Effect of phosphorus precursor on the catalytic performance of metal phosphides in the methanation of syngas
-
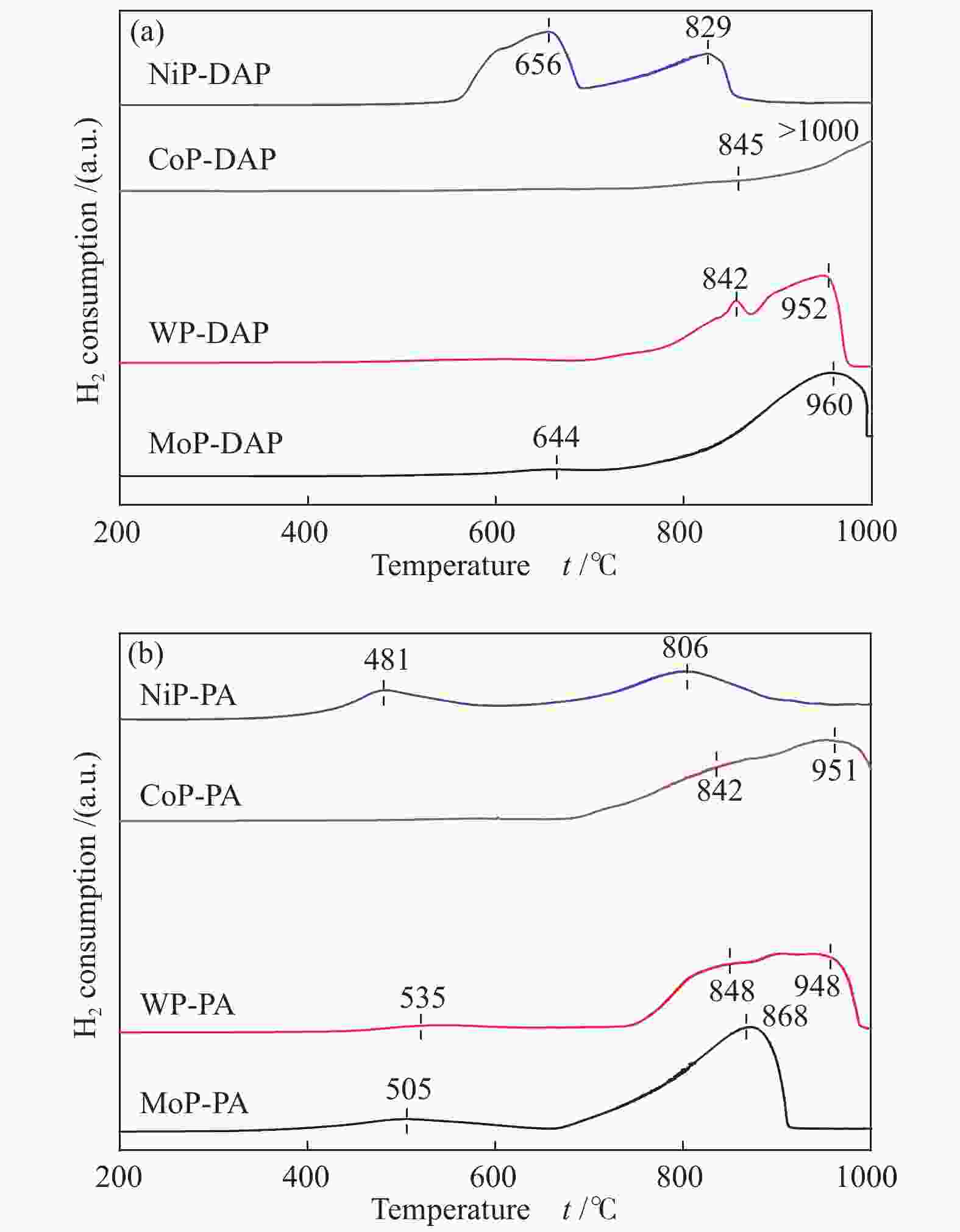
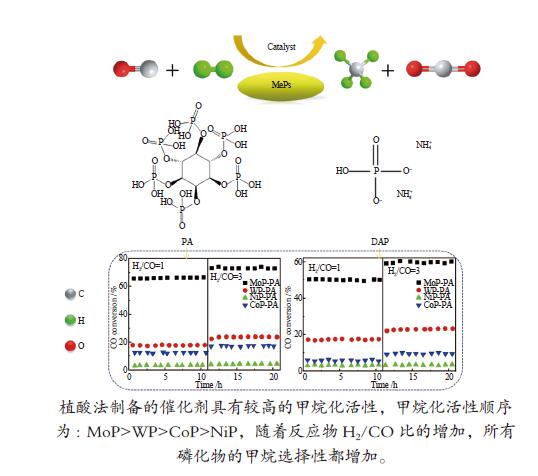
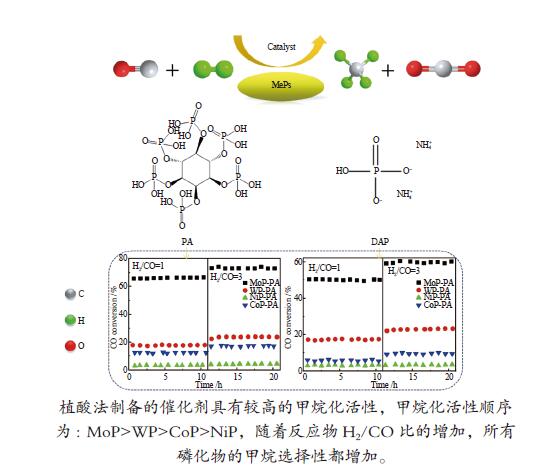
摘要: 采用氢气程序升温还原方法制备了系列金属磷化物催化剂,研究了磷前驱体和H2/CO比对其甲烷化催化性能的影响。结果表明,与磷酸氢二铵相比,由植酸制备的金属磷化物催化剂具有较高的甲烷化活性;植酸作为螯合剂可以有效地分散金属前驱体,降低还原温度,使催化剂具有较高的比表面积和较小的晶粒尺寸,并更好地还原为纯磷化物晶相。不同金属磷化物催化剂的活性顺序为MoP > WP > CoP > NiP。高H2/CO比有利于甲烷化反应进行,随着反应物H2/CO比的增加,所有磷化物催化剂的甲烷选择性都增加。Abstract: A series of metal phosphides including MoP, WP, CoP and NiP was prepared by temperature-programmed reduction with hydrogen from different phosphorus precursors. The effect of phosphorus precursor and feed H2/CO ratio on the catalytic performance of metal phosphides in the methanation was investigated. In comparison with diammonium hydrogen phosphate (DAP), phytic acid (PA) as a chelating agent can effectively disperse the metal precursor, reduce the reduction temperature, promote to form pure phosphide phase, and give the phosphide catalyst a higher surface area and a smaller particle size; as a result, the metal phosphides prepared with PA as a phosphorus precursor exhibit higher catalytic activity in methanation. In addition, the catalytic activity of various metal phosphides in methanation follows the sequence of MoP > WP > CoP > NiP. A high H2/CO ratio in the feed is favorable for the methanation over the phosphide catalysts; the selectivity to methane increases with an increase in the H2/CO ratio.
-
Key words:
- phytic acid /
- methanation /
- phosphate /
- metal phosphide /
- H2/CO ratio /
- catalytic performance
-
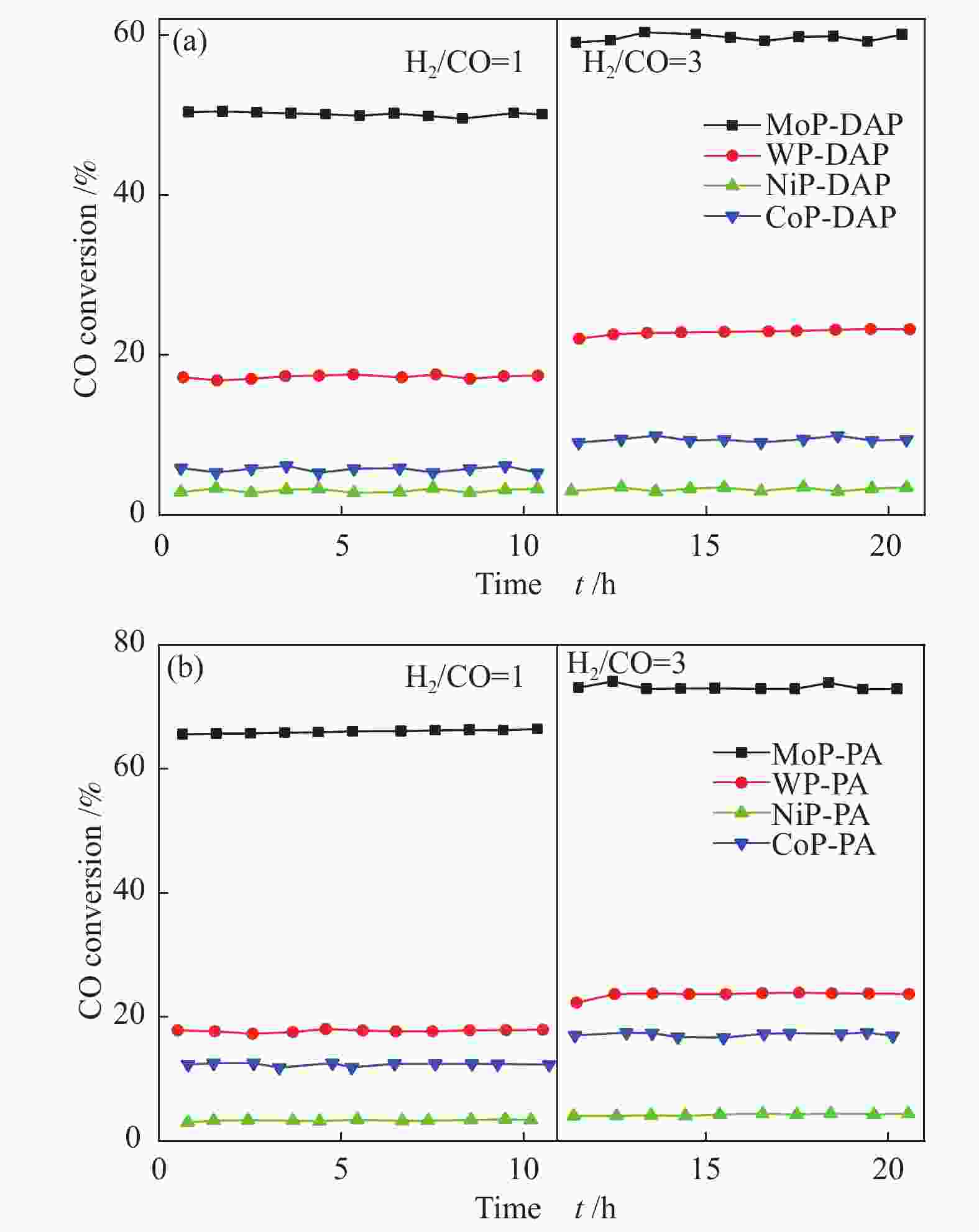
Table 1 Methanation performance of various metal phosphide catalysts at 550 °C, 3.0 MPa, and 5000 mL/(g·h)
Catalyst H2/CO = 1 H2/CO = 3 $x_{ {\rm{CO} } }$/% $ s_{{\rm{CO}}_2} $/% $ s_{{\rm{CH}}_4} $/% $ s_{{\rm{C}}_2{\rm{H}}_6} $/% $ x_{{\rm{CO}}} $/% $ s_{{\rm{CO}}_2} $/% $ s_{{\rm{CH}}_4} $/% $ s_{{\rm{C}}_2{\rm{H}}_6} $/% MoP-DAP 50.1 45.8 53.1 1.1 59.6 41.9 57.2 0.9 MoP-PA 65.6 46.8 51.9 1.3 72.5 31.9 67.5 0.6 WP-DAP 17.3 41.9 57.5 0.6 22.8 32.0 67.5 0.5 WP-PA 21.3 43.0 56.0 1.0 23.8 32.9 66.2 0.9 CoP-DAP 5.8 20.0 78.1 1.9 9.4 10.1 88.8 1.1 CoP-PA 12.4 23.6 75.8 0.4 17.3 16.8 82.1 1.1 NiP-DAP 3.2 13.4 84.0 2.6 3.4 7.0 90.3 2.7 NiP-PA 3.2 14.3 84.1 1.6 4.3 12.0 86.3 1.7 Table 2 Textural properties determined by N2 sorption of various phosphide catalysts
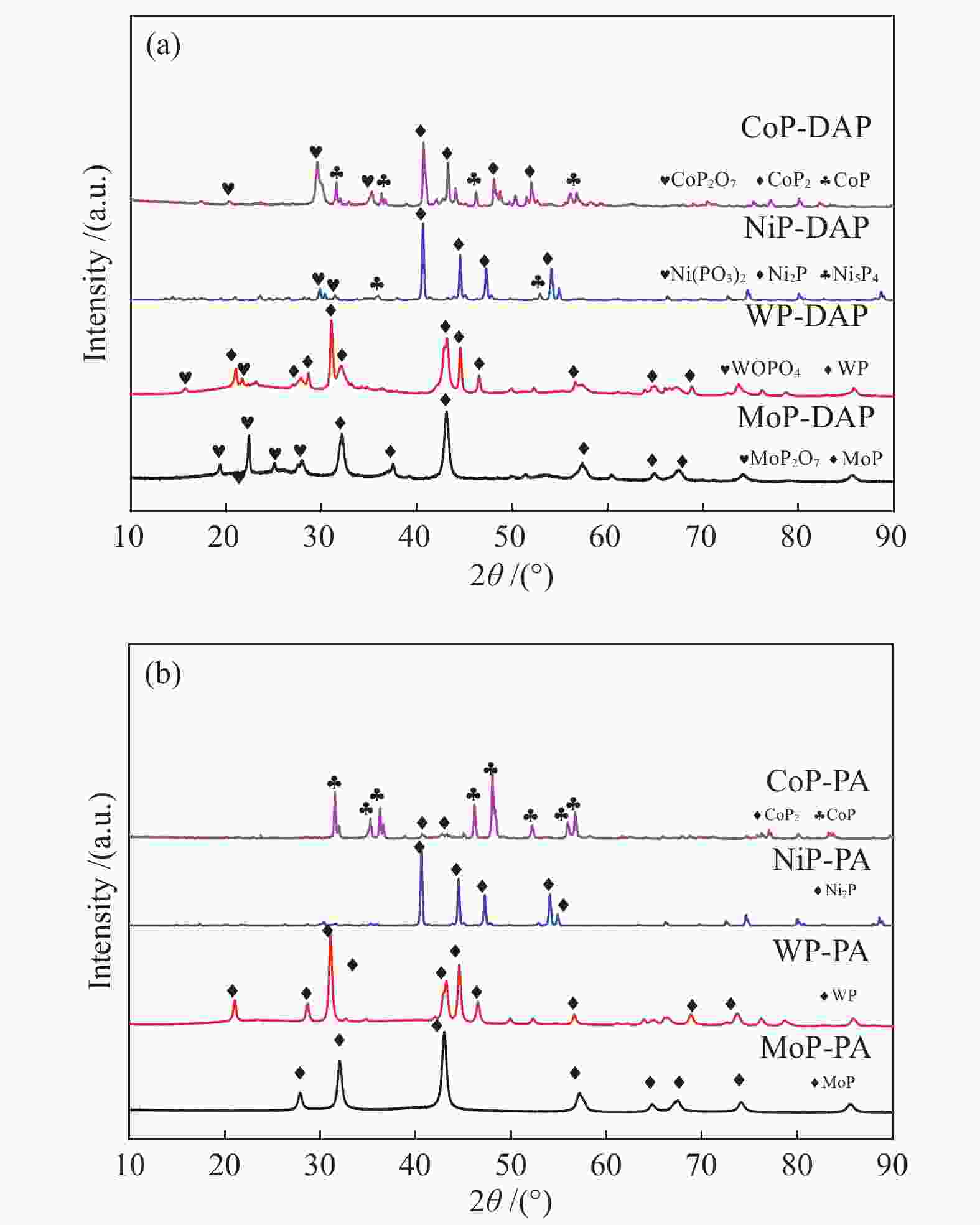
Catalyst Surface area A/(m2·g−1) Pore volume v/(cm3·g−1) Average pore size d/nm Particle size d/nm MoP-DAP 3.7 0.022 27.0 19.7 MoP-PA 40.1 0.010 8.9 14.7 WP-DAP 3.2 0.024 55.5 35.5 WP-PA 27.4 0.083 11.1 22.9 CoP-DAP 1.0 0.003 52.6 90.8 CoP-PA 10.9 0.062 30.2 38.1 NiP-DAP 4.0 0.029 48.1 25.8 NiP-PA 8.1 0.021 38.3 19.7 -
[1] KUSTOV A L, FREY A M, LARSEN K E, JOHANNESSEN T, NØRSKOV J K, CHRISTENSEN C H. CO methanation over supported bimetallic Ni-Fe catalysts: From computational studies towards catalyst optimization[J]. Appl Catal A: Gen,2007,320:98−104. doi: 10.1016/j.apcata.2006.12.017 [2] YAN Shuang-hua, SHUANG Jian-yong, HU Si-bin. Methane synthesis technology in coal to natural gas process[J]. Chem Fert Des,2010,48(2):19−21+32. [3] MENG F, LI X, SHAW G M, SMITH P J, MORGAN D J, PERDJON M, LI Z. Sacrificial carbon strategy toward enhancement of slurry methanation activity and stability over Ni-Zr/SiO2 catalyst[J]. Ind Eng Chem Res,2018,57(14):4798−4806. doi: 10.1021/acs.iecr.7b05157 [4] MENG F, LI X, LI M, CUI X, LI Z. Catalytic performance of CO methanation over La-promoted Ni/Al2O3 catalyst in a slurry-bed reactor[J]. Chem Eng J,2017,313:1548−1555. doi: 10.1016/j.cej.2016.11.038 [5] WANG G, XU S. Research progress of coal based alternative/synthetic natural gas technology[J]. Petrochem Technol,2016,45(1):1−9. [6] MENG F, LI X, LV X, LI Z. CO hydrogenation combined with water-gas-shift reaction for synthetic natural gas production: A thermodynamic and experimental study[J]. Int J Coal Sci Technol,2018,5(4):439−451. [7] LIU C, WANG W, XU Y, LI Z, WANG B, MA X. Effect of zirconia morphology on sulfur-resistant methanation performance of MoO3/ZrO2 catalyst[J]. Appl Surf Sci,2018,441:482−490. doi: 10.1016/j.apsusc.2018.02.019 [8] HU H, WANG W, LIU Z, WANG B, LI Z, MA X. Sulfur-resistant CO methanation to CH4 over MoS2/ZrO2 catalysts: Support size effect on morphology and performance of Mo species[J]. Catal Lett,2018,148(8):2585−2595. doi: 10.1007/s10562-018-2438-9 [9] HULLIGER F. Crystal chemistry of the chalcogenides and pnictides of the transition elements[J]. Struct Bond,1968,4(6):83−229. [10] PÖTTGEN R, HÖNLE W, SCHNERING H G. Phosphides: Solid - State Chemistry [M]. 2006, 1−16. [11] YANG Y, ZHONG Y, SHI Q, WANG Z, SUN K, WANG H. Electrocatalysis in lithium sulfur batteries under lean electrolyte conditions[J]. Angew Chem Int Ed,2018,57(47):15549−15552. doi: 10.1002/anie.201808311 [12] PRYTZ Ø, LØVVIK O, TAFTØ J. Comparison of theoretical and experimental dielectric functions: Electron energy-loss spectroscopy and density-functional calculations on skutterudites[J]. Phys Rev B,2006,74:245109. [13] OSHTRAKH MI, LARIONOV MY, GROKHOVSKY VI, SEMIONKIN VA. Study of rhabdite (iron nickel phosphide) microcrystals extracted from Sikhote–Alin iron meteorite by magnetization measurements and Mössbauer spectroscopy[J]. Mater Chem Phys,2011,130(1):373−380. [14] TEGUS O, BRÜCK E, BUSCHOW K H, DE BOER F R. Transition-metal-based magnetic refrigerants for room-temperature applications[J]. Nature,2002,415(6868):150−152. doi: 10.1038/415150a [15] BARZ H, KU H C, MEISNER G P, FISK Z, MATTHIAS B T. Ternary transition metal phosphides: High-temperature superconductors[J]. Proc Natl Acad Sci USA,1980,77(6):3132−3134. doi: 10.1073/pnas.77.6.3132 [16] MONTESINOS-CASTELLANOS A, ZEPEDA T A, PAWELEC B, LIMA E, FIERRO J L G, OLIVAS A, DE LOS REYES H J A. Influence of reduction temperature and metal loading on the performance of molybdenum phosphide catalysts for dibenzothiophene hydrodesulfurization[J]. Appl Catal A: Gen,2008,334(1):330−338. [17] GUO C, TIRUMALA VENKATESWARA RAO K, REYHANITASH E, YUAN Z, ROHANI S, XU C, HE S. Novel inexpensive transition metal phosphide catalysts for upgrading of pyrolysis oil via hydrodeoxygenation[J]. AIChE J,2016,62(10):3664−3672. doi: 10.1002/aic.15286 [18] INFANTES-MOLINA A, MORENO-LEÓN C, PAWELEC B, FIERRO J L G, RODRIGUEZ-CASTELLON E, JIMÉNEZ-LÓPEZ A. Simultaneous hydrodesulfurization and hydrodenitrogenation on MoP/SiO2 catalysts: Effect of catalyst preparation method[J]. Appl Catal B: Environ,2012,113-114:87−99. doi: 10.1016/j.apcatb.2011.11.022 [19] WANG D, ZHANG D, TANG C, ZHOU P, WU Z, FANG B. Hydrogen evolution catalyzed by cobalt-promoted molybdenum phosphide nanoparticles[J]. Catal Sci Technol,2016,6(6):1952−1956. doi: 10.1039/C5CY01457C [20] KIBSGAARD J, JARAMILLO TF. Molybdenum phosphosulfide: An active, acid-stable, earth-abundant catalyst for the hydrogen evolution reaction[J]. Angew Chem Int Ed,2014,53(52):14433−14437. doi: 10.1002/anie.201408222 [21] CHEN J, YANG Y, SHI H, LI M, CHU Y, PAN Z, YU X. Regulating product distribution in deoxygenation of methyl laurate on silica-supported Ni-Mo phosphides: Effect of Ni/Mo ratio[J]. Fuel,2014,129:1−10. doi: 10.1016/j.fuel.2014.03.049 [22] ZHANG G, WANG G, LIU Y, LIU H, QU J, LI J. Highly active and stable catalysts of phytic acid-derivative transition metal p for full water splitting[J]. J Am Chem Soc,2016,138(44):14686−14693. doi: 10.1021/jacs.6b08491 [23] WANG X, NA Z, YIN D, WANG C, WU Y, HUANG G, WANG L. Phytic acid-assisted formation of hierarchical porous CoP/C nanoboxes for enhanced lithium storage and hydrogen generation[J]. ACS Nano,2018,12(12):12238−12246. doi: 10.1021/acsnano.8b06039 [24] ZHUANG M, OU X, DOU Y, ZHANG L, ZHANG Q, WU R, DING Y, SHAO M, LUO Z. Polymer-embedded fabrication of Co2P nanoparticles encapsulated in N, P-doped graphene for hydrogen generation[J]. Nano Lett,2016,16(7):4691−4698. doi: 10.1021/acs.nanolett.6b02203 [25] PU Z, AMIINU IS, ZHANG C, WANG M, KOU Z, MU S. Phytic acid-derivative transition metal phosphides encapsulated in N, P-codoped carbon: an efficient and durable hydrogen evolution electrocatalyst in a wide pH range[J]. Nanoscale,2017,9(10):3555−3560. doi: 10.1039/C6NR09883E [26] ZHANG C, PU Z, AMIINU I S, ZHAO Y, ZHU J, TANG Y, MU S. Co2P quantum dot embedded N, P dual-doped carbon self-supported electrodes with flexible and binder-free properties for efficient hydrogen evolution reactions[J]. Nanoscale,2018,10(6):2902−2907. doi: 10.1039/C7NR08148K [27] WANG B, DING G, SHANG Y, LV J, WANG H, WANG E, LI Z, MA X, QIN S, SUN Q. Effects of MoO3 loading and calcination temperature on the activity of the sulphur-resistant methanation catalyst MoO3/γ-Al2O3[J]. Appl Catal A: Gen,2012,431-432:144−150. doi: 10.1016/j.apcata.2012.04.029 [28] LIU Z, XU Y, CHENG J, WANG W, WANG B, LI Z, MA X. Comparative study on cubic and tetragonal CexZr1-xO2 supported MoO3-catalysts for sulfur-resistant methanation[J]. Appl Surf Sci,2018,433:730−738. doi: 10.1016/j.apsusc.2017.10.103 [29] OYAMA S T. Novel catalysts for advanced hydroprocessing: transition metal phosphides[J]. J Catal,2003,216(1):343−352. [30] STINNER C, TANG Z, HAOUAS M, WEBER T, PRINS R. Preparation and 31P NMR characterization of nickel phosphides on silica[J]. J Catal,2002,208(2):456−466. [31] CHEN J, SHI H, LI L, LI K. Deoxygenation of methyl laurate as a model compound to hydrocarbons on transition metal phosphide catalysts[J]. Appl Catal B: Environ,2014,144:870−884. doi: 10.1016/j.apcatb.2013.08.026 [32] LIANG X, ZHANG D, WU Z, WANG D. The Fe-promoted MoP catalyst with high activity for water splitting[J]. Appl Catal A: Gen,2016,524:134−138. doi: 10.1016/j.apcata.2016.06.029 [33] WANG D, SHEN Y, ZHANG X, WU Z. Enhanced hydrogen evolution from the MoP/C hybrid by the modification of ketjen black[J]. J Mater Sci,2017,52(6):3337−3343. doi: 10.1007/s10853-016-0621-1 [34] PRINS R, BUSSELL M E. Metal phosphides: Preparation, characterization and catalytic reactivity[J]. Catal Lett,2012,142(12):1413−1436. doi: 10.1007/s10562-012-0929-7 [35] WHIFFEN V M L, SMITH K J, STRAUS S K. The influence of citric acid on the synthesis and activity of high surface area MoP for the hydrodeoxygenation of 4-methylphenol[J]. Appl Catal A: Gen,2012,419−420:111−125. doi: 10.1016/j.apcata.2012.01.018 [36] WANG R, SMITH K J. Hydrodesulfurization of 4, 6-dimethyldibenzothiophene over high surface area metal phosphides[J]. Appl Catal A: Gen,2009,361(1):18−25. [37] XIAO P, SK M A, THIA L, GE X, LIM R J, WANG J-Y, LIM K H, WANG X. Molybdenum phosphide as an efficient electrocatalyst for the hydrogen evolution reaction[J]. Energy Environ Sci,2014,7(8):2624−2629. doi: 10.1039/C4EE00957F [38] BU P, CECILIA J A, OYAMA S T, TAKAGAKI A, INFANTES-MOLINA A, ZHAO H, LI D, RODRÍGUEZ-CASTELLÓN E, JIMÉNEZ LÓPEZ A. Studies of the synthesis of transition metal phosphides and their activity in the hydrodeoxygenation of a biofuel model compound[J]. J Catal,2012,294:184−198. doi: 10.1016/j.jcat.2012.07.021 -




 下载:
下载: