The effect of Bi content on the photocatalytic performance of bismuth oxybromides
-
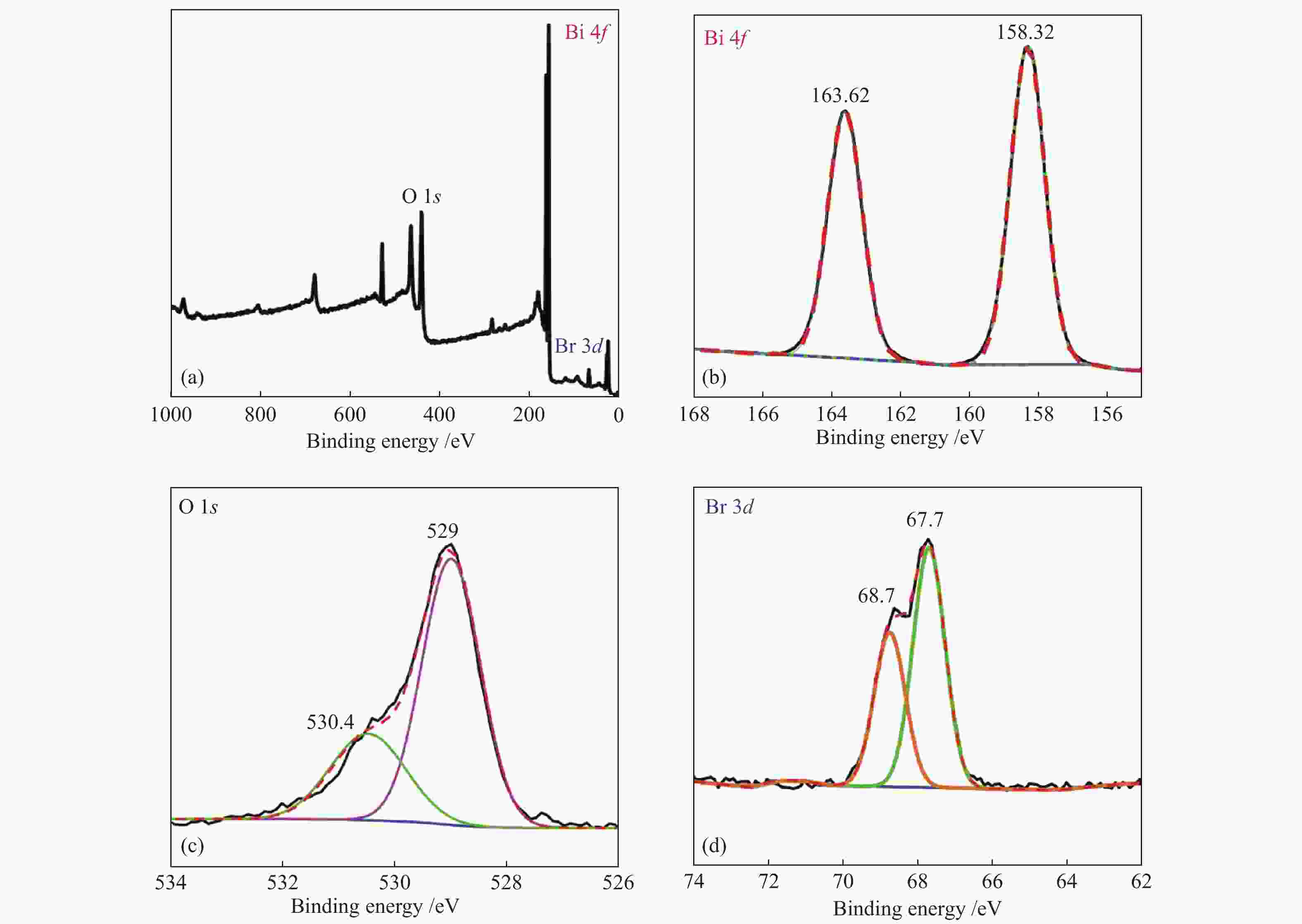
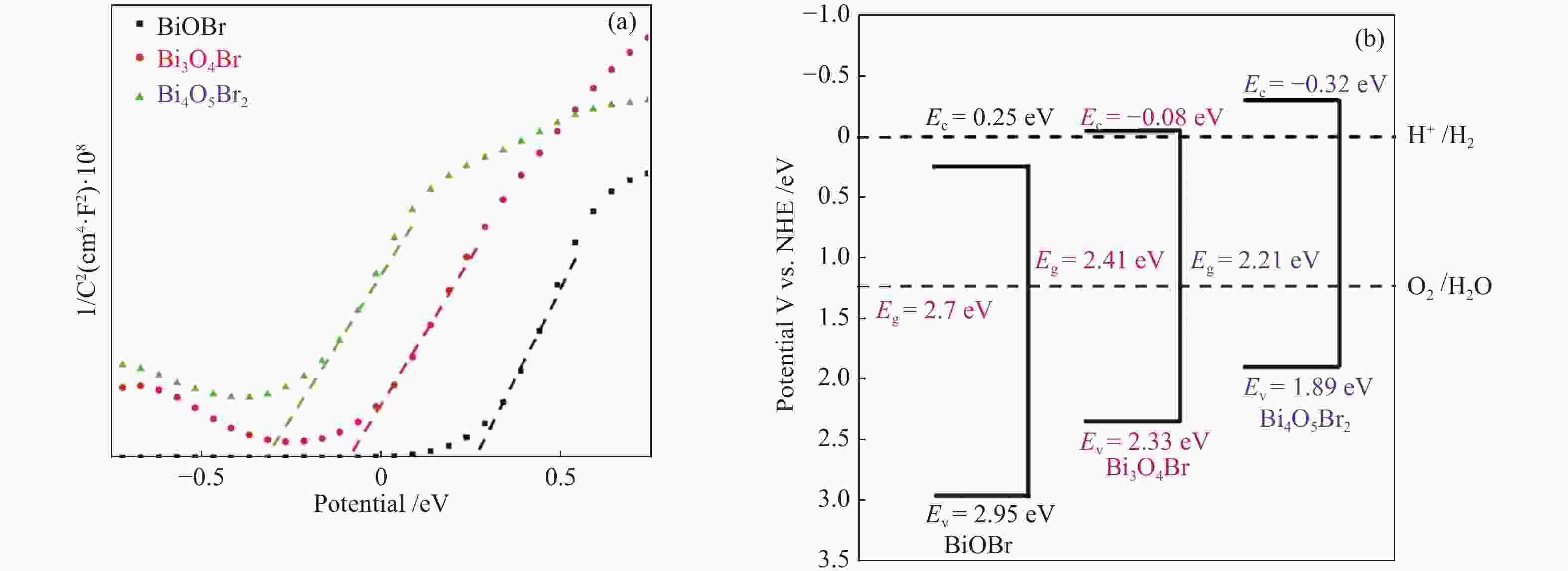
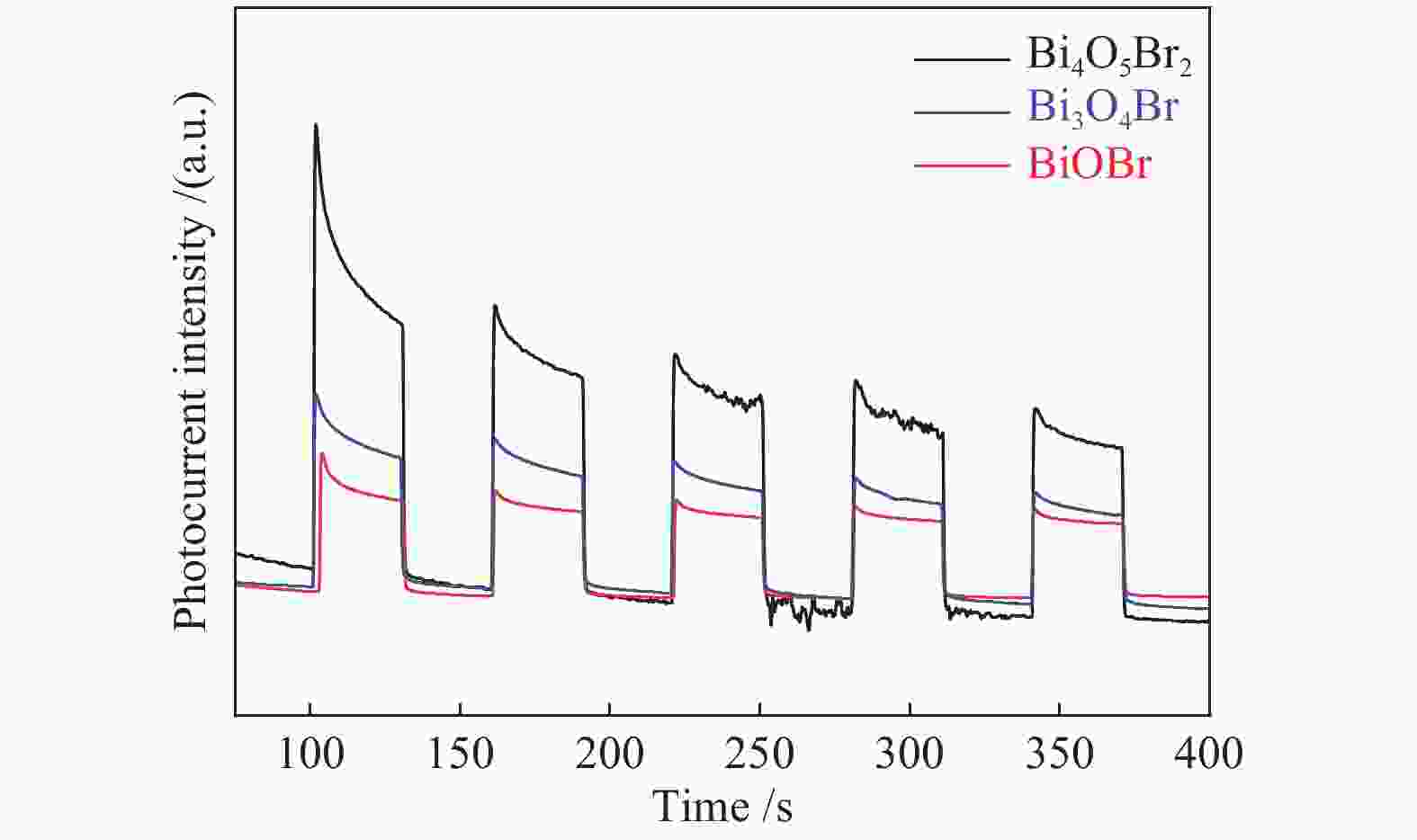
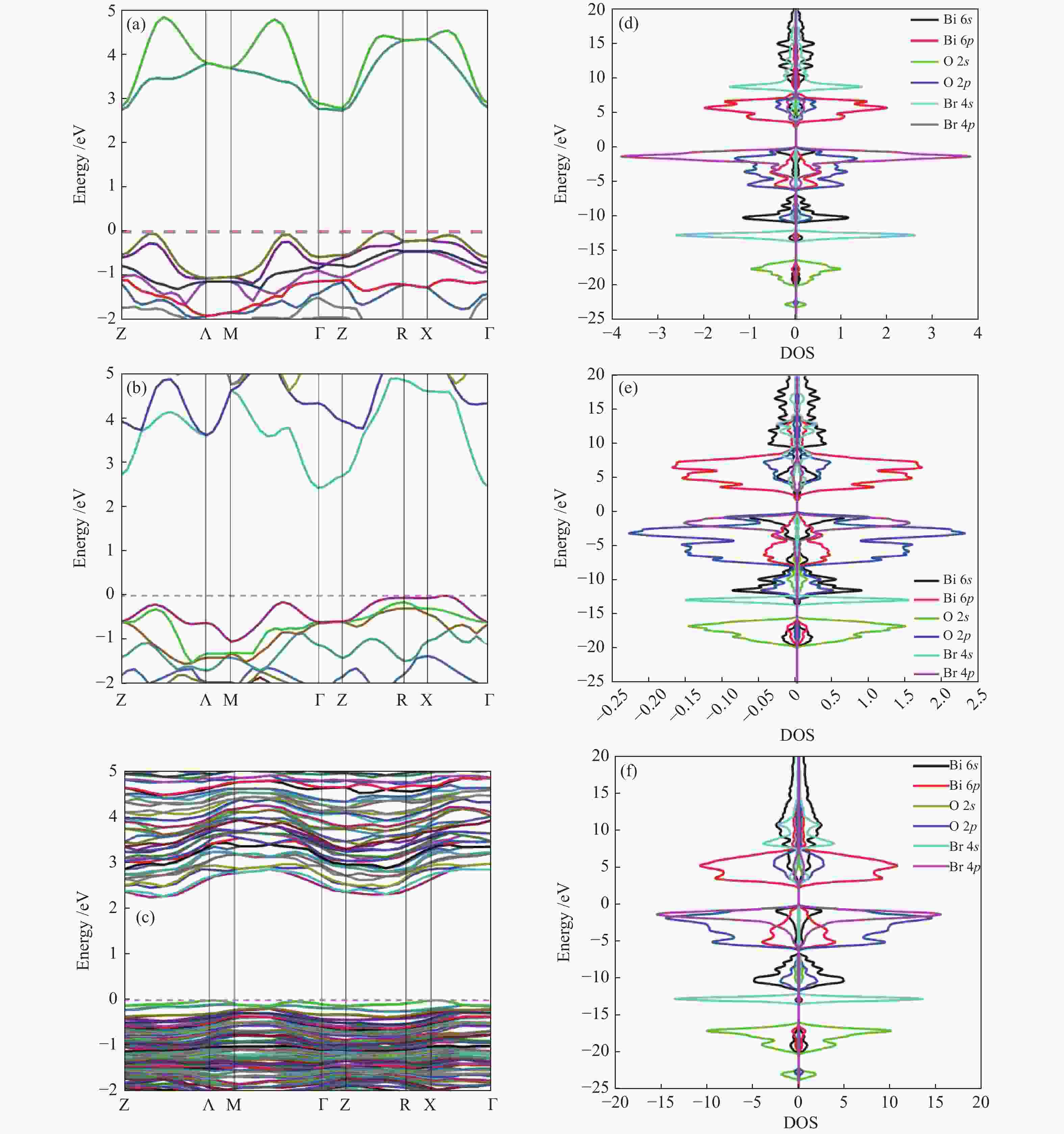
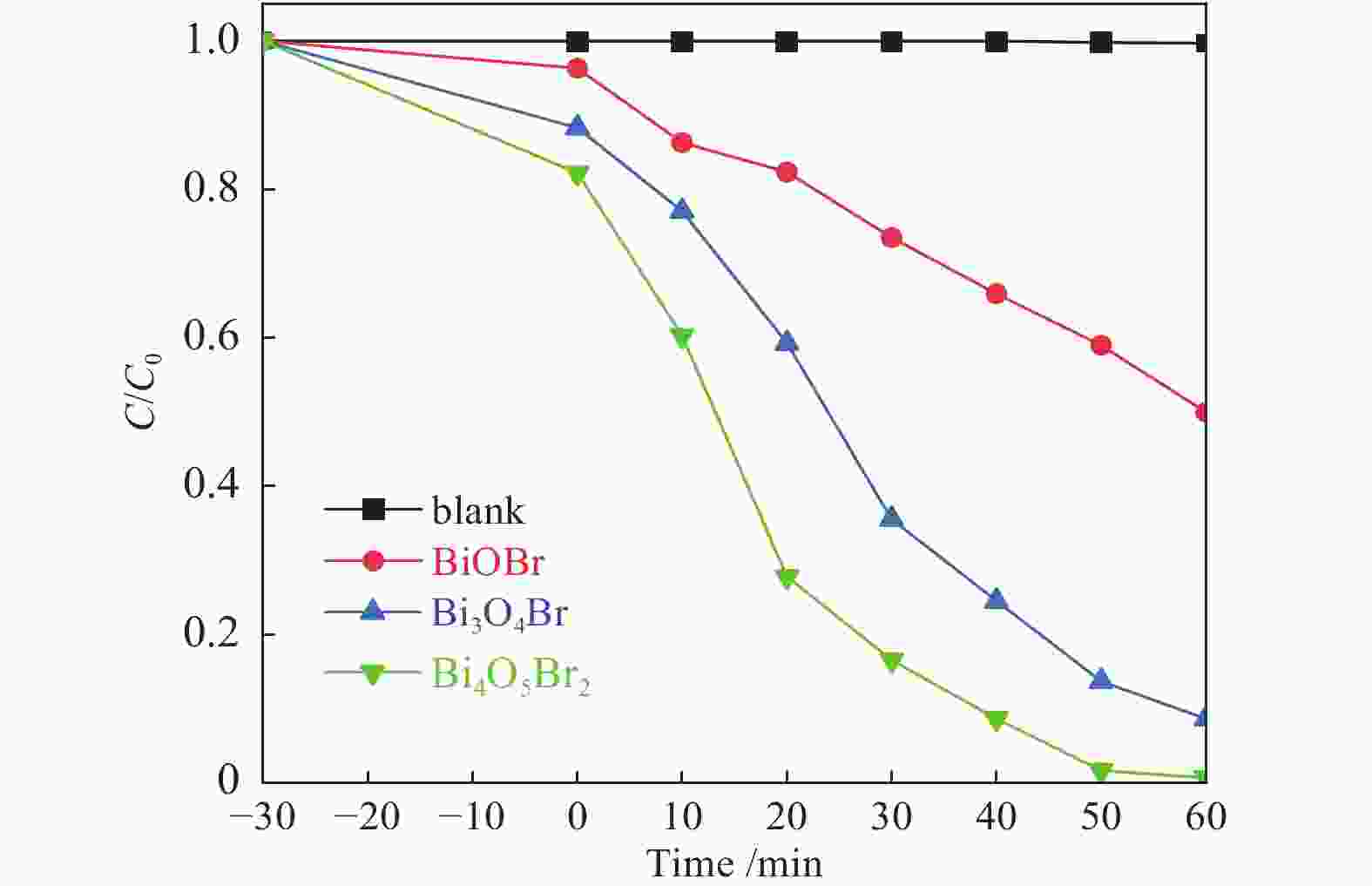
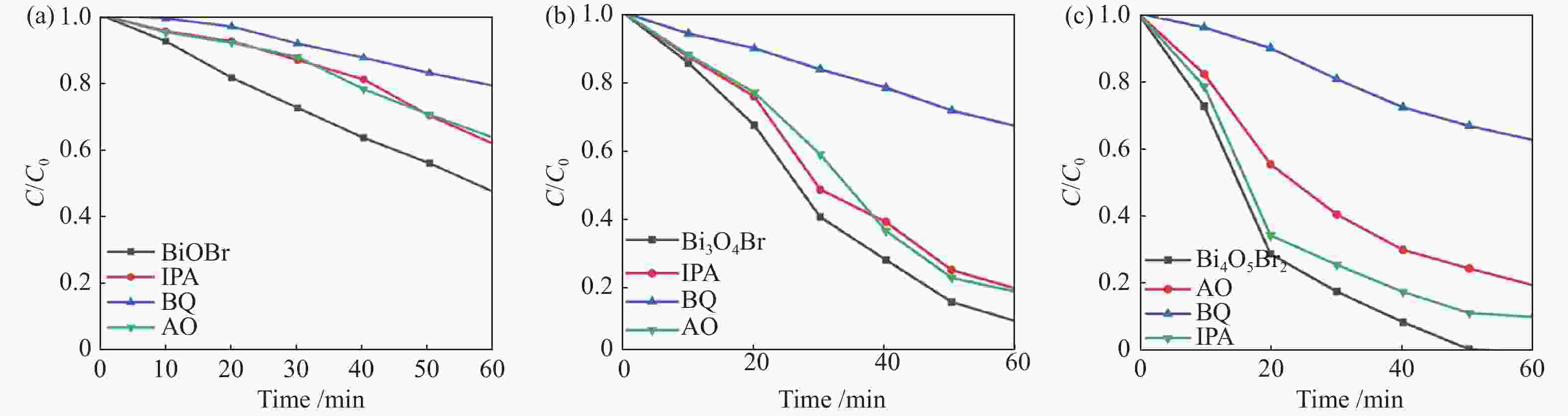
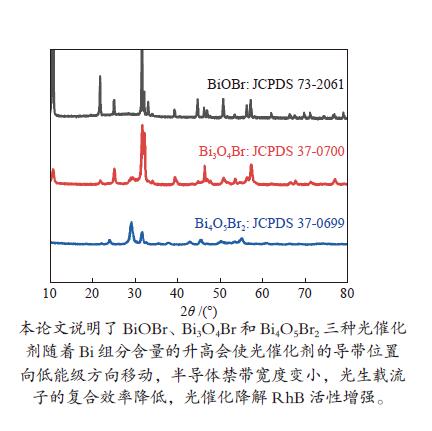
摘要: 采用水热法和溶剂热法制备BiOBr、Bi3O4Br和Bi4O5Br2三种光催化剂,通过X射线衍射(XRD)、扫描电子显微镜(SEM)、X射线光电子能谱(XPS)与紫外-可见漫反射光谱(UV-vis DRS)表征光催化剂的晶体结构、表面形貌和光学性能,利用密度泛函理论计算光催化剂的能带结构和态密度,在可见光照射下,通过降解RhB考察光催化剂的活性。结果表明,Bi的含量会影响光催化剂的导带位置和禁带宽度,Bi4O5Br2光催化剂降解效果最好,能够在50 min内将RhB完全降解。自由基捕获实验证明,超氧自由基(·
${\rm{O}}_2^- $ )是光催化降解RhB的主要活性物质。Abstract: BiOBr, Bi3O4Br and Bi4O5Br2 were prepared by hydrothermal and solvothermal methods. Their structural composition, surface morphology, chemical states and optical properties were characterized by XRD, SEM, XPS and UV-vis. The band structure and density of states of the photocatalysts were calculated by density functional theory (DFT). The photocatalytic activity was evaluated by degradation of RhB. The results show that band gap and the position of conduction band is affected by Bi content. The Bi4O5Br2 photocatalyst can completely degrade RhB in 50 min. Radical-trapping experiments proves that ·$ {\rm{O}}_2^- $ is the main active species in photocatalytic degradation of RhB.-
Key words:
- BiOBr /
- Bi content /
- DFT /
- photocatalytic performance
1) #: 共同第一作者 -
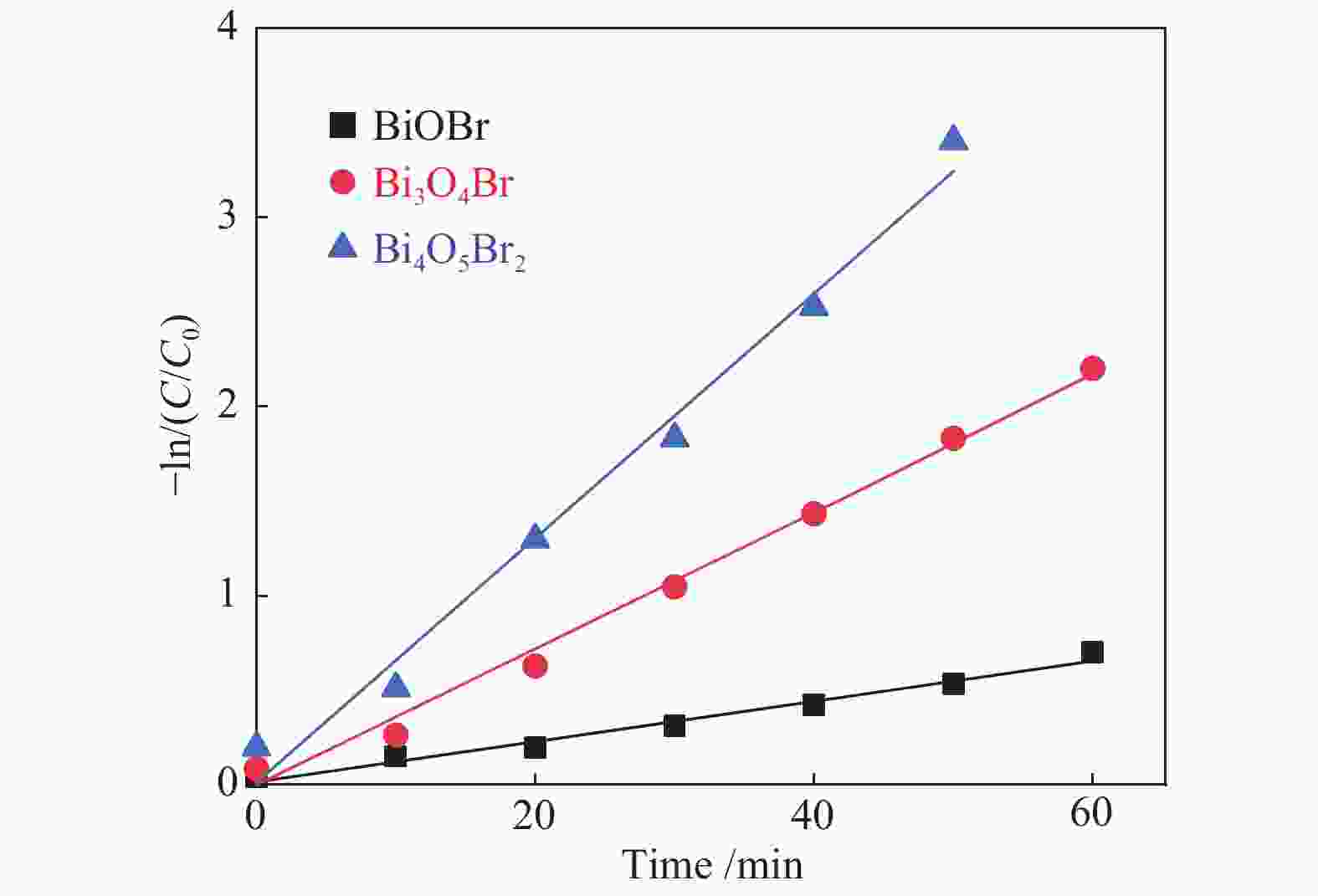
表 1 三种光催化剂的一级反应动力学参数
Table 1 First-order reaction kinetic parameters of three samples
Sample BiOBr Bi3O4Br Bi4O5Br2 k/min−1 0.010 0.0367 0.0642 R2 0.979 0.991 0.982 -
[1] VAYA D, SUROLIA P K. Semiconductor based photocatalytic degradation of pesticides: An overview[J]. Environ Technol Innovation,2020,20:101128. doi: 10.1016/j.eti.2020.101128 [2] IKREEDEEGH RR, TAHIR M. A critical review in recent developments of metal-organic-frameworks (MOFs) with band engineering alteration for photocatalytic CO2 reduction to solar fuels[J]. J CO2 Util,2021,43:101381. doi: 10.1016/j.jcou.2020.101381 [3] 胡庆松. 卤氧铋复合催化剂构建及其增强光催化去除水中污染物研究[D]. 上海: 华东师范大学, 2020.HU Qing-song. Construction of bismuth oxyhalide composite catalysts with enhanced photocatalytic activity for the removal of contaminants[D]. Shanghai: East China Normal University, 2020. [4] SUN J J, LI X Y, ZHAO Q D, LIU B J. Ultrathin nanoflake-assembled hierarchical BiOBr microflower with highly exposed {001} facets for efficient photocatalytic degradation of gaseous ortho-dichlorobenzene[J]. App Catal B: Environ,2021,281:119478. doi: 10.1016/j.apcatb.2020.119478 [5] MAO D J, DING S S, MENG L J, DAI Y X, SUN C, YANG S G, HE H. One-pot microemulsion-mediated synthesis of Bi-rich Bi4O5Br2 with controllable morphologies and excellent visible-light photocatalytic removal of pollutants[J]. App Catal B: Environ,2017,207:153−165. doi: 10.1016/j.apcatb.2017.02.010 [6] TIAN H D, CHENG R R, LIN M H, LI P, LV Y H, RAN S L. Oxygen-vacancy-rich ultrathin BiOBr nonosheets for high-performance supercapacitor electrodes[J]. Inorg Chem Commun,2020,118:108018. doi: 10.1016/j.inoche.2020.108018 [7] BAI Y, YANG P, WANG L, YANG B, XIE H Q, ZHOU Y, YE L Q. Ultrathin Bi4O5Br2 nanosheets for selective photocatalytic CO2 conversion into CO[J]. Chem Eng J,2019,360:473−482. doi: 10.1016/j.cej.2018.12.008 [8] Mao D J, Yuan J L, Qu X L, SUN C, YANG S G, HE H. Size tunable Bi3O4Br hierarchical hollow spheres assembled with {0 0 1}-facets exposed nanosheets for robust photocatalysis against phenolic pollutants[J]. J Catal,2019,369:209−221. doi: 10.1016/j.jcat.2018.11.016 [9] ZHANG H G, WANG W T, FENG LJ, LI C H WANG L. Effect of hydrothermal pH value on composition and morphology of bismuth oxybromide and their photocatalytic performance[J]. J Fuel Chem Technol,2019,47(5):582−589. doi: 10.1016/S1872-5813(19)30026-X [10] ZHANG W B, XIAO X, WU Q F, FAN Q, ZAHNG F C. Facile synthesis of novel Mn-doped Bi4O5Br2 for enhanced photocatalytic NO removal activity[J]. J Alloys Compd,2020,826:154204. doi: 10.1016/j.jallcom.2020.154204 [11] ZHANG Y, YANG W. Comment on ''generalized gradient approximation made simple''[J]. Phys Rev Lett,1998,80(4):891−891. doi: 10.1103/PhysRevLett.80.891 [12] MONKHORST H J, PACK J D. Special points for brillonin-zone integrations[J]. Phys Rev B,1976,13(12):5188−5192. doi: 10.1103/PhysRevB.13.5188 [13] LI K L, LEE WW, LU CS, DAI Y M, CHOU S Y, CHEN S L, LIN H P, CHEN C C. Synthesis of BiOBr, Bi3O4Br, and Bi12O17Br2 by controlled hydrothermal method and their photocatalytic properties[J]. J Taiwan Inst Chem Eng,2014,45(5):2688−2697. doi: 10.1016/j.jtice.2014.04.001 [14] JIN X L, LV C, ZHOU X, XIE H Q, SUN S F, LIU Y, MENG Q Q, CHEN G. A bismuth rich hollow Bi4O5Br2 photocatalyst enables dramatic CO2 reduction activity[J]. Nano Energy,2019,64:103955. doi: 10.1016/j.nanoen.2019.103955 [15] WANG X K, LIU Y X, WANG J N, ZHANG J M, HUANG Y H, WEI X M. Theoretical investigation of the photocatalytic mechanism of single Au adsorption on the Bi4O5Br2 (101) surface[J]. Chem Phys Lett,2020,757(6):137851. [16] GUO N N, CAO Y L, RONG Y L, JIA D Z. Green synthesis of BiOBr modified Bi2O2CO3 nanocomposites with enhanced visible-responsive photocatalytic properties[J]. RSC Adv,2016,6(108):106046. doi: 10.1039/C6RA22385K [17] YANG P, WANG J C, YUE G Z, YANG R Z, ZHAO P X, YANG L J, ZHANG X C, ASTRUC D. Constructing mesoporous g-C3N4/ZnO nanosheets catalyst for enhanced visible-light driven photocatalytic activity[J]. J Photoch Photobio A,2020,388:112169. doi: 10.1016/j.jphotochem.2019.112169 [18] LI R, XIE F X, LIU J X, WANG Y W, WANG Y F, ZHANG X C, FAN C M. Synthesis of Bi4O5Br2 from reorganization of BiOBr and its excellent visible light photocatalytic activity[J]. Dalton Trans,2016,45:9182−9186. doi: 10.1039/C6DT00997B [19] KANAGARAI T, THIRIPURANTHAGAN S. Photocatalytic activities of novel SrTiO3-BiOBr heterojunction catalysts towards the degradation of reactive dyes[J]. App Catal B: Environ,2017,207:218−232. doi: 10.1016/j.apcatb.2017.01.084 [20] AO Y H, WANG K D, WANG P F, WANG C, HOU J. Synthesis of novel 2D-2D p-n heterojunction BiOBr/La2Ti2O7 composite photocatalyst with enhanced photocatalytic performance under both UV and visible light irradiation[J]. App Catal B: Environ,2016,194:157−168. doi: 10.1016/j.apcatb.2016.04.050 -






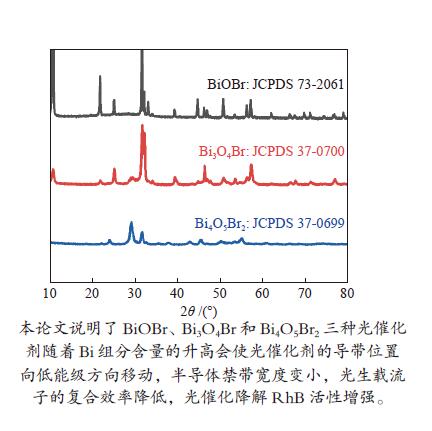
 下载:
下载: