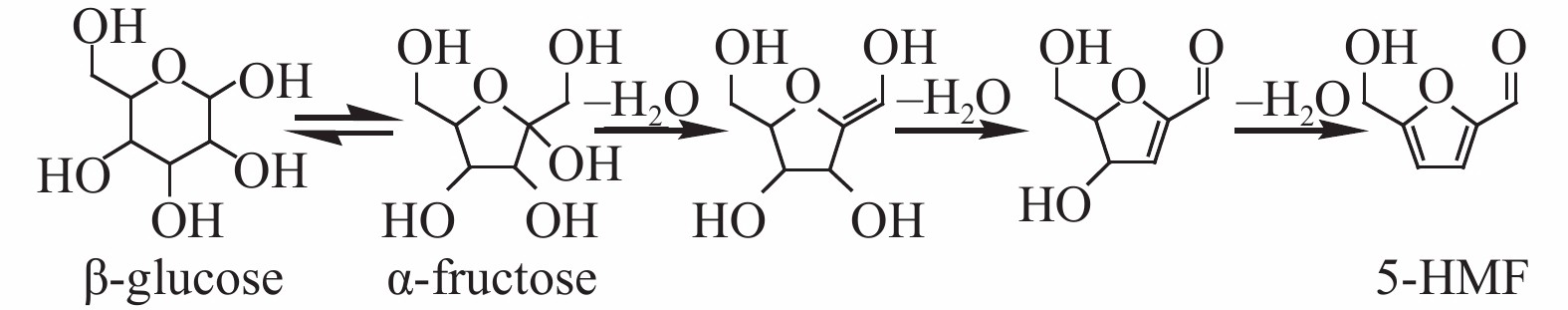
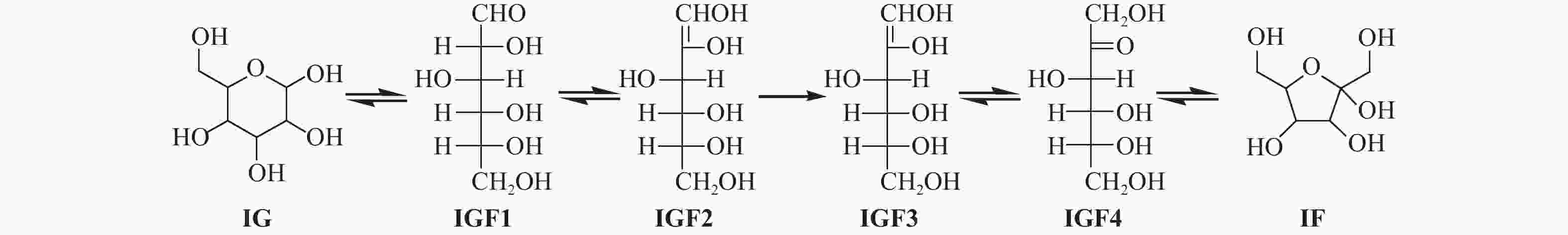
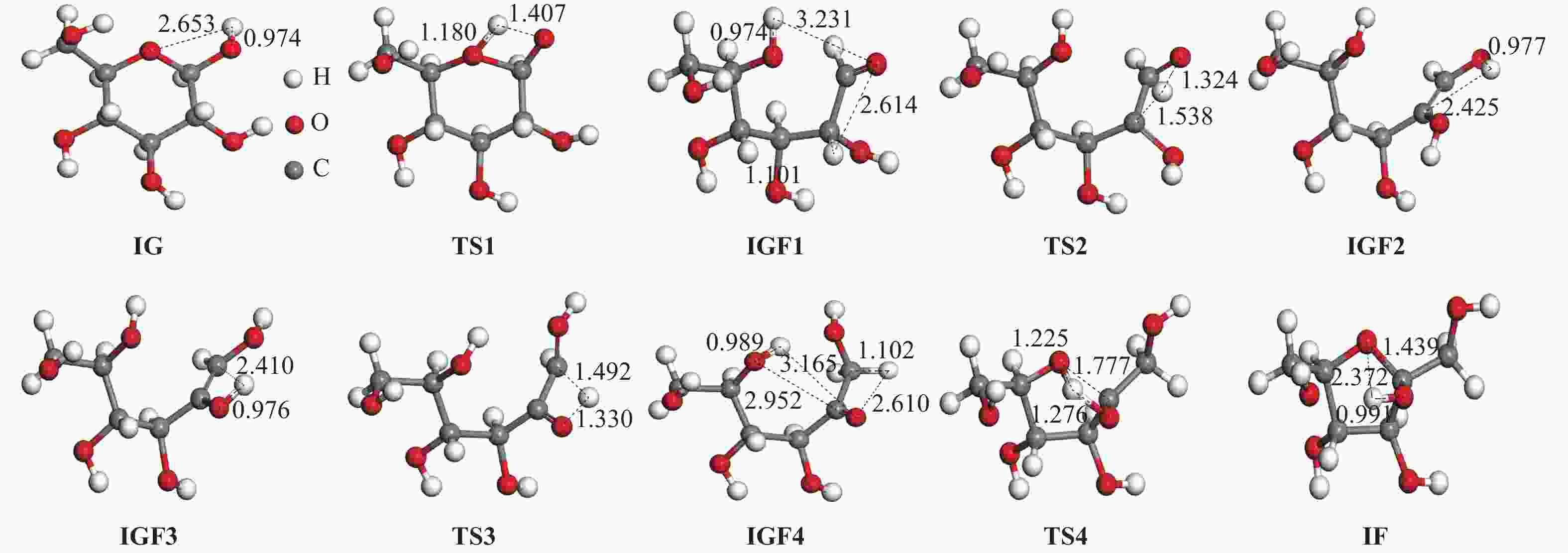
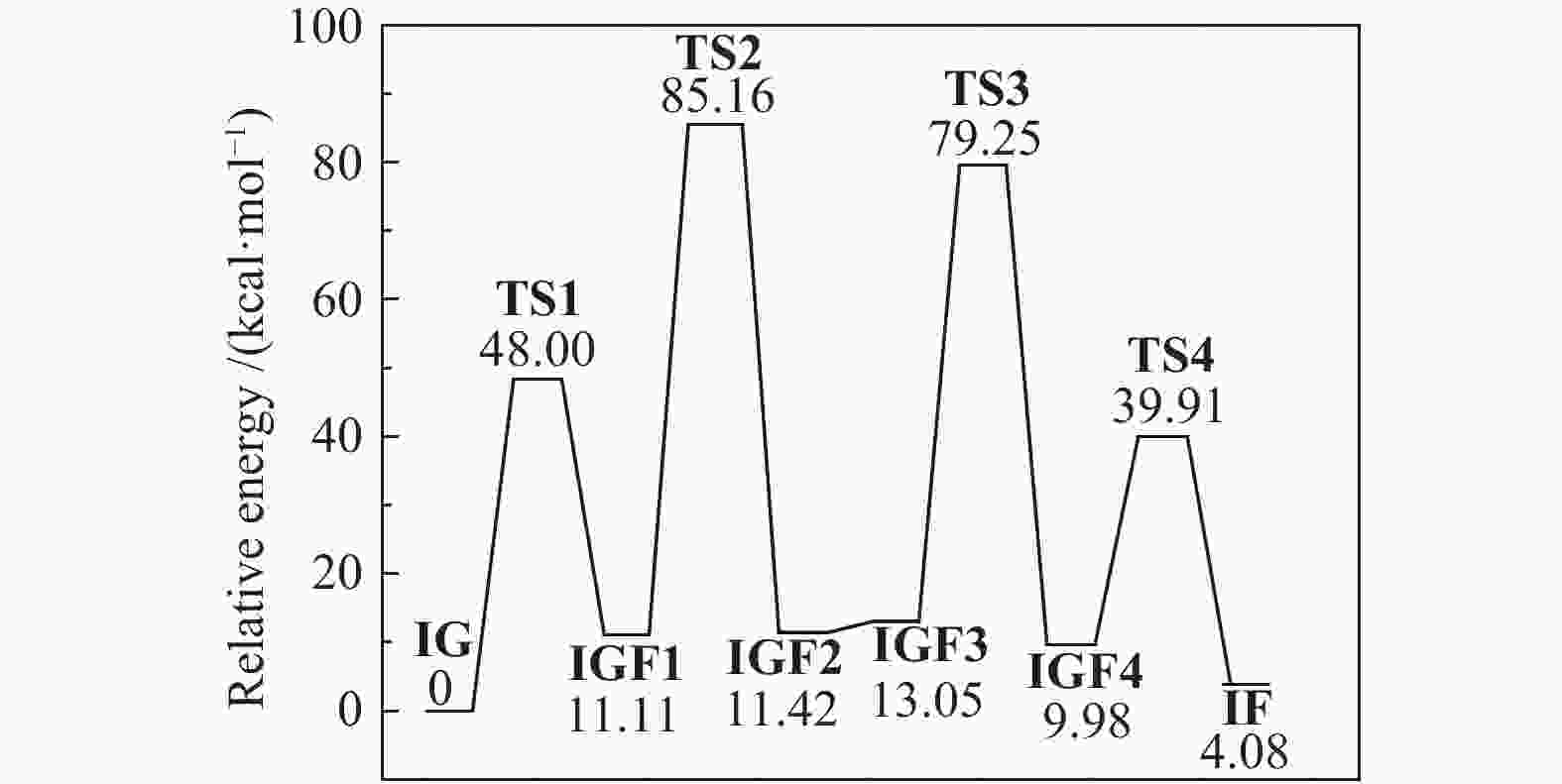
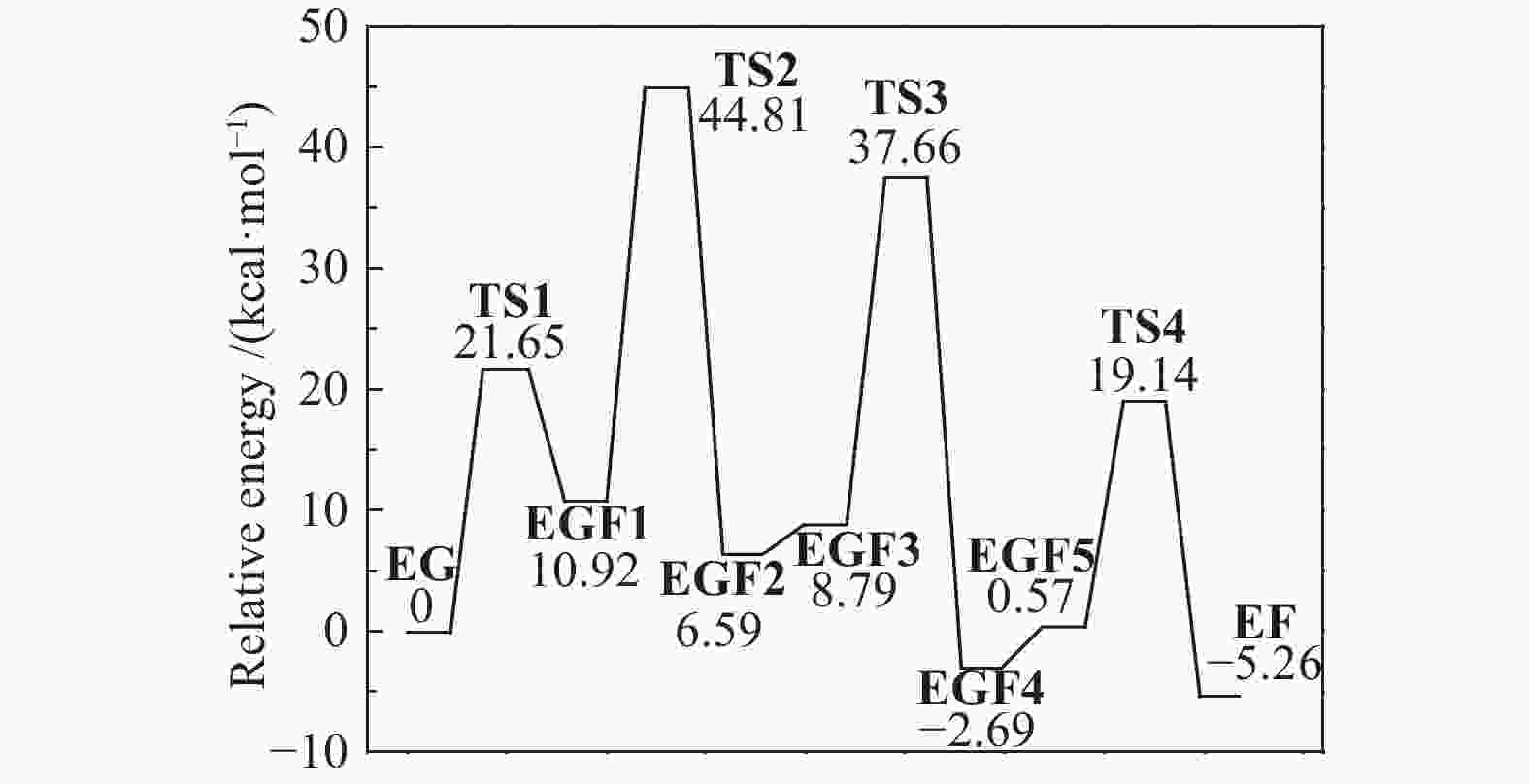
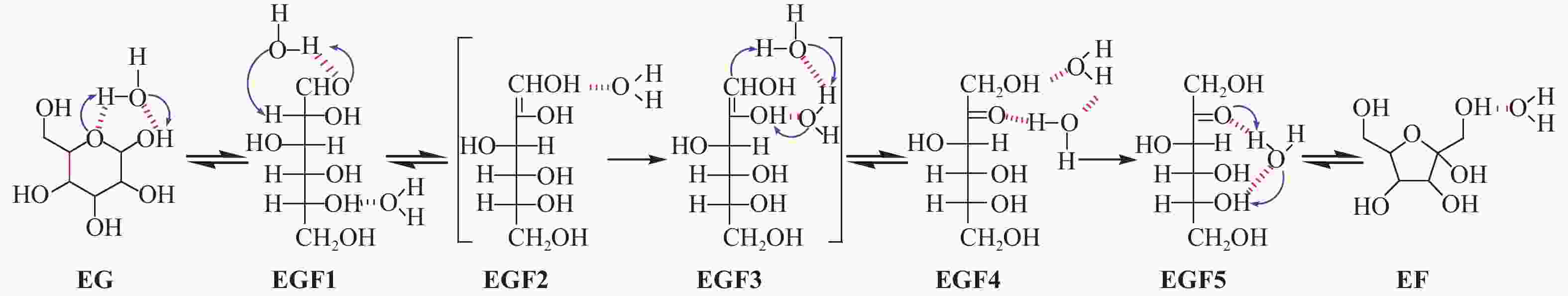
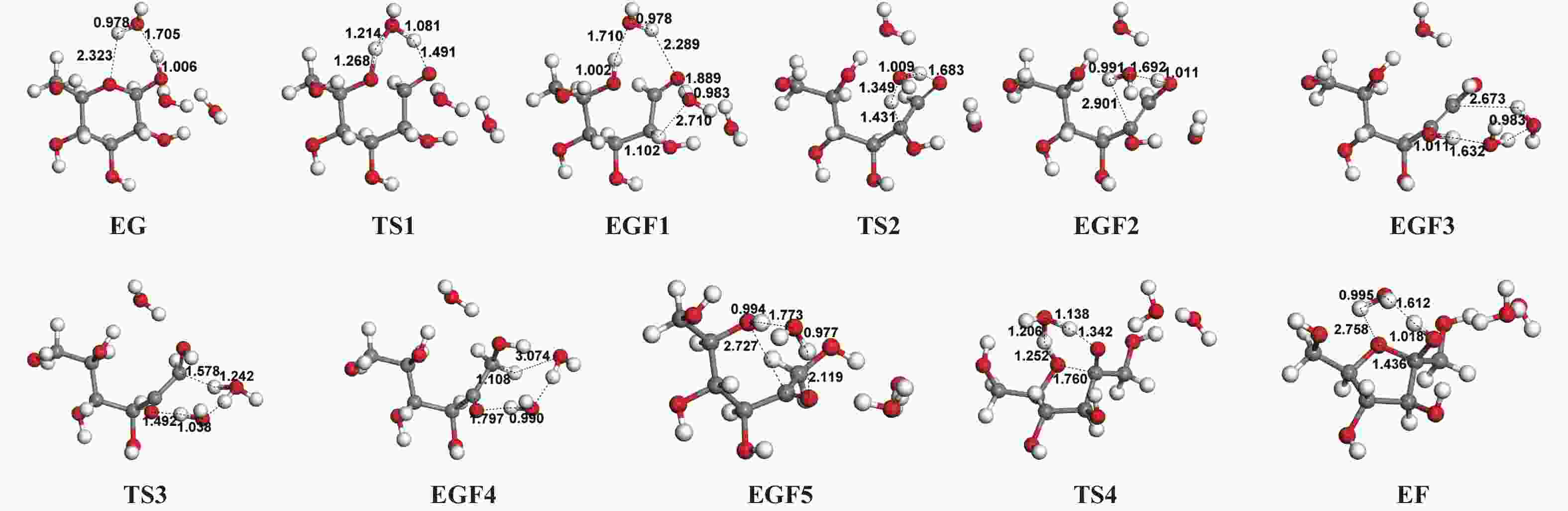
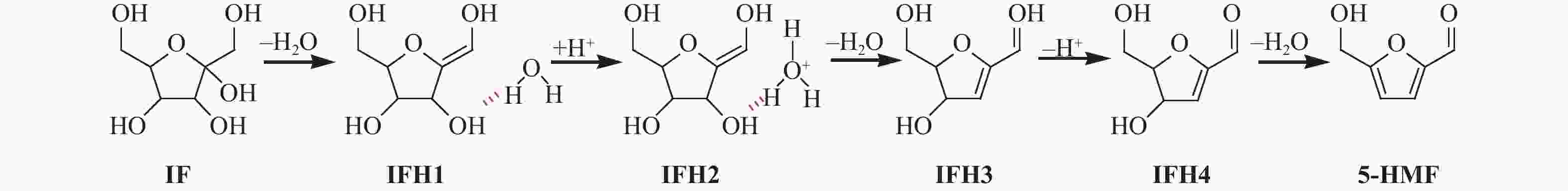
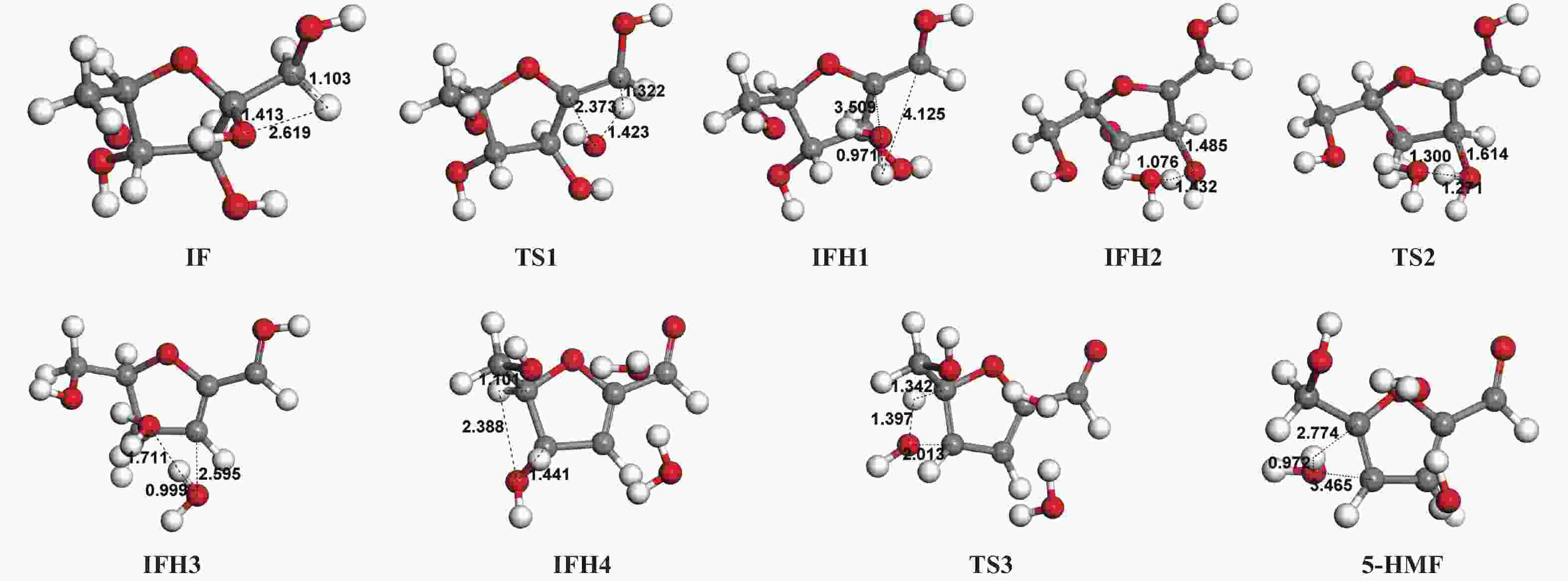
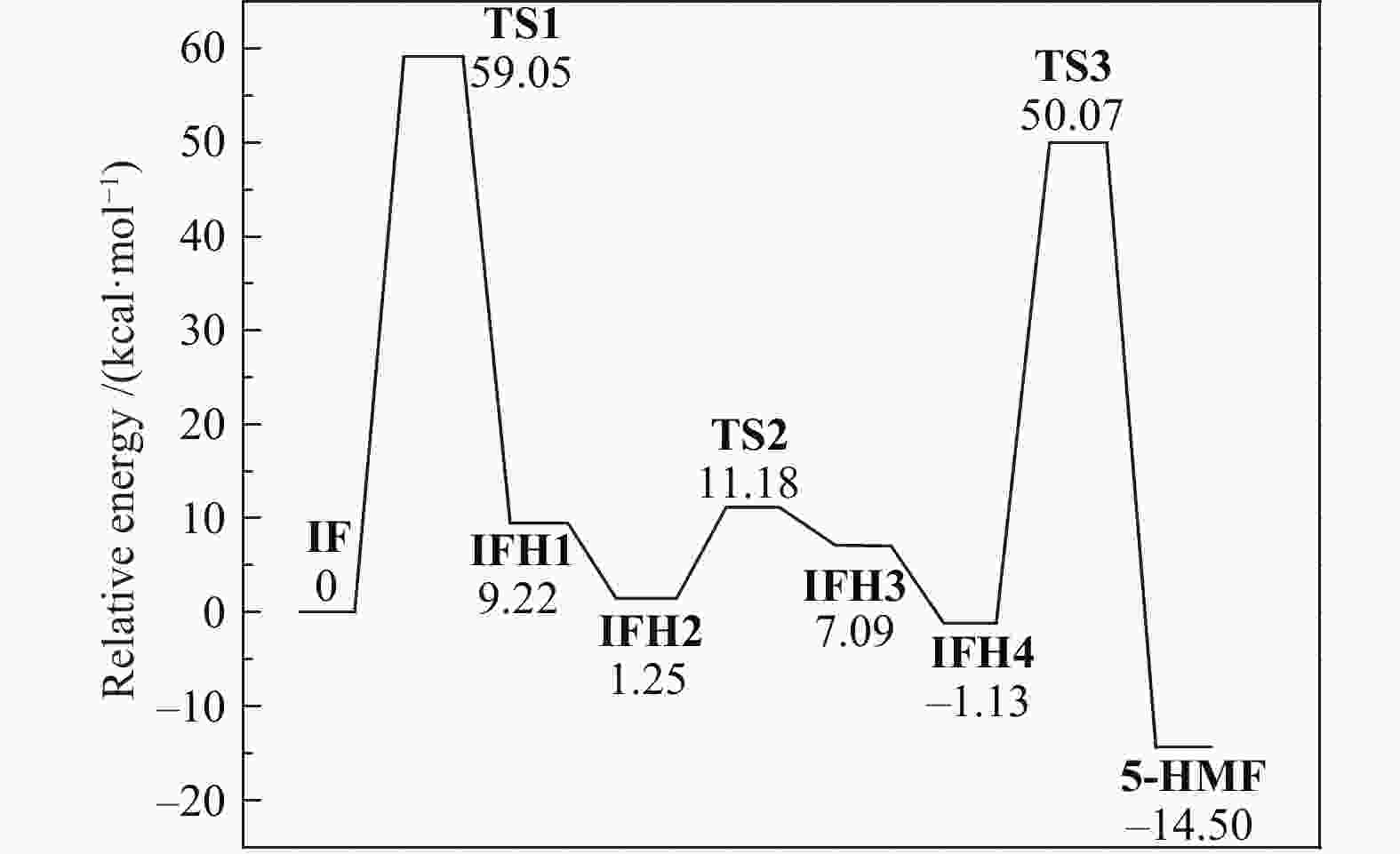
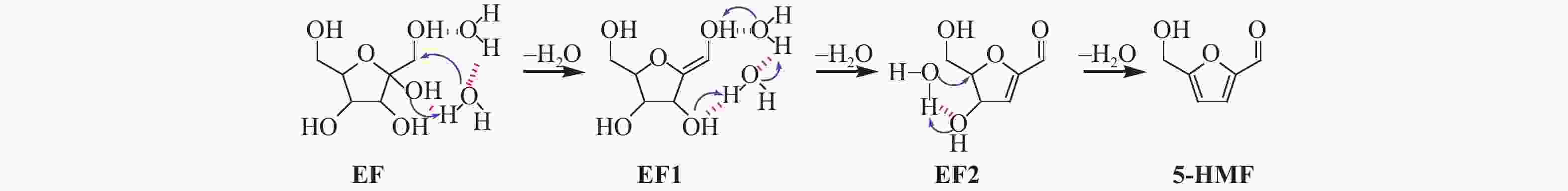
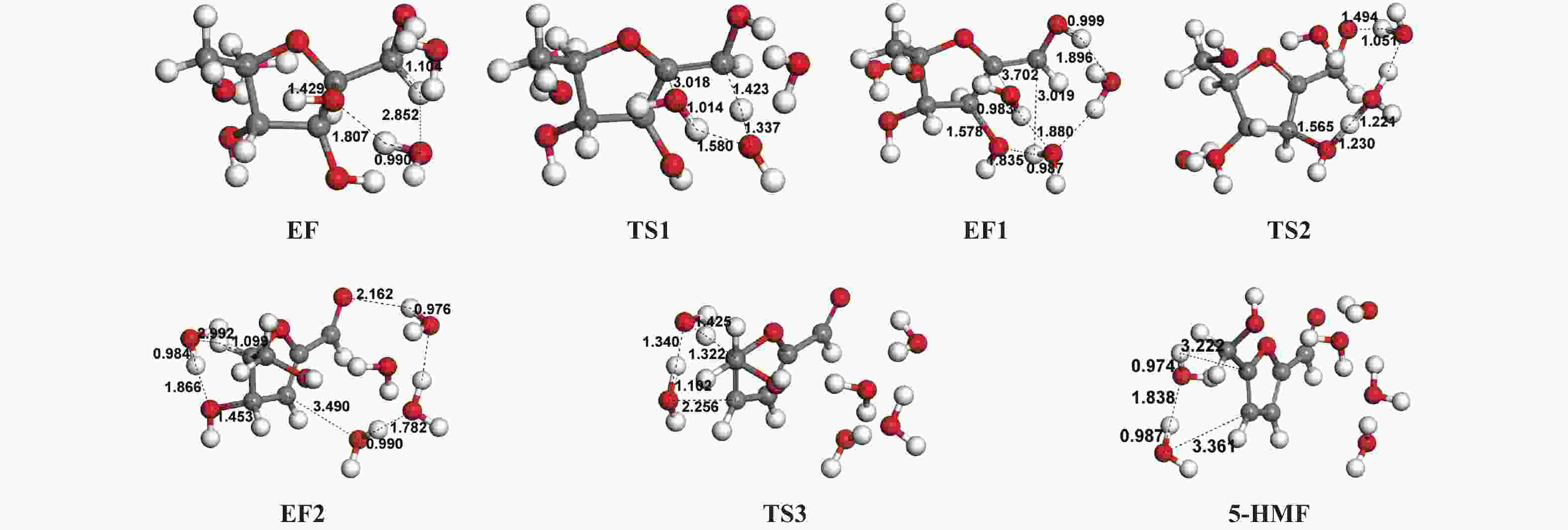
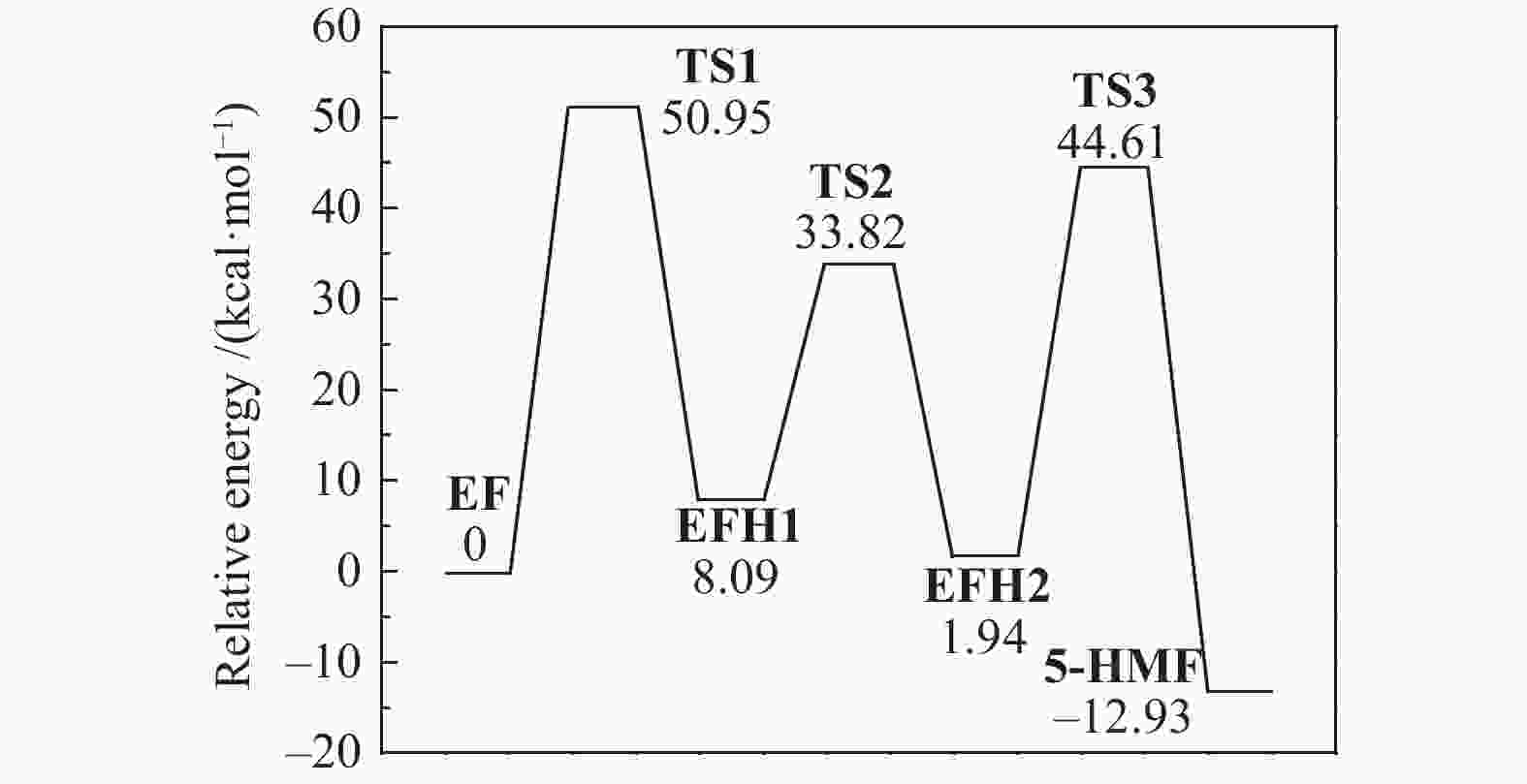
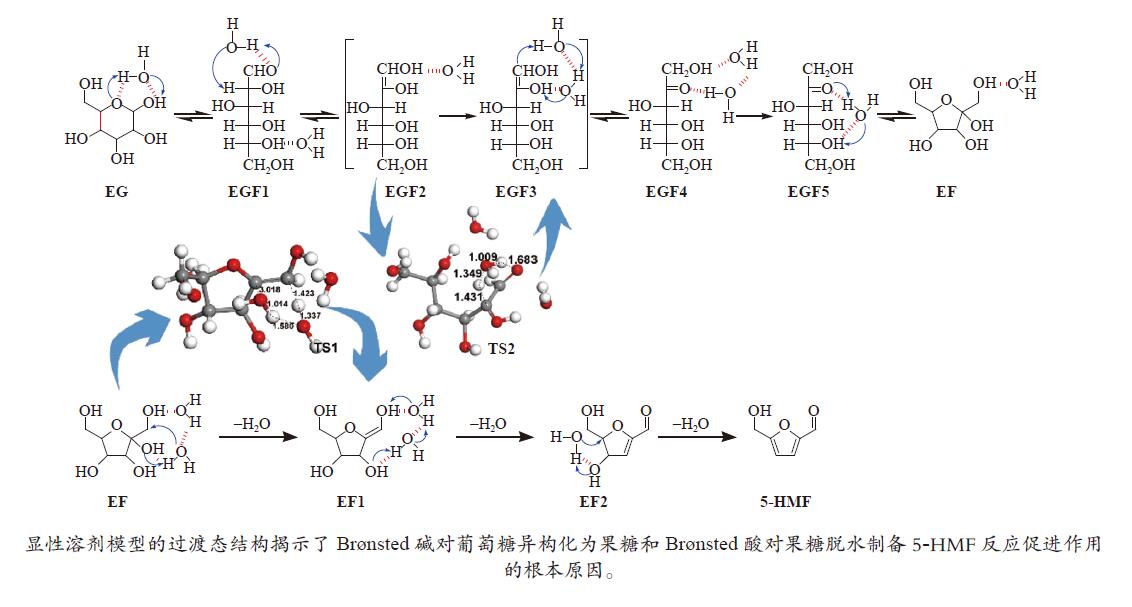
| [1] |
YAN Z, LIAN J, FENG Y, LI M, LONG F, CHENG R, SHI S, GUO H, LU J. A mechanistic insight into glucose conversion in subcritical water: Complex reaction network and the effects of acid-base catalysis[J]. Fuel,2021,289:119969. doi: 10.1016/j.fuel.2020.119969
|
| [2] |
张强, 喻蓬秋, 李林, 乐治平. ZnCl2溶液中微波辅助SnCl4催化纤维素制备5-HMF[J]. 燃料化学学报,2017,45(3):317−322. doi: 10.3969/j.issn.0253-2409.2017.03.009ZHANG Qiang, YU Peng-qiu, LI Lin, LE Zhi-ping. Preparation of 5-HMF from cellulose catalyzed by SnCl4 under microwave in ZnCl2 solution[J]. J Fuel Chem Technol,2017,45(3):317−322. doi: 10.3969/j.issn.0253-2409.2017.03.009
|
| [3] |
郑洪岩, 赵子龙, 肖鲁青山, 赵文诗, 梁栩彬, 薛彦峰, 杨红, 牛宇岚, 朱玉雷. 分子筛催化纤维素和淀粉转化制糠醛[J]. 燃料化学学报: 1−9[2021-06-14]. http://kns.cnki.net/kcms/detail/14.1140.TQ.20210422.2120.003.html.ZHENG Hong-yan, ZHAO Zi-long, XIAO Lu-qing-shan, ZHAO Wen-shi, LIANG Xu-bin, XUE Yan-feng, YANG Hong, NIU Yu-lan, ZHU Yu-lei. Catalytic conversion of cellulose and starch to furfural over zeolites[J]. J Fuel Chem Technol, 1−9[2021-06-14]. http://kns.cnki.net/kcms/detail/14.1140.TQ.20210422.2120.003.html.
|
| [4] |
VAN PUTTEN R-J, VAN DER WAAL J C, DE JONG E, RASRENDRA C B, HEERES H J, DE VRIES J G. Hydroxymethylfurfural, a versatile platform chemical made from renewable resources[J]. Chem Rev,2013,113(3):1499−1597. doi: 10.1021/cr300182k
|
| [5] |
PIDKO E A, DEGIRMENCI V, VAN SANTEN R A, HENSEN E J M. Glucose activation by transient Cr2+ Dimers[J]. Angew Chem Int Ed,2010,49(14):2530−2534. doi: 10.1002/anie.201000250
|
| [6] |
BINDER J B, CEFALI A V, BLANK J J, RAINES R T. Mechanistic insights on the conversion of sugars into 5-hydroxymethylfurfural[J]. Energy Environ Sci,2010,3(6):765−771. doi: 10.1039/b923961h
|
| [7] |
LIU C, CARRAHER J M, SWEDBERG J L, HERNDON C R, FLEITMAN C N, TESSONNIER J P. Selective base-catalyzed isomerization of glucose to fructose[J]. ACS Catal,2014,4(12):4295−4298. doi: 10.1021/cs501197w
|
| [8] |
张雄, 徐志祥, 李雪辉, 关建郁, 龙金星. 葡萄糖化学催化异构制备果糖研究进展[J]. 化工进展,2017,36(12):4575−4585.ZHANG Xiong, XU Zhi-xiang, LI Xue-hui, GUAN Jian-yu, LONG Jin-xing. Chemical isomerization of glucose into fructose[J]. Chem Ind Eng Prog,2017,36(12):4575−4585.
|
| [9] |
QI T, HE M-F, ZHU L-F, LYU Y-J, YANG H-Q, HU C-W. Cooperative catalytic performance of lewis and brønsted acids from AlCl3 salt in aqueous solution toward glucose-to-fructose isomerization[J]. J Phys Chem C,2019,123(8):4879−4891. doi: 10.1021/acs.jpcc.8b11773
|
| [10] |
张颖诗, 王艳, 万金泉, 马邕文, 连洁. 基于固体酸的纤维素非均相催化糖化的研究进展[J]. 化工进展,2014,33(11):2947−2955.ZHANG Ying-shi, WANG Yan, WAN Jin-quan, MA Yong-wen, LIAN Jie. Heterogeneous saccharification of cellulose by solid acid[J]. Chem Ind Eng Prog,2014,33(11):2947−2955.
|
| [11] |
YABUSHITA M, SHIBAYAMA N, NAKAJIMA K, FUKUOKA A. Selective glucose-to-fructose isomerization in ethanol catalyzed by hydrotalcites[J]. ACS Catal,2019,9(3):2101−2109. doi: 10.1021/acscatal.8b05145
|
| [12] |
唐伟强, 谢鹏, 徐小飞, 赵双良. 反应密度泛函理论的构建与初步应用[J]. 化工学报,2021,72(2):633−652.TANG Wei-qiang, XIE Peng, XU Xiao-fei, ZHAO Shuang-liang. Development and applications of reaction density functional theory[J]. CIESC J,2021,72(2):633−652.
|
| [13] |
周婷婷, 刘霞, 叶子航, 周奕鹏, 杨雅淇, 徐清. 三聚氯氰催化及溶剂效应实现温和高效的酮肟贝克曼重排反应[J]. 有机化学,2020,40:1−11. doi: 10.6023/cjoc201907028ZHOU Ting-ting, LIU Xia, YE Zi-hang, ZHOU Yi-peng, YANG Ya-qia, XU Qing. Cyanuric chloride catalysis and solvent effect leading to a mild and efficient Beckmann rearrangement of ketoximes[J]. Chin J Org Chem,2020,40:1−11. doi: 10.6023/cjoc201907028
|
| [14] |
宋英健, 崔晓静, 邓天昇, 秦张峰, 樊卫斌. 液相体系Ru-Co3O4催化CO2加氢制甲烷过程中的溶剂效应研究[J]. 燃料化学学报,2021,49(2):178−185. doi: 10.1016/S1872-5813(21)60013-0SONG Ying-jian, CUI Xiao-jing, DENG Tian-sheng, QIN Zhang-feng, FAN Wei-bin. Solvent effect on the activity of Ru-Co3O4 catalyst for liquid-phase hydrogenation of CO2 into methane[J]. J Fuel Chem Technol,2021,49(2):178−185. doi: 10.1016/S1872-5813(21)60013-0
|
| [15] |
SHIGEMOTO I, KAWAKAMI T, TAIKO H, OKUMURA M. A quantum chemical study on the thermal degradation reaction of polyesters[J]. Polym Degrad Stab,2012,97(6):941−947. doi: 10.1016/j.polymdegradstab.2012.03.020
|
| [16] |
CARTA D, CAO G, D’ANGELI C. Chemical recycling of poly(ethylene terephthalate) (pet) by hydrolysis and glycolysis[J]. Environ Sci Pollut Res,2003,10(6):390−394. doi: 10.1065/espr2001.12.104.8
|
| [17] |
MCMAHON W, BIRDSALL H A, JOHNSON G R, CAMILLI C T. Degradation studies of polyethylene terephthalate[J]. J Chem Eng Data,1959,4(1):57−79. doi: 10.1021/je60001a009
|
| [18] |
PICKETT J E, COYLE D J. Hydrolysis kinetics of condensation polymers under humidity aging conditions[J]. Polym Degrad Stab,2013,98(7):1311−1320. doi: 10.1016/j.polymdegradstab.2013.04.001
|
| [19] |
DUBELLEY F, PLANES E, BAS C, PONS E, YRIEIX B, FLANDIN L. Predictive durability of polyethylene terephthalate toward hydrolysis over large temperature and relative humidity ranges[J]. Polymer,2018,142:285−292. doi: 10.1016/j.polymer.2018.03.043
|
| [20] |
RAVENS D A S, WARD I M. Chemical reactivity of polyethylene terephthalate. Hydrolysis and esterification reactions in the solid phase[J]. Trans Faraday Soc,1961,57(0):150−159.
|
| [21] |
YOSHIOKA T, OKAYAMA N, OKUWAKI A. Kinetics of hydrolysis of PET powder in nitric acid by a modified shrinking-core model[J]. Ind Eng Chem Res,1998,37(2):336−340. doi: 10.1021/ie970459a
|
| [22] |
YAN Z, LIAN J, LI M, MENG L, ZHANG Y, GE C, LU J. Deeper insight into hydrolysis mechanisms of polyester/cotton blended fabrics for separation by explicit solvent models[J]. Int J Biol Macromol,2020,154:596−605. doi: 10.1016/j.ijbiomac.2020.03.130
|
| [23] |
WANG L-P, MARTINEZ T J, PANDE V S. Building force fields: An automatic, systematic, and reproducible approach[J]. J Phys Chem Lett,2014,5(11):1885−1891. doi: 10.1021/jz500737m
|
| [24] |
CHEN J, BROOKS C L, KHANDOGIN J. Recent advances in implicit solvent-based methods for biomolecular simulations[J]. Curr Opin Struc Biol,2008,18(2):140−148. doi: 10.1016/j.sbi.2008.01.003
|
| [25] |
BR LL D, KAUL C, KR MER A, KRAMMER P, RICHTER T, JUNG M, VOGEL H, ZEHNER P. Chemistry in supercritical water[J]. Angew Chem Int Ed,1999,38(20):2998−3014. doi: 10.1002/(SICI)1521-3773(19991018)38:20<2998::AID-ANIE2998>3.0.CO;2-L
|
| [26] |
ZHAO Y, LU W-J, WANG H-T, LI D. Combined supercritical and subcritical process for cellulose hydrolysis to fermentable hexoses[J]. Environ Sci Technol,2009,43(5):1565−1570. doi: 10.1021/es803122f
|
| [27] |
赵岩, 李冬, 陆文静, 王洪涛, 朴雪松. 纤维素超临界水预处理与水解研究[J]. 化学学报,2008,66(20):2295−2301. doi: 10.3321/j.issn:0567-7351.2008.20.018ZHAO Yan, LI Dong, LU Wen-jing, WANG Hong-tao, PIAO Xue-song. Supercritical pretreatment and hydrolyzation of cellulose[J]. Acta Chim Sin,2008,66(20):2295−2301. doi: 10.3321/j.issn:0567-7351.2008.20.018
|
| [28] |
PROMDEJ C, MATSUMURA Y. Temperature effect on hydrothermal decomposition of glucose in sub- and supercritical water[J]. Ind Eng Chem Res,2011,50(14):8492−8497. doi: 10.1021/ie200298c
|
| [29] |
SASAKI M, FANG Z, FUKUSHIMA Y, ADSCHIRI T, ARAI K. Dissolution and hydrolysis of cellulose in subcritical and supercritical water[J]. Ind Eng Chem Res,2000,39(8):2883−2890. doi: 10.1021/ie990690j
|
| [30] |
YU Y, LOU X, WU H. Some recent advances in hydrolysis of biomass in hot-compressed water and its comparisons with other hydrolysis methods[J]. Energy Fuels,2008,22(1):46−60. doi: 10.1021/ef700292p
|
| [31] |
KRUSE A, DINJUS E. Hot compressed water as reaction medium and reactant: Properties and synthesis reactions[J]. J Supercrit Fluids,2007,39(3):362−380. doi: 10.1016/j.supflu.2006.03.016
|
| [32] |
GRIMME S, ANTONY J, EHRLICH S, KRIEG H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu[J]. J Chem Phys,2010,132(15):154104−154119. doi: 10.1063/1.3382344
|
| [33] |
DELLEY B. An all-electron numerical method for solving the local density functional for polyatomic molecules[J]. J Chem Phys,1990,92(1):508−517. doi: 10.1063/1.458452
|
| [34] |
DELLEY B. Fast calculation of electrostatics in crystals and large molecules[J]. J Phys Chem,1996,100(15):6107−6110. doi: 10.1021/jp952713n
|
| [35] |
PEVERATI R, BALDRIDGE K K. Implementation and optimization of DFT-D/COSab with respect to basis set and functional: Application to polar processes of furfural derivatives in solution[J]. J Chem Theory Comput,2009,5(10):2772−2786. doi: 10.1021/ct900363n
|
| [36] |
SIROHIWAL A, NEESE F, PANTAZIS D A. Microsolvation of the redox-active Tyrosine-D in photosystem II: correlation of energetics with EPR spectroscopy and oxidation-induced proton transfer[J]. J Am Chem Soc,2019,141(7):3217−3231. doi: 10.1021/jacs.8b13123
|
| [37] |
AKIYA N, SAVAGE P E. Roles of water for chemical reactions in high-temperature water[J]. Chem Rev,2002,102(8):2725−2750. doi: 10.1021/cr000668w
|
| [38] |
HALGREN T A, LIPSCOMB W N. The synchronous-transit method for determining reaction pathways and locating molecular transition states[J]. Chem Phys Lett,1977,49(2):225−232. doi: 10.1016/0009-2614(77)80574-5
|
| [39] |
PENG C, BERNHARD SCHLEGEL H. Combining synchronous transit and quasi-newton methods to find transition states[J]. Isr J Chem,1993,33(4):449−454. doi: 10.1002/ijch.199300051
|
| [40] |
PERDEW J P, BURKE K, ERNZERHOF M. Generalized gradient approximation made simple[J]. Phys Rev Lett,1996,77(18):3865−3868. doi: 10.1103/PhysRevLett.77.3865
|




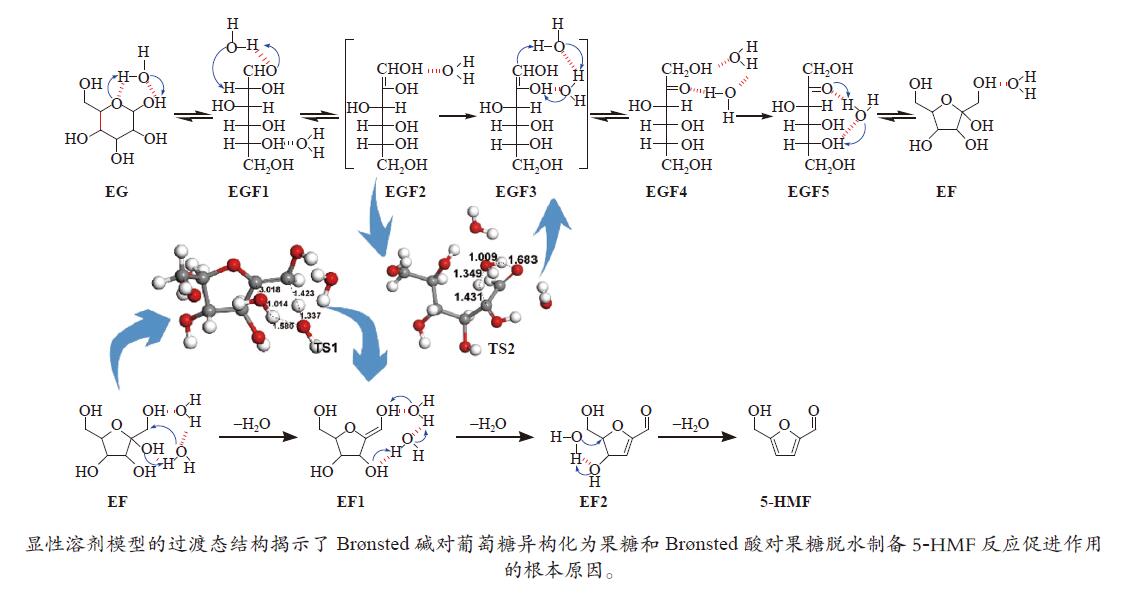
 下载:
下载: