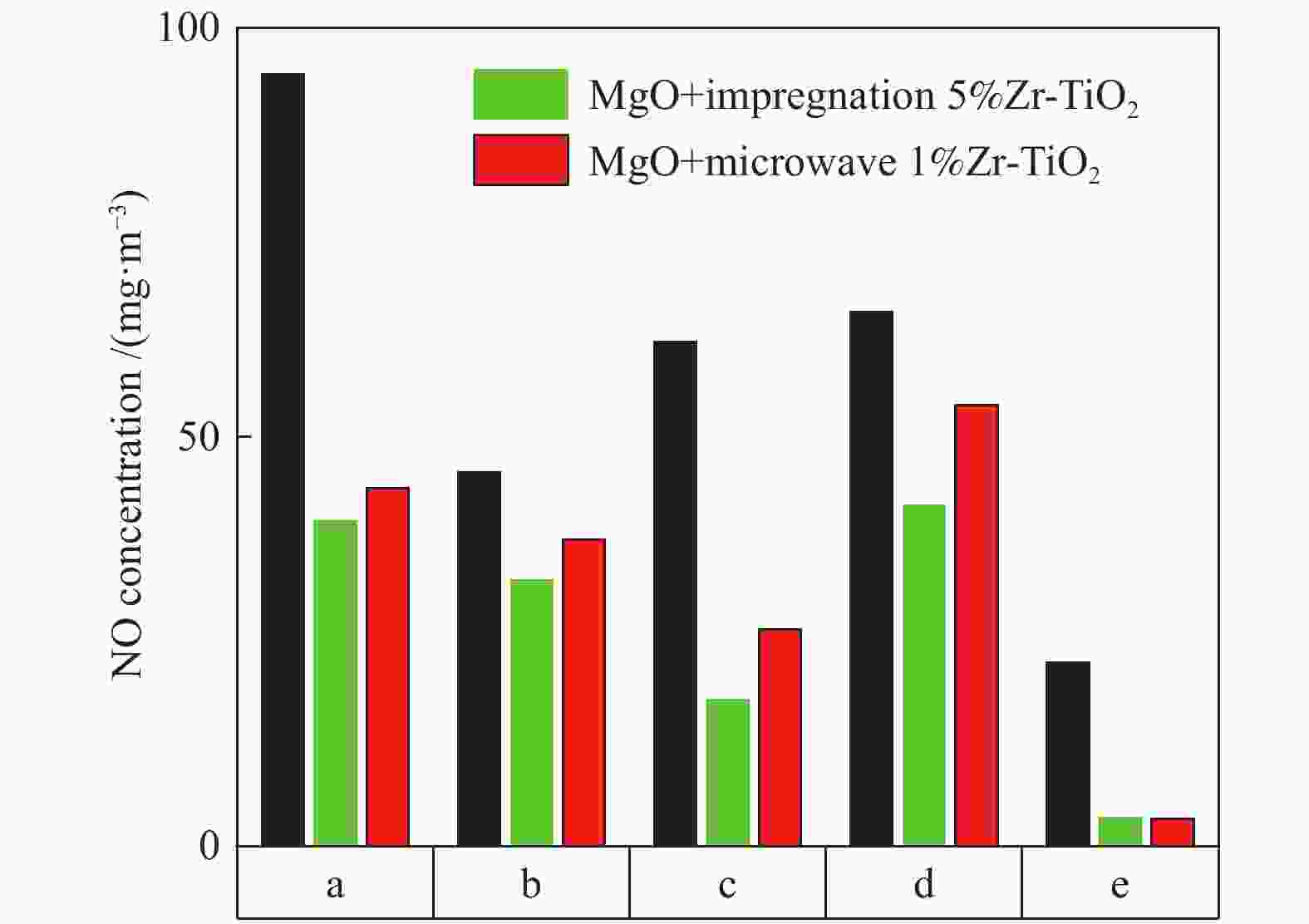
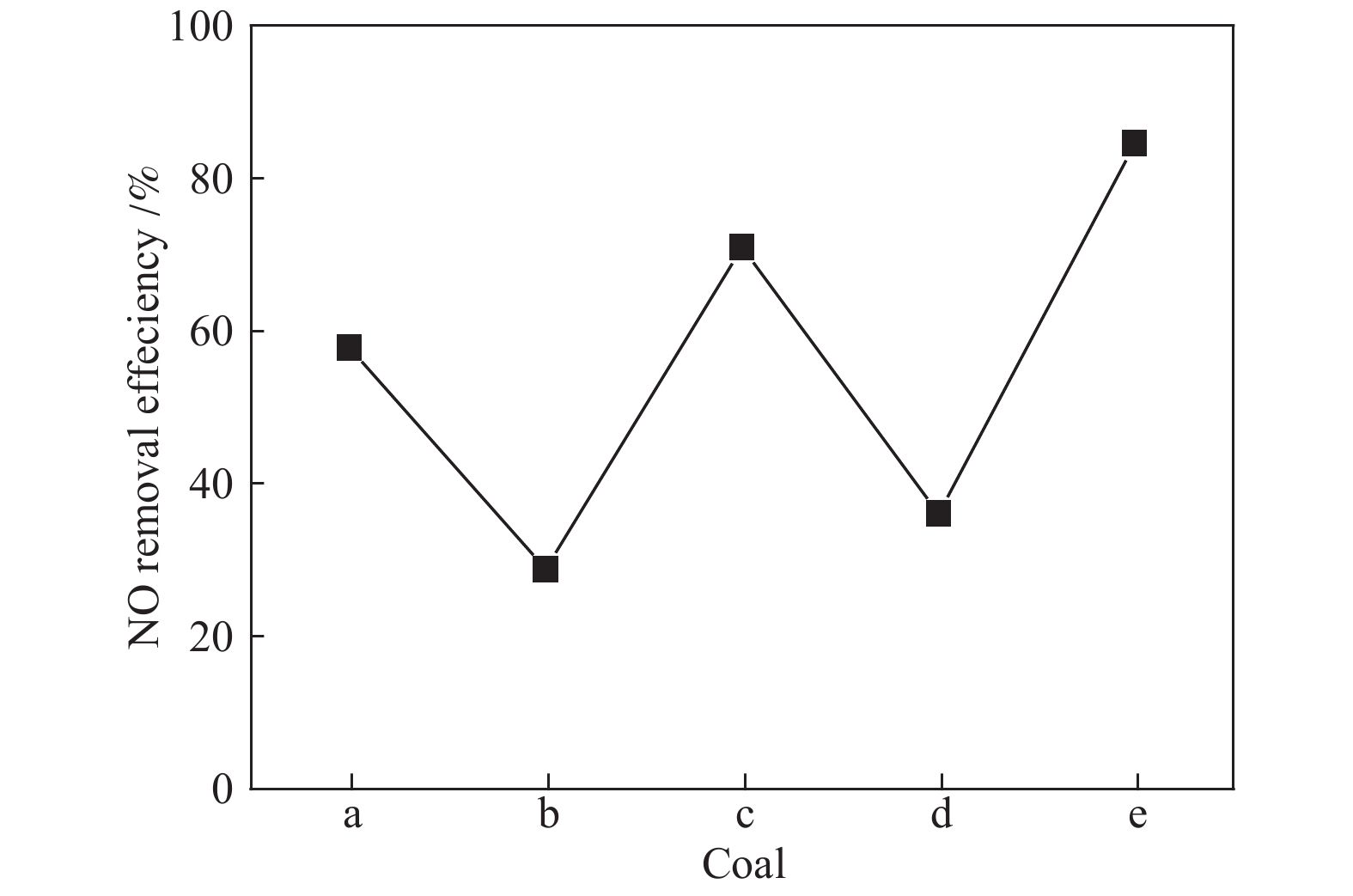
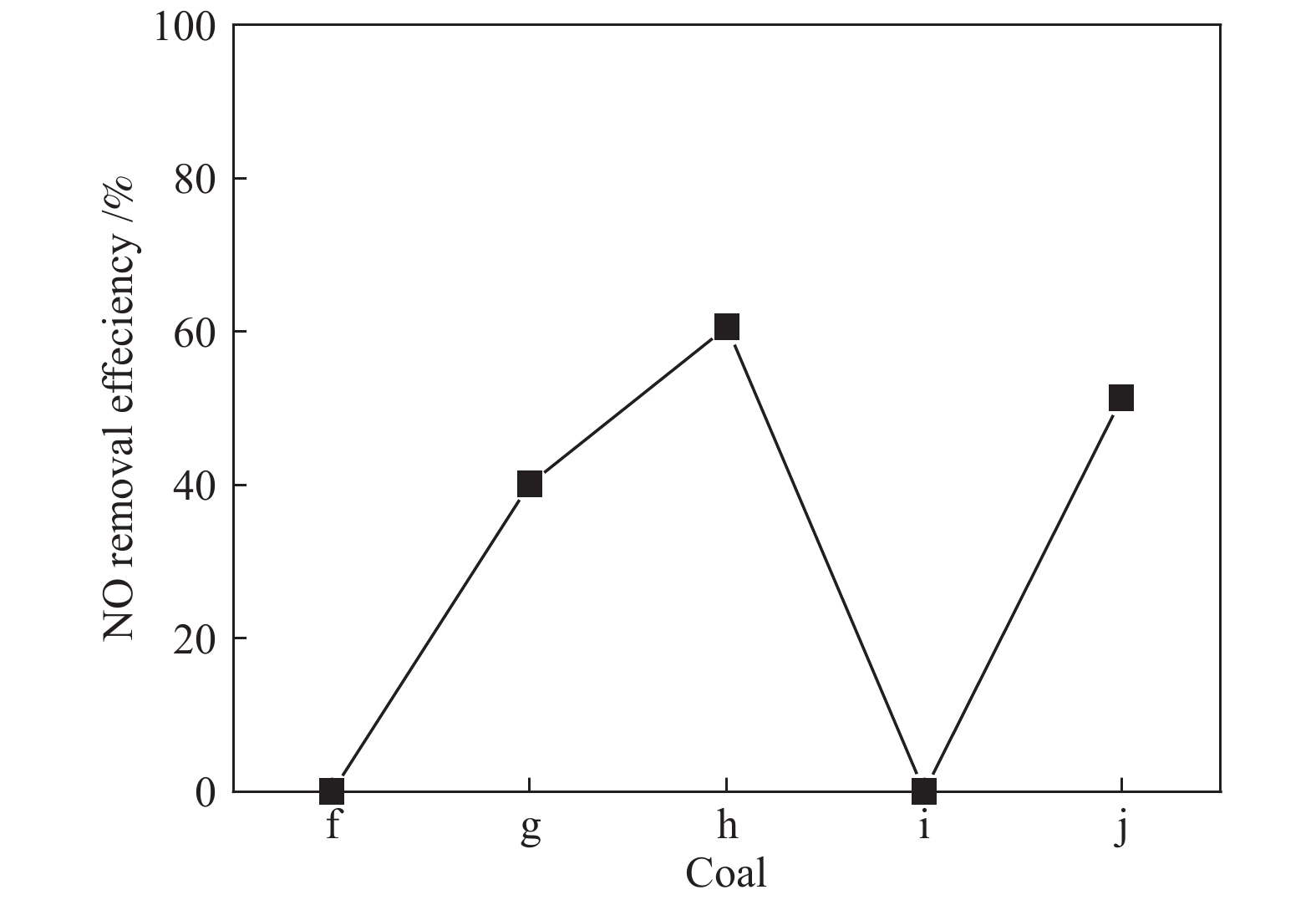
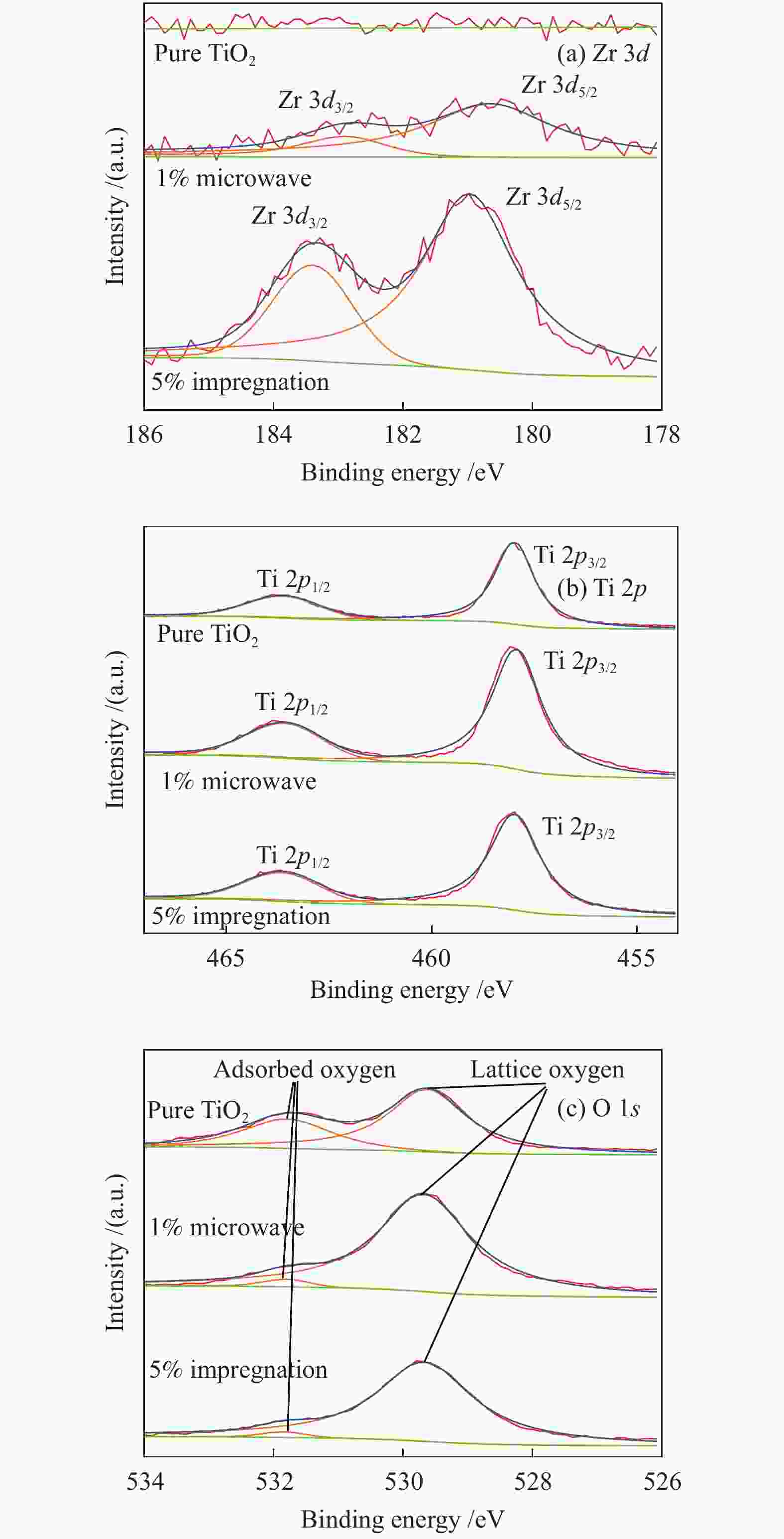
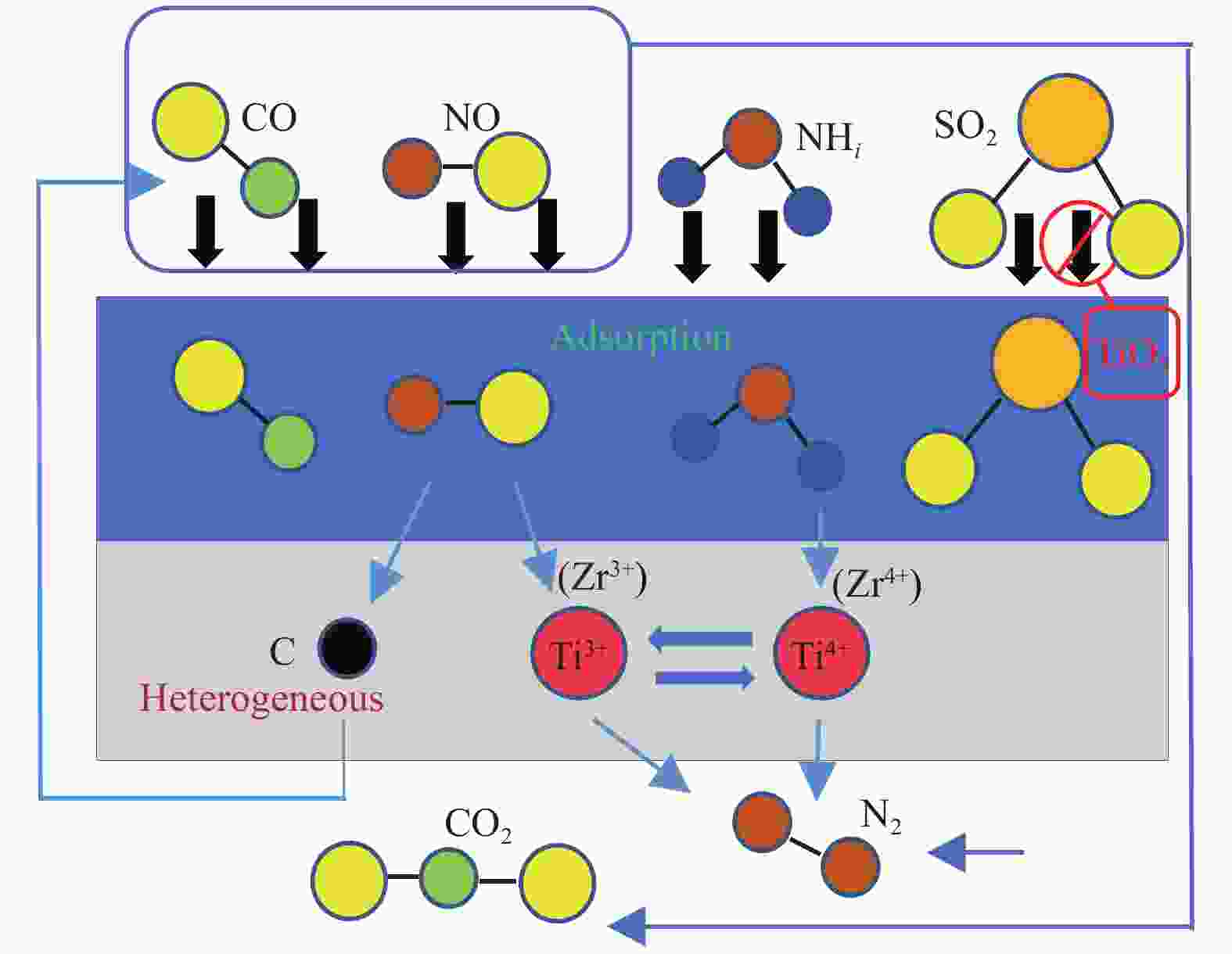
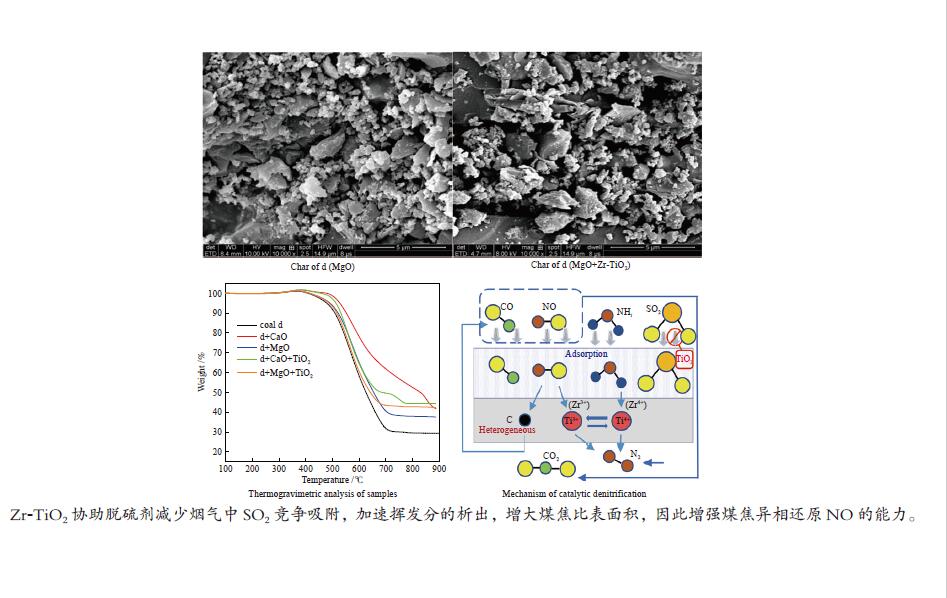
摘要: 采用浸渍法与微波法制取改性的Zr-TiO2,利用管式炉进行劣质煤NO析出实验,探究了脱硫剂、不同方法制备的改性TiO2对劣质煤NO排放的影响及煤种的适应性,并对制备的Zr-TiO2及其煤焦进行XRD、BET、SEM、XPS、TGA表征分析,得出脱硝机理。结果表明,脱硫剂的添加会促进NO的析出,在燃烧温度850 ℃、氧气流量40 mL/min、脱硫剂为MgO时,催化剂为浸渍法5% Zr-TiO2时NO的排放量最低,比纯TiO2低51.0%,比同条件下纯煤燃烧时低84.6%;对选用的10个煤种的适应性实验结果表明,制备的浸渍法5% Zr-TiO2催化剂能够适用于硫分 < 3%、灰分 < 30%的煤种,拓宽了催化剂的应用范围;Zr的掺杂抑制了晶粒增长,增强了活性组分,使得吸附氧含量增多,促进了催化过程中元素价态的转化,加速了挥发分的析出,起到了助燃作用,增大了煤焦的比表面积,增强了煤焦的异相还原能力。Abstract: The effect of desulfurizer, modified TiO2 prepared by different methods on the NO emission from inferior coal burning and the adaptability of coal type were studied by using a tube furnace. The prepared Zr-TiO2 and the char were characterized by XRD, BET, SEM, XPS, TGA, and the mechanism of denitrification was explored. The results show that the addition of desulfurizer can promote NO emission. When the combustion temperature is 850 ℃, oxygen flow rate is 40 mL/min and the desulfurizer is MgO, the NO emission is the lowest as the catalyst of 5% Zr-TiO2 prepared by impregnation method is added, which is 51.0% lower than that with addition of pure TiO2 and 84.6% lower than that with pure coal under the same conditions. The catalyst 5% Zr-TiO2 can be applied to the coal with sulfur < 3% and ash < 30%, having a wide application range. The doping of Zr can inhibit the growth of grain, enhance the active component, increase the content of adsorbed oxygen, promote the transformation of the valence states of the elements, accelerate the devolatilization of volatiles, promote the combustion, increase the specific surface area of chars, and enhance the ability of heterogeneous reduction of chars.
-
Key words:
- inferior coal /
- NO emission characteristics /
- Zr-TiO2 /
- catalysis
-
Table 1 Results of ultimate analysis of the coals
Sample name Ultimate analysis w/% C H N S a 67.91 4.20 1.21 0.69 b 47.25 3.27 0.74 1.37 c 64.01 2.83 1.00 2.01 d 69.36 4.01 0.96 3.82 e 67.71 4.50 0.90 2.82 f 39.01 2.56 0.62 1.35 g 62.29 3.55 0.85 1.31 h 67.90 4.46 0.87 0.85 i 71.51 3.30 1.19 3.25 j 60.38 3.10 0.85 1.81 Table 2 Results of proximate analysis of the coals
Sample name Mad/% Vad/% Aad/% FCad/% Qnet,ad/(MJ·kg−1) Calorific value (kilocalorie)/(cal·g−1) a 9.67 36.87 19.74 33.72 19.24 4601 b 2.93 28.06 36.50 32.51 17.99 4302 c 2.49 13.09 23.65 60.77 24.48 5854 d 2.05 25.60 18.18 54.17 26.03 6225 e 8.71 49.01 13.55 28.73 21.47 5134 f 4.21 20.73 53.56 21.50 10.88 2602 g 5.41 20.21 24.60 49.78 21.53 5149 h 4.90 37.08 7.18 50.84 27.54 6586 i 3.14 15.89 20.41 60.56 25.10 6002 j 3.18 16.51 27.47 52.84 22.24 5319 Table 3 BET data of catalysts
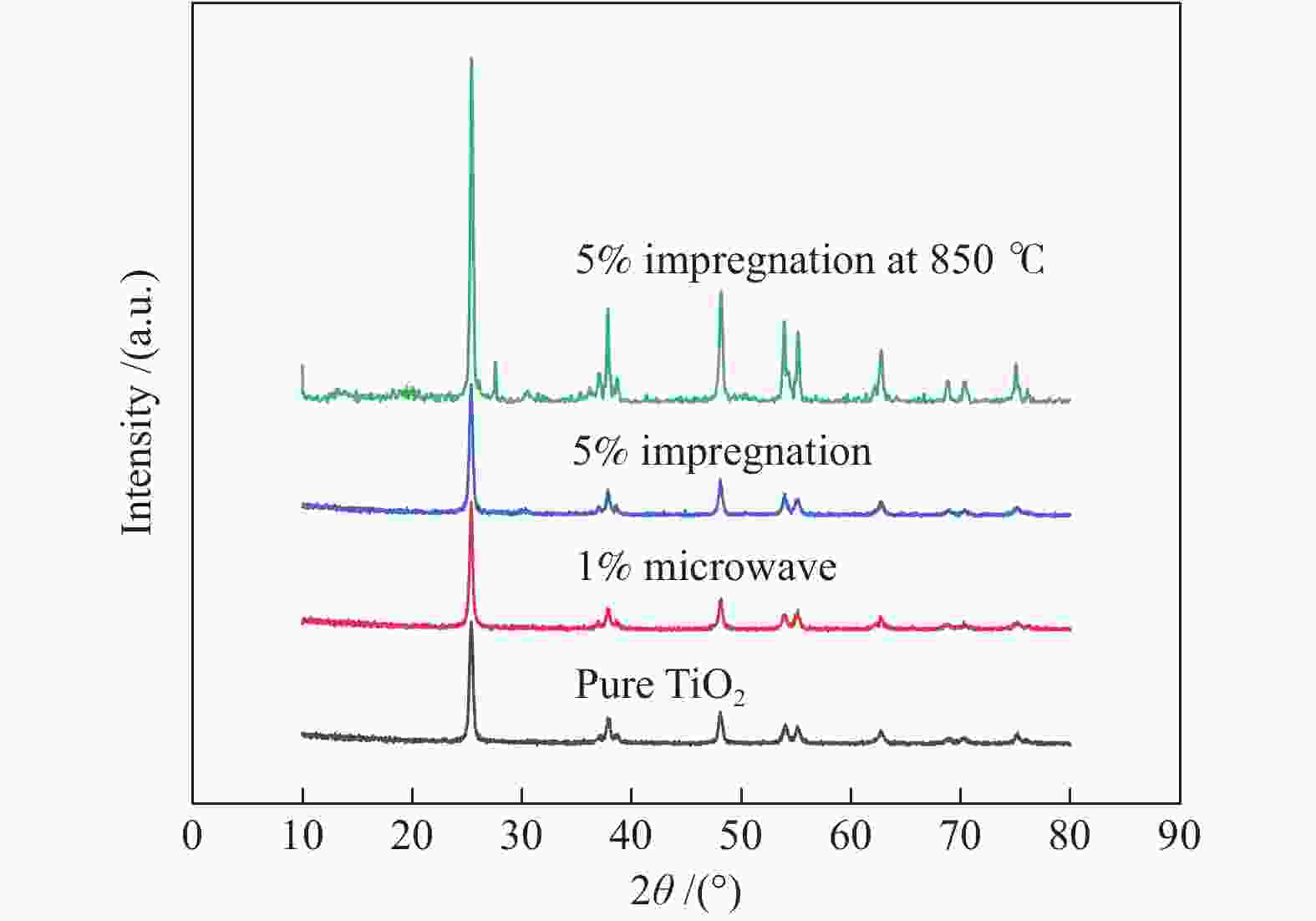
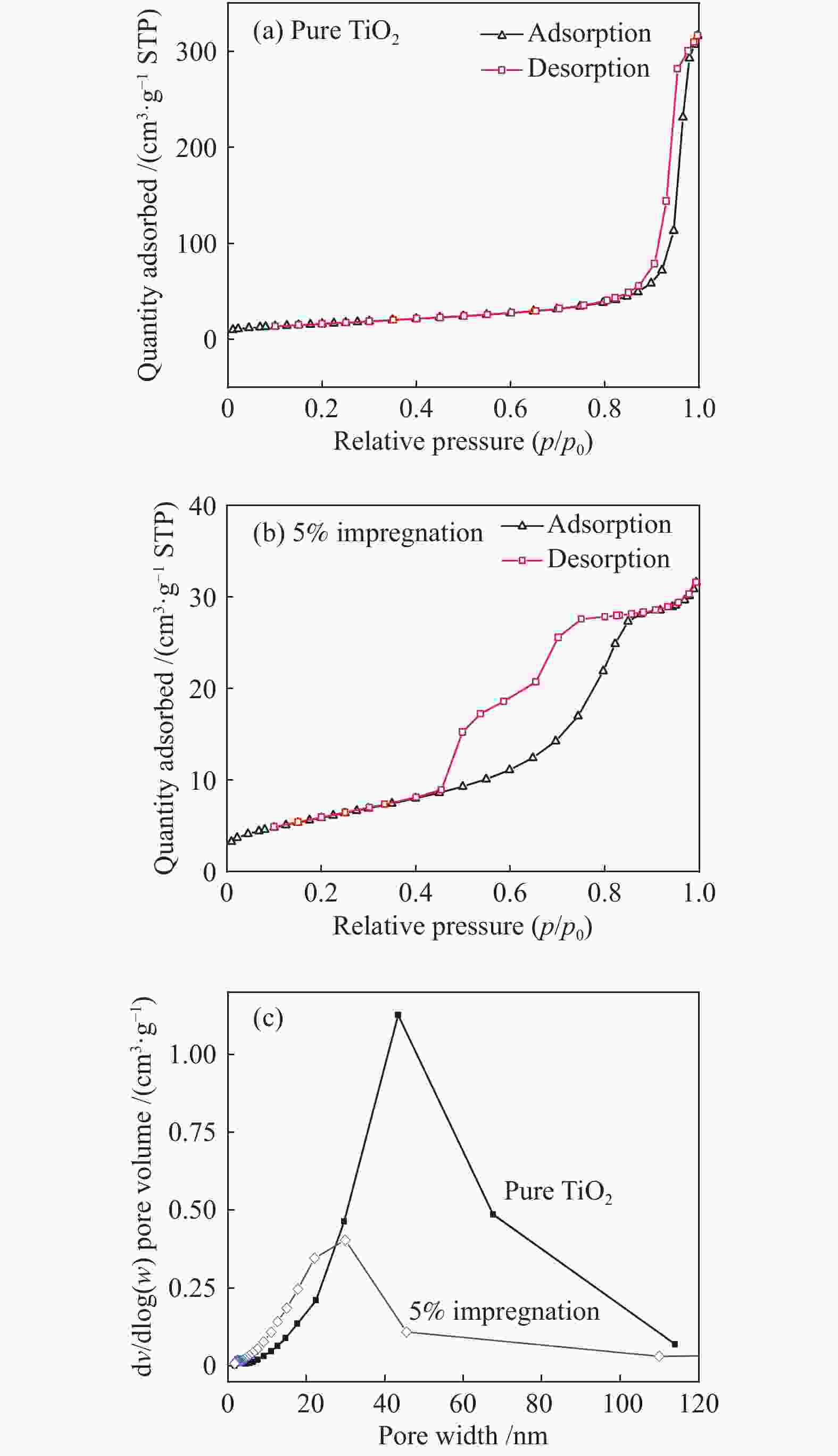
Sample Specific surface area A/(m2·g−1) Average pore size d/nm Pore volume v/(cm3·g−1) Pure TiO2 22.28 31.89 0.0493 5% impregnation 59.42 18.27 0.4747 1% microwave 51.66 8.64 0.2390 Table 4 Ignition and burnout temperature of samples
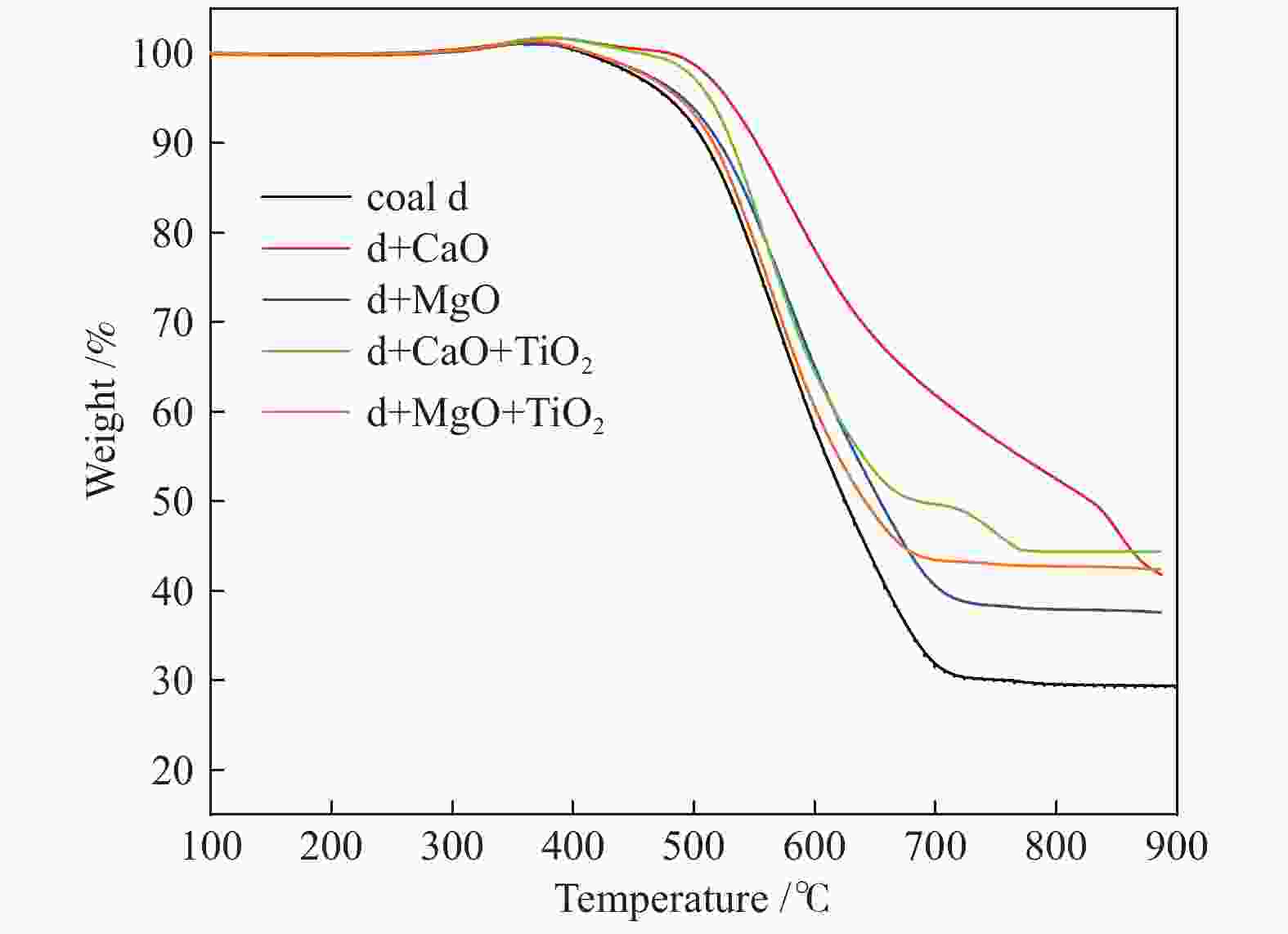
Sample Ignition temperature/℃ Burnout temperature/℃ Coal d 490.55 709.25 d + CaO 514.42 − d + MgO 503.39 714.86 d + CaO + TiO2 503.81 759.49 d + MgO + TiO2 500.9 686.63 Table 5 NO removal efficiency of coal char before and after adding additives
Coal char sample a b c d e Without adding
additives98.3% 89.1% 96.8% 91.1% 96.7% With adding
additives98.0% 97.2% 98.8% 97.4% 98.0% Table 6 BET data of coal char
Coal char BET surface area A/(m2·g−1) Pore size d/nm Pore volume v/(cm3·g−1) d + MgO 2.51 2.28 0.0097 d + MgO + TiO2 39.99 16.66 0.0112 -
[1] ZHANG J X, ZHOU A N, RUN N, CHENG F X, HE X F, YANG Z Y, ZHANG Y T. Controllable preparation of magnetic Mo/HZSM-5@SiO2@Fe3O4 catalyst and its application in coal pyrolysis[J/OL]. J China Coal Soc, 1-10 [2020-07-21]. https://doi.org/10.13225/j.cnki.jccs.2019.1807. [2] ZHANG Y G. Utilization technology of inferior coal[J]. Coal Chem Ind,2002,30(5):24−27. [3] LIANG L T, ZHANG Q, HUANG W, et al. Characteristics of pore structure and gasification reactivity of CO2 in catalytic defocusing of low-rank coals [J/OL]. J China Coal Soc, 1-8[2020-07-21]. https://doi.org/10.13225/j.cnki.jccs.2019.1194. [4] LI H. Experimental study on photocatalytic reduction of CO2 by metal-doped nano-TiO2[D]. Huainan: Anhui University of Science and Technology, 2019. [5] WANG S Q, FU P, HAO L X. Kinetics and mechanism of visible-light photocatalytic oxidation of SO2 over vanadium-doped TiO2[J]. Acta Energ Sol Sin,2015,36(11):2690−2697. [6] QI X. Study on modified TiO2 supported Fe-Mn catalyst for denitrification[D]. Tangshan: North China University of Technology, 2018. [7] WANG L J. Preparation, modification and photocatalytic activity of supported TiO2[D]. Dalian: Dalian University of Technology, 2016. [8] WANG S Q. Experimental study on desulfurization and denitrification with nano-combustion-supporting additive[D]. Baoding: North China Electric Power University, 2009. [9] YIN D Q. Effect of additives on the efficiency of denitration and mercury removal of inferior coal during combustion[D]. Baoding: North China Electric Power University, 2018. [10] LIU M Z. Effect of modified TiO2 on mercury removal and denitration efficiency during biomass and coal combustion[D]. Baoding: North China Electric Power University, 2017. [11] OKASHA F. Enhancing sulphur self-retention by building-in CaO in straw-bitumen pellets[J]. Fuel Process Technol,2007,88(4):401−408. doi: 10.1016/j.fuproc.2006.12.001 [12] LIN R, SU Y X, CHENG J H, ZHANG X W, WEN N N, DENHG W Y, ZHOU H, ZHJAO B T. Reduction of NO by methane over Fe/Ga2O3-Al2O3 catalyst[J]. Chin J Environ Eng,2020,14(6):1592−1604. [13] JIANG Y, LIANG G T, BAO C Z, RUN Y, WANG X C, XING Z M. Selective catalytic reduction of NOx in flue gas over Zr-modified CeO2/TiO2 catalyst[J]. J China Univ Pet (Nat Sci Ed),2017,41(5):139−145. [14] LIU Q, CHENG X L, LI D H, YANG Z J. Effect of Zr doping on electronic structure and optical properties of Anatase TiO2[J]. Sci China,2011,41(1):66−70. [15] XIE C, YAG S H, LI B B, WANG H K, SHI J W, LI G D, NIU C M. C-doped mesoporous anatase TiO2 comprising 10 nm crystallites[J]. J Colloid Interface Sci,2016,1(80):1−8. [16] LI H R, LIANG L R, CHENG S, CHENG X W. Preparation, characterization and properties of Zr-TiO2 photocatalyst[J]. New Chem Mater,2016,44(8):172−174. [17] WANG S Q, LIU M Z, SUN L L, CHENG W L. Study on the mechanism of desulfurization and denitrification catalyzed by TiO2 in the combustion with biomass and coal[J]. Korean J Chem Eng,2017,34(6):1882−1888. doi: 10.1007/s11814-017-0051-z [18] LU L M, KONG C H, VEENA S, HARRIS D. Char Structural ordering during pyrolysis and combustion and its influence on char reactivity[J]. Fuel,2002,81(9):1215−1225. doi: 10.1016/S0016-2361(02)00035-2 -





 下载:
下载: